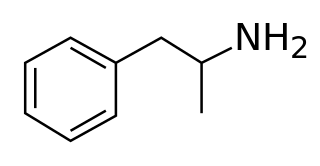
Amphetamine is a central nervous system (CNS) stimulant that is used in the treatment of attention deficit hyperactivity disorder (ADHD), narcolepsy, and obesity; it is also used to treat binge eating disorder in the form of its inactive prodrug lisdexamfetamine. Amphetamine was discovered as a chemical in 1887 by Lazăr Edeleanu, and then as a drug in the late 1920s. It exists as two enantiomers: levoamphetamine and dextroamphetamine. Amphetamine properly refers to a specific chemical, the racemic free base, which is equal parts of the two enantiomers in their pure amine forms. The term is frequently used informally to refer to any combination of the enantiomers, or to either of them alone. Historically, it has been used to treat nasal congestion and depression. Amphetamine is also used as an athletic performance enhancer and cognitive enhancer, and recreationally as an aphrodisiac and euphoriant. It is a prescription drug in many countries, and unauthorized possession and distribution of amphetamine are often tightly controlled due to the significant health risks associated with recreational use.

Orexin, also known as hypocretin, is a neuropeptide that regulates arousal, wakefulness, and appetite. It exists in the forms of orexin-A and orexin-B. The most common form of narcolepsy, type 1, in which the individual experiences brief losses of muscle tone, is caused by a lack of orexin in the brain due to destruction of the cells that produce it.

Monoamine transporters (MATs) are proteins that function as integral plasma-membrane transporters to regulate concentrations of extracellular monoamine neurotransmitters. The three major classes are serotonin transporters (SERTs), dopamine transporters (DATs), and norepinephrine transporters (NETs) and are responsible for the reuptake of their associated amine neurotransmitters. MATs are located just outside the synaptic cleft (peri-synaptically), transporting monoamine transmitter overflow from the synaptic cleft back to the cytoplasm of the pre-synaptic neuron. MAT regulation generally occurs through protein phosphorylation and post-translational modification. Due to their significance in neuronal signaling, MATs are commonly associated with drugs used to treat mental disorders as well as recreational drugs. Compounds targeting MATs range from medications such as the wide variety of tricyclic antidepressants, selective serotonin reuptake inhibitors such as fluoxetine (Prozac) to stimulant medications such as methylphenidate (Ritalin) and amphetamine in its many forms and derivatives methamphetamine (Desoxyn) and lisdexamfetamine (Vyvanse). Furthermore, drugs such as MDMA and natural alkaloids such as cocaine exert their effects in part by their interaction with MATs, by blocking the transporters from mopping up dopamine, serotonin, and other neurotransmitters from the synapse.

Cholecystokinin is a peptide hormone of the gastrointestinal system responsible for stimulating the digestion of fat and protein. Cholecystokinin, formerly called pancreozymin, is synthesized and secreted by enteroendocrine cells in the duodenum, the first segment of the small intestine. Its presence causes the release of digestive enzymes and bile from the pancreas and gallbladder, respectively..

Dopamine receptors are a class of G protein-coupled receptors that are prominent in the vertebrate central nervous system (CNS). Dopamine receptors activate different effectors through not only G-protein coupling, but also signaling through different protein interactions. The neurotransmitter dopamine is the primary endogenous ligand for dopamine receptors.
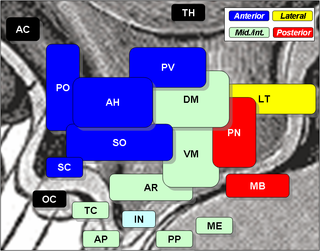
The arcuate nucleus of the hypothalamus (ARH), or ARC, is also known as the infundibular nucleus to distinguish it from the arcuate nucleus of the medulla oblongata in the brainstem. The arcuate nucleus is an aggregation of neurons in the mediobasal hypothalamus, adjacent to the third ventricle and the median eminence. The arcuate nucleus includes several important and diverse populations of neurons that help mediate different neuroendocrine and physiological functions, including neuroendocrine neurons, centrally projecting neurons, and astrocytes. The populations of neurons found in the arcuate nucleus are based on the hormones they secrete or interact with and are responsible for hypothalamic function, such as regulating hormones released from the pituitary gland or secreting their own hormones. Neurons in this region are also responsible for integrating information and providing inputs to other nuclei in the hypothalamus or inputs to areas outside this region of the brain. These neurons, generated from the ventral part of the periventricular epithelium during embryonic development, locate dorsally in the hypothalamus, becoming part of the ventromedial hypothalamic region. The function of the arcuate nucleus relies on its diversity of neurons, but its central role is involved in homeostasis. The arcuate nucleus provides many physiological roles involved in feeding, metabolism, fertility, and cardiovascular regulation.
Neuropeptides are chemical messengers made up of small chains of amino acids that are synthesized and released by neurons. Neuropeptides typically bind to G protein-coupled receptors (GPCRs) to modulate neural activity and other tissues like the gut, muscles, and heart.

Neuropeptide Y (NPY) is a 36 amino-acid neuropeptide that is involved in various physiological and homeostatic processes in both the central and peripheral nervous systems. It is secreted alongside other neurotransmitters such as GABA and glutamate.

The dopamine transporter is a membrane-spanning protein coded for in humans by the SLC6A3 gene, that pumps the neurotransmitter dopamine out of the synaptic cleft back into cytosol. In the cytosol, other transporters sequester the dopamine into vesicles for storage and later release. Dopamine reuptake via DAT provides the primary mechanism through which dopamine is cleared from synapses, although there may be an exception in the prefrontal cortex, where evidence points to a possibly larger role of the norepinephrine transporter.

Agouti-related protein (AgRP), also called agouti-related peptide, is a neuropeptide produced in the brain by the AgRP/NPY neuron. It is synthesized in neuropeptide Y (NPY)-containing cell bodies located in the ventromedial part of the arcuate nucleus in the hypothalamus. AgRP is co-expressed with NPY and acts to increase appetite and decrease metabolism and energy expenditure. It is one of the most potent and long-lasting of appetite stimulators. In humans, the agouti-related peptide is encoded by the AGRP gene.
Neuromodulation is the physiological process by which a given neuron uses one or more chemicals to regulate diverse populations of neurons. Neuromodulators typically bind to metabotropic, G-protein coupled receptors (GPCRs) to initiate a second messenger signaling cascade that induces a broad, long-lasting signal. This modulation can last for hundreds of milliseconds to several minutes. Some of the effects of neuromodulators include altering intrinsic firing activity, increasing or decreasing voltage-dependent currents, altering synaptic efficacy, increasing bursting activity and reconfiguring synaptic connectivity.

Calcitonin gene-related peptide (CGRP) is a member of the calcitonin family of peptides consisting of calcitonin, amylin, adrenomedullin, adrenomedullin 2 (intermedin) and calcitonin‑receptor‑stimulating peptide. Calcitonin is mainly produced by thyroid C cells whilst CGRP is secreted and stored in the nervous system. This peptide, in humans, exists in two forms: CGRP alpha, and CGRP beta. α-CGRP is a 37-amino acid neuropeptide and is formed by alternative splicing of the calcitonin/CGRP gene located on chromosome 11. β-CGRP is less studied. In humans, β-CGRP differs from α-CGRP by three amino acids and is encoded in a separate, nearby gene. The CGRP family includes calcitonin (CT), adrenomedullin (AM), and amylin (AMY).

DNQX (6,7-dinitroquinoxaline-2,3-dione) is a competitive antagonist at AMPA and kainate receptors, two ionotropic glutamate receptor (iGluR) subfamilies. It is used in a variety of molecular biology subfields, notably neurophysiology, to assist researchers in determining the properties of various types of ion channels and their potential applications in medicine.
The neuropeptide FF receptors are members of the G-protein coupled receptor superfamily of integral membrane proteins which bind the pain modulatory neuropeptides AF and FF. The Neuropeptide FF receptor family is a member of the G protein-coupled receptor superfamily containing two subtypes, NPFF1 and NPFF2, which exhibit a high affinity for Neuropeptide FF (NPFF) peptides. NPFF1 is broadly distributed in the central nervous system with the highest levels found in the limbic system and the hypothalamus. NPFF2 is present in high density, particularly in mammals in the superficial layers of the spinal cord where it is involved in nociception and modulation of opioid functions. These receptors participate to the modulation of opioid receptor function in the brain and spinal cord, and can either reduce or increase opioid receptor function depending which tissue they are released in, reflecting a complex role for neuropeptide FF in pain responses.

The cholecystokinin B receptor also known as CCKBR or CCK2 is a protein that in humans is encoded by the CCKBR gene.

Neuropeptide Y receptor type 2 (Y2R) is a member of the neuropeptide Y receptor family of G-protein coupled receptors, that in humans is encoded by the NPY2R gene.

Probable G-protein coupled receptor 160 is a protein that in humans is encoded by the GPR160 gene. It has been identified as the receptor for Cocaine- and amphetamine-regulated transcript, also known as CART.

Pancreatic polypeptide receptor 1, also known as Neuropeptide Y receptor type 4, is a protein that in humans is encoded by the PPYR1 gene.

A monoamine releasing agent (MRA), or simply monoamine releaser, is a drug that induces the release of a monoamine neurotransmitter from the presynaptic neuron into the synapse, leading to an increase in the extracellular concentrations of the neurotransmitter. Many drugs induce their effects in the body and/or brain via the release of monoamine neurotransmitters, e.g., trace amines, many substituted amphetamines, and related compounds.

Neuropeptide S (NPS) is a neuropeptide found in human and mammalian brain, mainly produced by neurons in the amygdala and between Barrington's nucleus and the locus coeruleus, although NPS-responsive neurons extend projections into many other brain areas. NPS binds specifically to a G protein-coupled receptor, NPSR. Animal studies show that NPS suppresses anxiety and appetite, induces wakefulness and hyperactivity, including hyper-sexuality, and plays a significant role in the extinction of conditioned fear. It has also been shown to significantly enhance dopamine activity in the mesolimbic pathway, and inhibits motility and increases permeability in neurocrine fashion acting through NO in the myenteric plexus in rats and humans.
















