
In vitro studies are performed with microorganisms, cells, or biological molecules outside their normal biological context. Colloquially called "test-tube experiments", these studies in biology and its subdisciplines are traditionally done in labware such as test tubes, flasks, Petri dishes, and microtiter plates. Studies conducted using components of an organism that have been isolated from their usual biological surroundings permit a more detailed or more convenient analysis than can be done with whole organisms; however, results obtained from in vitro experiments may not fully or accurately predict the effects on a whole organism. In contrast to in vitro experiments, in vivo studies are those conducted in living organisms, including humans, known as clinical trials, and whole plants.
In toxicology, the median lethal dose, LD50 (abbreviation for "lethal dose, 50%"), LC50 (lethal concentration, 50%) or LCt50 is a toxic unit that measures the lethal dose of a given substance. The value of LD50 for a substance is the dose required to kill half the members of a tested population after a specified test duration. LD50 figures are frequently used as a general indicator of a substance's acute toxicity. A lower LD50 is indicative of higher toxicity.

A toxin is a naturally occurring poison produced by metabolic activities of living cells or organisms. They occur especially as proteins, often conjugated. The term was first used by organic chemist Ludwig Brieger (1849–1919) and is derived from the word "toxic".

Toxicology is a scientific discipline, overlapping with biology, chemistry, pharmacology, and medicine, that involves the study of the adverse effects of chemical substances on living organisms and the practice of diagnosing and treating exposures to toxins and toxicants. The relationship between dose and its effects on the exposed organism is of high significance in toxicology. Factors that influence chemical toxicity include the dosage, duration of exposure, route of exposure, species, age, sex, and environment. Toxicologists are experts on poisons and poisoning. There is a movement for evidence-based toxicology as part of the larger movement towards evidence-based practices. Toxicology is currently contributing to the field of cancer research, since some toxins can be used as drugs for killing tumor cells. One prime example of this is ribosome-inactivating proteins, tested in the treatment of leukemia.

A poison is any chemical substance that is harmful or lethal to living organisms. The term is used in a wide range of scientific fields and industries, where it is often specifically defined. It may also be applied colloquially or figuratively, with a broad sense.

Toxicity is the degree to which a chemical substance or a particular mixture of substances can damage an organism. Toxicity can refer to the effect on a whole organism, such as an animal, bacterium, or plant, as well as the effect on a substructure of the organism, such as a cell (cytotoxicity) or an organ such as the liver (hepatotoxicity). Sometimes the word is more or less synonymous with poisoning in everyday usage.
In vitro toxicity testing is the scientific analysis of the toxic effects of chemical substances on cultured bacteria or mammalian cells. In vitro testing methods are employed primarily to identify potentially hazardous chemicals and/or to confirm the lack of certain toxic properties in the early stages of the development of potentially useful new substances such as therapeutic drugs, agricultural chemicals and food additives.

Piperonyl butoxide (PBO) is a pale yellow to light brown liquid organic compound used as an adjuvant component of pesticide formulations for synergy. That is, despite having no pesticidal activity of its own, it enhances the potency of certain pesticides such as carbamates, pyrethrins, pyrethroids, and rotenone. It is a semisynthetic derivative of safrole and is produced from the condensation of the sodium salt of 2-(2-butoxyethoxy) ethanol and the chloromethyl derivative of hydrogenated safrole (dihydrosafrole); or through 1,2-Methylenedioxybenzene.

Hormesis is a two-phased dose-response relationship to an environmental agent whereby low-dose amounts have a beneficial effect and high-dose amounts are either inhibitory to function or toxic. Within the hormetic zone, the biological response to low-dose amounts of some stressors is generally favorable. An example is the breathing of oxygen, which is required in low amounts via respiration in living animals, but can be toxic in high amounts, even in a managed clinical setting.
In toxicology, the lethal dose (LD) is an indication of the lethal toxicity of a given substance or type of radiation. Because resistance varies from one individual to another, the "lethal dose" represents a dose at which a given percentage of subjects will die. The lethal concentration is a lethal dose measurement used for gases or particulates. The LD may be based on the standard person concept, a theoretical individual that has perfectly "normal" characteristics, and thus not apply to all sub-populations.

Aquatic toxicology is the study of the effects of manufactured chemicals and other anthropogenic and natural materials and activities on aquatic organisms at various levels of organization, from subcellular through individual organisms to communities and ecosystems. Aquatic toxicology is a multidisciplinary field which integrates toxicology, aquatic ecology and aquatic chemistry.
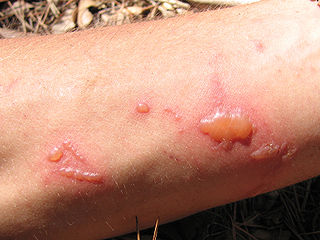
Phototoxicity, also called photoirritation, is a chemically induced skin irritation, requiring light, that does not involve the immune system. It is a type of photosensitivity.

Phosmet is a phthalimide-derived, non-systemic, organophosphate insecticide used on plants and animals. It is mainly used on apple trees for control of codling moth, though it is also used on a wide range of fruit crops, ornamentals, and vines for the control of aphids, suckers, mites, and fruit flies.
Safety pharmacology is a branch of pharmacology specialising in detecting and investigating potential undesirable pharmacodynamic effects of new chemical entities (NCEs) on physiological functions in relation to exposure in the therapeutic range and above.

Developmental toxicity is any developmental malformation that is caused by the toxicity of a chemical or pathogen. It is the structural or functional alteration, reversible or irreversible, which interferes with homeostasis, normal growth, differentiation, development or behavior. Developmental toxicity is caused by environmental insult, which includes drugs, alcohol, diet, toxic chemicals, and physical factors.
In vitro to in vivo extrapolation (IVIVE) refers to the qualitative or quantitative transposition of experimental results or observations made in vitro to predict phenomena in vivo, biological organisms.
Occupational toxicology is the application of toxicology to chemical hazards in the workplace. It focuses on substances and conditions that people may be exposed to in workplaces, including inhalation and dermal exposures, which are most prevalent when discussing occupational toxicology. These environmental and individual exposures can impact health, and there is a focus on identifying early adverse affects that are more subtle than those presented in clinical medicine.

Poisoning is the harmful effect which occurs when toxic substances are introduced into the body. The term "poisoning" is a derivative of poison, a term describing any chemical substance that may harm or kill a living organism upon ingestion. Poisoning can be brought on by swallowing, inhaling, injecting or absorbing toxins through the skin. Toxicology is the practice and study of symptoms, mechanisms, diagnoses, and treatments correlated to poisoning.
A bioassay is an analytical method to determine the potency or effect of a substance by its effect on living animals or plants, or on living cells or tissues. A bioassay can be either quantal or quantitative, direct or indirect. If the measured response is binary, the assay is quantal; if not, it is quantitative.
Threshold dose is the minimum dose of drug that triggers minimal detectable biological effect in an animal. At extremely low doses, biological responses are absent for some of the drugs. The increase in dose above threshold dose induces an increase in the percentage of biological responses. Several benchmarks have been established to describe the effects of a particular dose of drug in a particular species, such as NOEL(no-observed-effect-level), NOAEL(no-observed-adverse-effect-level) and LOAEL(lowest-observed-adverse-effect-level). They are established by reviewing the available studies and animal studies. The application of threshold dose in risk assessment safeguards the participants in human clinical trials and evaluates the risks of chronic exposure to certain substances. However, the nature of animal studies also limits the applicability of experimental results in the human population and its significance in evaluating potential risk of certain substances. In toxicology, there are some other safety factors including LD50, LC50 and EC50.













