
Thiamine, also known as thiamin and vitamin B1, is a vitamin, an essential micronutrient, that cannot be made in the body. It is found in food and commercially synthesized to be a dietary supplement or medication. Phosphorylated forms of thiamine are required for some metabolic reactions, including the breakdown of glucose and amino acids.
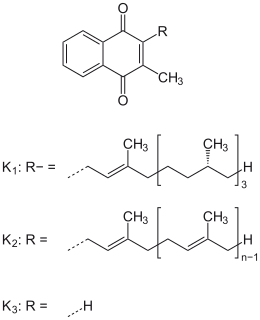
Vitamin K refers to structurally similar, fat-soluble vitamers found in foods and marketed as dietary supplements. The human body requires vitamin K for post-synthesis modification of certain proteins that are required for blood coagulation or for controlling binding of calcium in bones and other tissues. The complete synthesis involves final modification of these so-called "Gla proteins" by the enzyme gamma-glutamyl carboxylase that uses vitamin K as a cofactor.
Niacin, also known as nicotinic acid, is an organic compound and a form of vitamin B3, an essential human nutrient. It can be manufactured by plants and animals from the amino acid tryptophan. Niacin is obtained in the diet from a variety of whole and processed foods, with highest contents in fortified packaged foods, meat, poultry, red fish such as tuna and salmon, lesser amounts in nuts, legumes and seeds. Niacin as a dietary supplement is used to treat pellagra, a disease caused by niacin deficiency. Signs and symptoms of pellagra include skin and mouth lesions, anemia, headaches, and tiredness. Many countries mandate its addition to wheat flour or other food grains, thereby reducing the risk of pellagra.

Pantothenic acid, also called vitamin B5 is a water-soluble B vitamin and therefore an essential nutrient. All animals require pantothenic acid in order to synthesize coenzyme A (CoA) – essential for fatty acid metabolism – as well as to, in general, synthesize and metabolize proteins, carbohydrates, and fats.

Vitamin B6 is one of the B vitamins, and thus an essential nutrient. The term refers to a group of six chemically similar compounds, i.e., "vitamers", which can be interconverted in biological systems. Its active form, pyridoxal 5′-phosphate, serves as a coenzyme in more than 140 enzyme reactions in amino acid, glucose, and lipid metabolism.
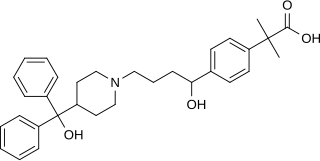
Fexofenadine, sold under the brand name Allegra among others, is an antihistamine pharmaceutical drug used in the treatment of allergy symptoms, such as hay fever and urticaria.

Menadione is a natural organic compound with the formula C6H4(CO)2C2H(CH3). It is an analog of 1,4-naphthoquinone with a methyl group in the 2-position. It is sometimes called vitamin K3. Use is allowed as a nutritional supplement in animal feed because of its vitamin K activity.

Arecoline is a nicotinic acid-based mild parasympathomimetic stimulant alkaloid found in the areca nut, the fruit of the areca palm. It is an odourless oily liquid. It can bring a sense of enhanced alertness and energy, euphoria and relaxation. Its psychoactive effects are comparable to that of nicotine.

Hordenine is an alkaloid of the phenethylamine class that occurs naturally in a variety of plants, taking its name from one of the most common, barley. Chemically, hordenine is the N-methyl derivative of N-methyltyramine, and the N,N-dimethyl derivative of the well-known biogenic amine tyramine, from which it is biosynthetically derived and with which it shares some pharmacological properties. As of September 2012, hordenine is widely sold as an ingredient of nutritional supplements, with the claims that it is a stimulant of the central nervous system, and has the ability to promote weight loss by enhancing metabolism. In experimental animals, given sufficiently large doses parenterally, hordenine does produce an increase in blood pressure, as well as other disturbances of the cardiovascular, respiratory, and nervous systems. These effects are generally not reproduced by oral administration of the drug in test animals, and virtually no scientific reports of the effects of hordenine in human beings have been published.

Ketobemidone, sold under the brand name Ketogan among others, is a powerful synthetic opioid painkiller. Its effectiveness against pain is in the same range as morphine, and it also has some NMDA-antagonist properties imparted, in part, by its metabolite norketobemidone. This may make it useful for some types of pain that do not respond well to other opioids. It is marketed in Denmark, Iceland, Norway and Sweden and is used for severe pain.

Methyllycaconitine (MLA) is a diterpenoid alkaloid found in many species of Delphinium (larkspurs). In common with many other diterpenoid alkaloids, it is toxic to animals, although the acute toxicity varies with species. Early research was focused on identifying, and characterizing the properties of methyllycaconitine as one of the principal toxins in larkspurs responsible for livestock poisoning in the mountain rangelands of North America. Methyllycaconitine has been explored as a possible therapeutic agent for the treatment of spastic paralyses in man, and it has been shown to have insecticidal properties. Most recently, it has become an important molecular probe for studying the pharmacology of the nicotinic acetylcholine receptor.

β-Methylphenethylamine is an organic compound of the phenethylamine class, and a positional isomer of the drug amphetamine, with which it shares some properties. In particular, both amphetamine and β-methylphenethylamine are human TAAR1 agonists. In appearance, it is a colorless or yellowish liquid.

Menatetrenone (INN), also known as MK-4, is one of the nine forms of vitamin K2.
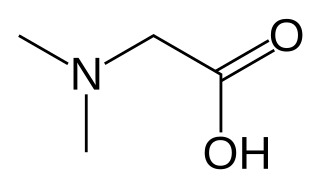
Dimethylglycine (DMG) is a derivative of the amino acid glycine with the structural formula (CH3)2NCH2COOH. It can be found in beans and liver, and has a sweet taste. It can be formed from trimethylglycine upon the loss of one of its methyl groups. It is also a byproduct of the metabolism of choline.

Remacemide is a drug which acts as a low-affinity NMDA antagonist with sodium channel blocking properties. It has been studied for the treatment of acute ischemic stroke, epilepsy, Huntington's disease, and Parkinson's disease.

N-Methyltyramine (NMT), also known as 4-hydroxy-N-methylphenethylamine, is a human trace amine and natural phenethylamine alkaloid found in a variety of plants. As the name implies, it is the N-methyl analog of tyramine, which is a well-known biogenic trace amine with which NMT shares many pharmacological properties. Biosynthetically, NMT is produced by the N-methylation of tyramine via the action of the enzyme phenylethanolamine N-methyltransferase in humans and tyramine N-methyltransferase in plants.

The pharmacokinetics of progesterone, concerns the pharmacodynamics, pharmacokinetics, and various routes of administration of progesterone.
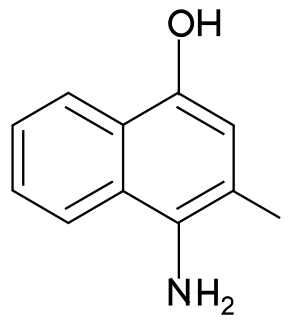
4-amino-3-methyl-1-naphthol is a synthetic menadione analog. It is also known as vitamin K7, and was named as such in 1950 when it was recognized as a compound with vitamin K activity.

2-Methylnaphthalene-1,4-diamine is a synthetic menadione analog with vitamin K activity.
Megavitamin-B6 syndrome is a collection of symptoms that can result from chronic supplementation, or acute overdose, of vitamin B6. While it is also known as hypervitaminosis B6, vitamin B6 toxicity and vitamin B6 excess, megavitamin-b6 syndrome is the name used in the ICD-10.



















