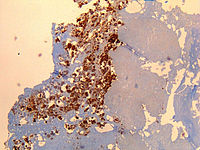
Salmonella is a genus of rod-shaped (bacillus) gram-negative bacteria of the family Enterobacteriaceae. The two known species of Salmonella are Salmonella enterica and Salmonella bongori. S. enterica is the type species and is further divided into six subspecies that include over 2,600 serotypes. Salmonella was named after Daniel Elmer Salmon (1850–1914), an American veterinary surgeon.

Q fever or query fever is a disease caused by infection with Coxiella burnetii, a bacterium that affects humans and other animals. This organism is uncommon, but may be found in cattle, sheep, goats, and other domestic mammals, including cats and dogs. The infection results from inhalation of a spore-like small-cell variant, and from contact with the milk, urine, feces, vaginal mucus, or semen of infected animals. Rarely, the disease is tick-borne. The incubation period can range from 9 to 40 days. Humans are vulnerable to Q fever, and infection can result from even a few organisms. The bacterium is an obligate intracellular pathogenic parasite.

Rickettsia is a genus of nonmotile, gram-negative, nonspore-forming, highly pleomorphic bacteria that may occur in the forms of cocci, bacilli, or threads. The genus was named after Howard Taylor Ricketts in honor of his pioneering work on tick-borne spotted fever.
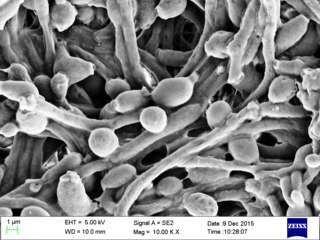
Candida albicans is an opportunistic pathogenic yeast that is a common member of the human gut flora. It can also survive outside the human body. It is detected in the gastrointestinal tract and mouth in 40–60% of healthy adults. It is usually a commensal organism, but it can become pathogenic in immunocompromised individuals under a variety of conditions. It is one of the few species of the genus Candida that cause the human infection candidiasis, which results from an overgrowth of the fungus. Candidiasis is, for example, often observed in HIV-infected patients. C. albicans is the most common fungal species isolated from biofilms either formed on (permanent) implanted medical devices or on human tissue. C. albicans, C. tropicalis, C. parapsilosis, and C. glabrata are together responsible for 50–90% of all cases of candidiasis in humans. A mortality rate of 40% has been reported for patients with systemic candidiasis due to C. albicans. By one estimate, invasive candidiasis contracted in a hospital causes 2,800 to 11,200 deaths yearly in the US. Nevertheless, these numbers may not truly reflect the true extent of damage this organism causes, given new studies indicating that C. albicans can cross the blood–brain barrier in mice.
Rickettsia prowazekii is a species of gram-negative, alphaproteobacteria, obligate intracellular parasitic, aerobic bacillus bacteria that is the etiologic agent of epidemic typhus, transmitted in the feces of lice. In North America, the main reservoir for R. prowazekii is the flying squirrel. R. prowazekii is often surrounded by a protein microcapsular layer and slime layer; the natural life cycle of the bacterium generally involves a vertebrate and an invertebrate host, usually an arthropod, typically the human body louse. A form of R. prowazekii that exists in the feces of arthropods remains stably infective for months. R. prowazekii also appears to be the closest free-living relative of mitochondria, based on genome sequencing.

Rickettsia rickettsii is a Gram-negative, intracellular, coccobacillus bacterium that was first discovered in 1902. R. rickettsii is the causative agent of Rocky Mountain Spotted Fever and is transferred to its host via a tick bite. It is one of the most pathogenic Rickettsia species and affects a large majority of the Western Hemisphere, most commonly the Americas. The pathogenic agent has been found on every continent, except Antarctica; however, Rocky Mountain Spotted Fever occurs mostly in North, Central, and South America. This prevalence is due to R. rickettsi thriving in warm, damp environments.

Legionella pneumophila is an aerobic, pleomorphic, flagellated, non-spore-forming, Gram-negative bacterium of the genus Legionella. L. pneumophila is the primary human pathogenic bacterium in this group. In nature, L. pneumophila infects freshwater and soil amoebae of the genera Acanthamoeba and Naegleria. This pathogen is found commonly near freshwater environments and will then invade the amoebae found in these environments, using them to carry out metabolic functions.
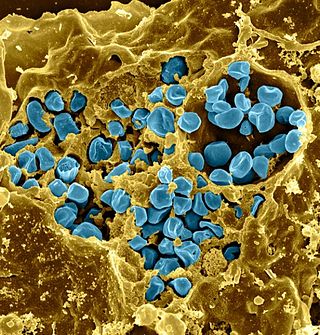
Francisella tularensis is a pathogenic species of Gram-negative coccobacillus, an aerobic bacterium. It is nonspore-forming, nonmotile, and the causative agent of tularemia, the pneumonic form of which is often lethal without treatment. It is a fastidious, facultative intracellular bacterium, which requires cysteine for growth. Due to its low infectious dose, ease of spread by aerosol, and high virulence, F. tularensis is classified as a Tier 1 Select Agent by the U.S. government, along with other potential agents of bioterrorism such as Yersinia pestis, Bacillus anthracis, and Ebola virus. When found in nature, Francisella tularensis can survive for several weeks at low temperatures in animal carcasses, soil, and water. In the laboratory, F. tularensis appears as small rods, and is grown best at 35–37 °C.

In cell biology, a phagosome is a vesicle formed around a particle engulfed by a phagocyte via phagocytosis. Professional phagocytes include macrophages, neutrophils, and dendritic cells (DCs).
Intracellular parasites are microparasites that are capable of growing and reproducing inside the cells of a host. They are also called intracellular pathogens.

Shigella flexneri is a species of Gram-negative bacteria in the genus Shigella that can cause diarrhea in humans. Several different serogroups of Shigella are described; S. flexneri belongs to group B. S. flexneri infections can usually be treated with antibiotics, although some strains have become resistant. Less severe cases are not usually treated because they become more resistant in the future. Shigella are closely related to Escherichia coli, but can be differentiated from E.coli based on pathogenicity, physiology and serology.

In biology, a phagolysosome, or endolysosome, is a cytoplasmic body formed by the fusion of a phagosome with a lysosome in a process that occurs during phagocytosis. Formation of phagolysosomes is essential for the intracellular destruction of microorganisms and pathogens. It takes place when the phagosome's and lysosome's membranes 'collide', at which point the lysosomal contents—including hydrolytic enzymes—are discharged into the phagosome in an explosive manner and digest the particles that the phagosome had ingested. Some products of the digestion are useful materials and are moved into the cytoplasm; others are exported by exocytosis.
In biology, axenic describes the state of a culture in which only a single species, variety, or strain of organism is present and entirely free of all other contaminating organisms. The earliest axenic cultures were of bacteria or unicellular eukaryotes, but axenic cultures of many multicellular organisms are also possible. Axenic culture is an important tool for the study of symbiotic and parasitic organisms in a controlled environment.

Ehrlichia is a genus of Rickettsiales bacteria that are transmitted to vertebrates by ticks. These bacteria cause the disease ehrlichiosis, which is considered zoonotic, because the main reservoirs for the disease are animals.

Orientia tsutsugamushi is a mite-borne bacterium belonging to the family Rickettsiaceae and is responsible for a disease called scrub typhus in humans. It is a natural and an obligate intracellular parasite of mites belonging to the family Trombiculidae. With a genome of only 2.0–2.7 Mb, it has the most repeated DNA sequences among bacterial genomes sequenced so far. The disease, scrub typhus, occurs when infected mite larvae accidentally bite humans. Primarily indicated by undifferentiated febrile illnesses, the infection can be complicated and often fatal.

Rhodococcus equi is a Gram-positive coccobacillus bacterium. The organism is commonly found in dry and dusty soil and can be important for diseases of domesticated animals. The frequency of infection can reach near 60%. R. equi is an important pathogen causing pneumonia in foals. Since 2008, R. equi has been known to infect wild boar and domestic pigs. R. equi can infect humans. At-risk groups are immunocompromised people, such as HIV-AIDS patients or transplant recipients. Rhodococcus infection in these patients resemble clinical and pathological signs of pulmonary tuberculosis. It is facultative intracellular.

Bacillus anthracis is a gram-positive and rod-shaped bacterium that causes anthrax, a deadly disease to livestock and, occasionally, to humans. It is the only permanent (obligate) pathogen within the genus Bacillus. Its infection is a type of zoonosis, as it is transmitted from animals to humans. It was discovered by a German physician Robert Koch in 1876, and became the first bacterium to be experimentally shown as a pathogen. The discovery was also the first scientific evidence for the germ theory of diseases.
Rickettsia typhi is a small, aerobic, obligate intracellular, rod shaped gram negative bacterium. It belongs to the typhus group of the Rickettsia genus, along with R. prowazekii. R. typhi has an uncertain history, as it may have long gone shadowed by epidemic typhus. This bacterium is recognized as a biocontainment level 2/3 organism. R. typhi is a flea-borne disease that is best known to be the causative agent for the disease murine typhus, which is an endemic typhus in humans that is distributed worldwide. As with all rickettsial organisms, R. typhi is a zoonotic agent that causes the disease murine typhus, displaying non-specific mild symptoms of fevers, headaches, pains and rashes. There are two cycles of R. typhi transmission from animal reservoirs containing R. typhi to humans: a classic rat-flea-rat cycle that is most well studied and common, and a secondary periodomestic cycle that could involve cats, dogs, opossums, sheep, and their fleas.
Rickettsia australis is a bacterium that causes a medical condition called Queensland tick typhus. The probable vectors are the tick species, Ixodes holocyclus and Ixodes tasmani. Small marsupials are suspected reservoirs of this bacterium.

Paul Fiset was a Canadian-American microbiologist and virologist. His research helped to develop one of the first successful Q fever vaccines, noted by The New York Times. Fiset was born in Quebec, Canada, and attended Laval University, where he earned a Doctor of Medicine degree in 1949. He subsequently attended Cambridge University, where he received a PhD degree in 1956. As a professor at the University of Maryland School of Medicine, he also researched other bacterial diseases such as typhus and Rocky Mountain spotted fever, in addition to Q fever.