Related Research Articles

Bacterial conjugation is the transfer of genetic material between bacterial cells by direct cell-to-cell contact or by a bridge-like connection between two cells. This takes place through a pilus. It is a parasexual mode of reproduction in bacteria.

Enterobacteria phage λ is a bacterial virus, or bacteriophage, that infects the bacterial species Escherichia coli. It was discovered by Esther Lederberg in 1950. The wild type of this virus has a temperate life cycle that allows it to either reside within the genome of its host through lysogeny or enter into a lytic phase, during which it kills and lyses the cell to produce offspring. Lambda strains, mutated at specific sites, are unable to lysogenize cells; instead, they grow and enter the lytic cycle after superinfecting an already lysogenized cell.
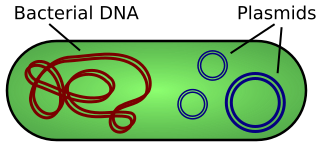
A plasmid is a small, extrachromosomal DNA molecule within a cell that is physically separated from chromosomal DNA and can replicate independently. They are most commonly found as small circular, double-stranded DNA molecules in bacteria; however, plasmids are sometimes present in archaea and eukaryotic organisms. In nature, plasmids often carry genes that benefit the survival of the organism and confer selective advantage such as antibiotic resistance. While chromosomes are large and contain all the essential genetic information for living under normal conditions, plasmids are usually very small and contain only additional genes that may be useful in certain situations or conditions. Artificial plasmids are widely used as vectors in molecular cloning, serving to drive the replication of recombinant DNA sequences within host organisms. In the laboratory, plasmids may be introduced into a cell via transformation. Synthetic plasmids are available for procurement over the internet.

Genetic recombination is the exchange of genetic material between different organisms which leads to production of offspring with combinations of traits that differ from those found in either parent. In eukaryotes, genetic recombination during meiosis can lead to a novel set of genetic information that can be further passed on from parents to offspring. Most recombination occurs naturally and can be classified into two types: (1) interchromosomal recombination, occurring through independent assortment of alleles whose loci are on different but homologous chromosomes ; & (2) intrachromosomal recombination, occurring through crossing over.

A cloning vector is a small piece of DNA that can be stably maintained in an organism, and into which a foreign DNA fragment can be inserted for cloning purposes. The cloning vector may be DNA taken from a virus, the cell of a higher organism, or it may be the plasmid of a bacterium. The vector contains features that allow for the convenient insertion of a DNA fragment into the vector or its removal from the vector, for example through the presence of restriction sites. The vector and the foreign DNA may be treated with a restriction enzyme that cuts the DNA, and DNA fragments thus generated contain either blunt ends or overhangs known as sticky ends, and vector DNA and foreign DNA with compatible ends can then be joined by molecular ligation. After a DNA fragment has been cloned into a cloning vector, it may be further subcloned into another vector designed for more specific use.

A high-frequency recombination cell is a bacterium with a conjugative plasmid integrated into its chromosomal DNA. The integration of the plasmid into the cell's chromosome is through homologous recombination. A conjugative plasmid capable of chromosome integration is also called an episome. When conjugation occurs, Hfr cells are very efficient in delivering chromosomal genes of the cell into recipient F− cells, which lack the episome.

A prophage is a bacteriophage genome that is integrated into the circular bacterial chromosome or exists as an extrachromosomal plasmid within the bacterial cell. Integration of prophages into the bacterial host is the characteristic step of the lysogenic cycle of temperate phages. Prophages remain latent in the genome through multiple cell divisions until activation by an external factor, such as UV light, leading to production of new phage particles that will lyse the cell and spread. As ubiquitous mobile genetic elements, prophages play important roles in bacterial genetics and evolution, such as in the acquisition of virulence factors.

Transduction is the process by which foreign DNA is introduced into a cell by a virus or viral vector. An example is the viral transfer of DNA from one bacterium to another and hence an example of horizontal gene transfer. Transduction does not require physical contact between the cell donating the DNA and the cell receiving the DNA, and it is DNase resistant. Transduction is a common tool used by molecular biologists to stably introduce a foreign gene into a host cell's genome.

Lysogeny, or the lysogenic cycle, is one of two cycles of viral reproduction. Lysogeny is characterized by integration of the bacteriophage nucleic acid into the host bacterium's genome or formation of a circular replicon in the bacterial cytoplasm. In this condition the bacterium continues to live and reproduce normally, while the bacteriophage lies in a dormant state in the host cell. The genetic material of the bacteriophage, called a prophage, can be transmitted to daughter cells at each subsequent cell division, and later events can release it, causing proliferation of new phages via the lytic cycle.
Merozygote is a state when a cell, usually bacterial, is temporarily partial diploid as result of DNA transfer processes like conjugation.
The F-plasmid allows genes to be transferred from one bacterium carrying the factor to another bacterium lacking the factor by conjugation. The F factor was the first plasmid to be discovered. Unlike other plasmids, F factor is constitutive for transfer proteins due to a mutation in the gene finO. The F plasmid belongs to F-like plasmids, a class of conjugative plasmids that control sexual functions of bacteria with a fertility inhibition (Fin) system.
Microbial genetics is a subject area within microbiology and genetic engineering. Microbial genetics studies microorganisms for different purposes. The microorganisms that are observed are bacteria, and archaea. Some fungi and protozoa are also subjects used to study in this field. The studies of microorganisms involve studies of genotype and expression system. Genotypes are the inherited compositions of an organism. Genetic Engineering is a field of work and study within microbial genetics. The usage of recombinant DNA technology is a process of this work. The process involves creating recombinant DNA molecules through manipulating a DNA sequence. That DNA created is then in contact with a host organism. Cloning is also an example of genetic engineering.
Allan McCulloch Campbell was an American microbiologist and geneticist and the Barbara Kimball Browning Professor Emeritus in the Department of Biology at Stanford University. His pioneering work on Lambda phage helped to advance molecular biology in the late 20th century. An important collaborator and member of his laboratory at Stanford University was biochemist Alice del Campillo Campbell, his wife.
Fosmids are similar to cosmids but are based on the bacterial F-plasmid. The cloning vector is limited, as a host can only contain one fosmid molecule. Fosmids can hold DNA inserts of up to 40 kb in size; often the source of the insert is random genomic DNA. A fosmid library is prepared by extracting the genomic DNA from the target organism and cloning it into the fosmid vector. The ligation mix is then packaged into phage particles and the DNA is transfected into the bacterial host. Bacterial clones propagate the fosmid library. The low copy number offers higher stability than vectors with relatively higher copy numbers, including cosmids. Fosmids may be useful for constructing stable libraries from complex genomes. Fosmids have high structural stability and have been found to maintain human DNA effectively even after 100 generations of bacterial growth. Fosmid clones were used to help assess the accuracy of the Public Human Genome Sequence.

Esther Miriam Zimmer Lederberg was an American microbiologist and a pioneer of bacterial genetics. She discovered the bacterial virus λ and the bacterial fertility factor F, devised the first implementation of replica plating, and furthered the understanding of the transfer of genes between bacteria by specialized transduction.

Bacteriophage P2, scientific name Escherichia virus P2, is a temperate phage that infects E. coli. It is a tailed virus with a contractile sheath and is thus classified in the genus Peduovirus, subfamily Peduovirinae, family Myoviridae within order Caudovirales. This genus of viruses includes many P2-like phages as well as the satellite phage P4.

Escherichia coli is a Gram-negative gammaproteobacterium commonly found in the lower intestine of warm-blooded organisms (endotherms). The descendants of two isolates, K-12 and B strain, are used routinely in molecular biology as both a tool and a model organism.
Bacterial genetics is the subfield of genetics devoted to the study of bacterial genes. Bacterial genetics are subtly different from eukaryotic genetics, however bacteria still serve as a good model for animal genetic studies. One of the major distinctions between bacterial and eukaryotic genetics stems from the bacteria's lack of membrane-bound organelles, necessitating protein synthesis occur in the cytoplasm.
Bacterial recombination is a type of genetic recombination in bacteria characterized by DNA transfer from one organism called donor to another organism as recipient. This process occurs in three main ways:
Integrative and conjugative elements (ICEs) are mobile genetic elements present in both gram-positive and gram-negative bacteria. In a donor cell, ICEs are located primarily on the chromosome, but have the ability to excise themselves from the genome and transfer to recipient cells via bacterial conjugation.
References
- ↑ Griffiths AJ, Miller JH, Suzuki DT, Lewontin RC, Gelbart WM. "An Introduction to Genetic Analysis". NCBI Bookshelf. Retrieved 7 November 2016.
- ↑ Brock TD (1990). The Emergence of Bacterial Genetics. Cold Spring Harbor, N.Y.: Cold Spring Harbor Laboratory Press. p. 97. ISBN 978-0879693503.
- ↑ Brock TD (1990). The Emergence of Bacterial Genetics. Cold Spring Harbor, N.Y.: Cold Spring Harbor Laboratory Press. p. 181. ISBN 978-0879693503.
- ↑ Lederberg J, Tatum EL (1946). "Gene recombination in Escherichia coli". Nature. 158 (4016): 558. Bibcode:1946Natur.158..558L. doi: 10.1038/158558a0 . PMID 21001945. S2CID 1826960.
- ↑ Brock TD (1990). The Emergence of Bacterial Genetics. Cold Spring Harbor, N.Y.: Cold Spring Harbor Laboratory Press. pp. 96–97. ISBN 978-0879693503.
- ↑ Lederberg J (1955). "Genetic recombination in bacteria". Science. 122 (3176): 920. Bibcode:1955Sci...122..920L. doi:10.1126/science.122.3176.920. PMID 13274050. S2CID 33350056.
- ↑ Wollman EL, Jacob F, Hayes W (1956). "Conjugation and genetic recombination in Escherichia coli K-12". Cold Spring Harbor Symposia on Quantitative Biology. 21: 141–62. doi:10.1101/sqb.1956.021.01.012. PMID 13433587.