Related Research Articles
An allergen is a type of antigen that produces an abnormally vigorous immune response in which the immune system fights off a perceived threat that would otherwise be harmless to the body. Such reactions are called allergies.
A radioallergosorbent test (RAST) is a blood test using radioimmunoassay test to detect specific IgE antibodies in order to determine the substances a subject is allergic to. This is different from a skin allergy test, which determines allergy by the reaction of a person's skin to different substances.
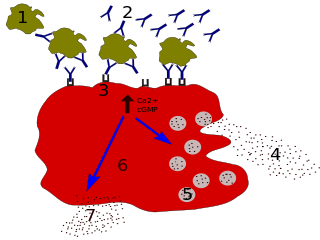
Immunoglobulin E (IgE) is a type of antibody that has been found only in mammals. IgE is synthesised by plasma cells. Monomers of IgE consist of two heavy chains and two light chains, with the ε chain containing four Ig-like constant domains (Cε1–Cε4). IgE is thought to be an important part of the immune response against infection by certain parasitic worms, including Schistosoma mansoni, Trichinella spiralis, and Fasciola hepatica. IgE is also utilized during immune defense against certain protozoan parasites such as Plasmodium falciparum. IgE may have evolved as a defense to protect against venoms.

Hives, also known as urticaria, is a kind of skin rash with red, raised, itchy bumps. Hives may burn or sting. The patches of rash may appear on different body parts, with variable duration from minutes to days, and does not leave any long-lasting skin change. Fewer than 5% of cases last for more than six weeks. The condition frequently recurs.
Omalizumab, sold under the brand name Xolair, is a medication to treat asthma, nasal polyps, and urticaria (hives).
Food intolerance is a detrimental reaction, often delayed, to a food, beverage, food additive, or compound found in foods that produces symptoms in one or more body organs and systems, but generally refers to reactions other than food allergy. Food hypersensitivity is used to refer broadly to both food intolerances and food allergies.

Atopy is the tendency to produce an exaggerated immunoglobulin E (IgE) immune response to otherwise harmless substances in the environment. Allergic diseases are clinical manifestations of such inappropriate, atopic responses.

Activation-induced cytidine deaminase, also known as AICDA, AID and single-stranded DNA cytosine deaminase, is a 24 kDa enzyme which in humans is encoded by the AICDA gene. It creates mutations in DNA by deamination of cytosine base, which turns it into uracil. In other words, it changes a C:G base pair into a U:G mismatch. The cell's DNA replication machinery recognizes the U as a T, and hence C:G is converted to a T:A base pair. During germinal center development of B lymphocytes, AID also generates other types of mutations, such as C:G to A:T. The mechanism by which these other mutations are created is not well understood. It is a member of the APOBEC family.

Physical urticaria is a distinct subgroup of urticaria (hives) that are induced by an exogenous physical stimulus rather than occurring spontaneously. There are seven subcategories that are recognized as independent diseases. Physical urticaria is known to be painful, itchy and physically unappealing; it can recur for months to years.

Selective immunoglobulin A (IgA) deficiency (SIgAD) is a kind of immunodeficiency, a type of hypogammaglobulinemia. People with this deficiency lack immunoglobulin A (IgA), a type of antibody that protects against infections of the mucous membranes lining the mouth, airways, and digestive tract. It is defined as an undetectable serum IgA level in the presence of normal serum levels of IgG and IgM, in persons older than 4 years. It is the most common of the primary antibody deficiencies. Most such persons remain healthy throughout their lives and are never diagnosed.

Wheat allergy is an allergy to wheat which typically presents itself as a food allergy, but can also be a contact allergy resulting from occupational exposure. Like all allergies, wheat allergy involves immunoglobulin E and mast cell response. Typically the allergy is limited to the seed storage proteins of wheat. Some reactions are restricted to wheat proteins, while others can react across many varieties of seeds and other plant tissues. Wheat allergy is rare. Prevalence in adults was found to be 0.21% in a 2012 study in Japan.
Tanox was a biopharmaceutical company based in Houston, Texas. The company was founded by two biomedical research scientists, Nancy T. Chang and Tse Wen Chang in March 1986 with $250,000, which was a large part of their family savings at that time. Both Changs grew up and received college education in chemistry in National Tsing Hua University in Taiwan and obtained Ph.D. degrees from Harvard University. For postdoctoral training, Tse Wen shifted to immunology and did research with Herman N. Eisen at the Center for Cancer Research, M.I.T. The two Changs successively became research managers and worked with a range of monoclonal antibody projects in Centocor, Inc. based in Malvern, Pennsylvania, from 1981 to 1985. The Changs were recruited by Baylor College of Medicine toward the end of 1985 and offered faculty positions in the Division of Molecular Virology. Soon after their arrival, they were encouraged by a high-ranking Baylor official and local business leaders to start a biotech venture in Houston. This was in a period of time when the economy of Houston was in slump as the result of the collapse of the oil industry.
HLA A1-B8 is a multigene haplotype that covers the MHC Class I region of the human major histocompatibility complex on chromosome 6. A multigene haplotype is set of inherited alleles covering several genes, or gene-alleles; common multigene haplotypes are generally the result of identity by descent from a common ancestor. Chromosomal recombination fragments multigene haplotypes as the distance to that ancestor increases in number of generations.
Ligelizumab is a humanized IgG1 monoclonal antibody designed for the treatment of severe asthma and chronic spontaneous urticaria. It is an anti-IgE that binds to IGHE an acts as an immunomodulator.
Tse Wen Chang is an immunology researcher, whose career spans across academia and industry. His early research involving the Immunoglobulin E (IgE) pathway and antibody-based therapeutics lead to the development of omalizumab, a medication that has been approved for the treatment of severe allergic asthma and severe chronic spontaneous urticaria. Chang is a cofounder of Tanox, a biopharmaceutical company specialized in anti-IgE therapies for the treatment of allergic diseases. After Tanox's tripartite partnership with Genentech and Novartis was forged in 1996, Chang returned to his alma mater, the National Tsing Hua University in Taiwan and served as the Dean (1996–1999) of the College of Life Sciences. Chang was appointed by the Taiwanese government as President of the Development Center for Biotechnology (DCB) in 2000, and served as a Science and Technology Advisor of the Executive Yuan from 2002 to 2006. From 2006 to 2016, he was tenured as Distinguished Research Fellow at the Genomics Research Center, Academia Sinica. He founded Immunwork, Inc. in 2014.

PLAID syndrome is an inherited condition characterised by antibody deficiency and immune dysregulation, first described in 2012. The name is an acronym of "PLCG2-associated antibody deficiency and immune dysregulation". It is characterised by cold-induced urticaria, autoimmunity, atopy and humoral immune deficiency.

Fish allergy is an immune hypersensitivity to proteins found in fish. Symptoms can be either rapid or gradual in onset. The latter can take hours to days to appear. The former may include anaphylaxis, a potentially life-threatening condition which requires treatment with epinephrine. Other presentations may include atopic dermatitis or inflammation of the esophagus. Fish is one of the eight common food allergens which are responsible for 90% of allergic reactions to foods: cow's milk, eggs, wheat, shellfish, peanuts, tree nuts, fish, and soy beans.
Lirentelimab is a humanized nonfucosylated monoclonal antibody that targets sialic acid-binding Ig-like lectin 8 (SIGLEC8). In a randomized clinical trial, lirentelimab was found to improve eosinophil counts and symptoms in individuals with eosinophilic gastritis and duodenitis. Adverse reactions include infusion reactions, which are mild to moderate and typically occur following the first infusion.

A food allergy to sesame seeds has prevalence estimates in the range of 0.1–0.2% of the general population, and are higher in the Middle East and other countries where sesame seeds are used in traditional foods. Reporting of sesame seed allergy has increased in the 21st century, either due to a true increase from exposure to more sesame foods or due to an increase in awareness. Increasing sesame allergy rates have induced more countries to regulate food labels to identify sesame ingredients in products and the potential for allergy. In the United States, sesame became the ninth food allergen with mandatory labeling, effective 1 January 2023.
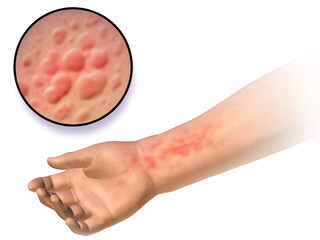
Autoimmune urticaria, also known as chronic autoimmune urticaria, is a type of chronic urticaria characterized by the presence of autoantibodies in the patient's immune system that target the body's own mast cells, leading to episodes of hives (urticaria). This immunologically distinct type of urticaria is considered autoimmune because the immune system, which normally protects the body from foreign organisms, mistakenly attacks the body's own cells, causing inflammation and other symptoms.
References
- ↑ Statement On A Nonproprietary Name Adopted By The USAN Council - Quilizumab, American Medical Association .
- ↑ World Health Organization (2011). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 106" (PDF). WHO Drug Information. 25 (4).
- ↑ Harris JM, Cabanski CR, Scheerens H, Samineni D, Bradley MS, Cochran C, Staubach P, Metz M, Sussman G, Maurer M (December 2016). "A randomized trial of quilizumab in adults with refractory chronic spontaneous urticaria". J. Allergy Clin. Immunol. 138 (6): 1730–1732. doi:10.1016/j.jaci.2016.06.023. PMID 27567329.
- ↑ Harris JM, Maciuca R, Bradley MS, Cabanski CR, Scheerens H, Lim J, Cai F, Kishnani M, Liao XC, Samineni D, Zhu R, Cochran C, Soong W, Diaz JD, Perin P, Tsukayama M, Dimov D, Agache I, Kelsen SG (March 2016). "A randomized trial of the efficacy and safety of quilizumab in adults with inadequately controlled allergic asthma". Respir. Res. 17: 29. doi: 10.1186/s12931-016-0347-2 . PMC 4797126 . PMID 26993628.
- ↑ Gauvreau GM, Harris JM, Boulet LP, Scheerens H, Fitzgerald JM, Putnam WS, Cockcroft DW, Davis BE, Leigh R, Zheng Y, Dahlén B, Wang Y, Maciuca R, Mayers I, Liao XC, Wu LC, Matthews JG, O'Byrne PM (July 2014). "Targeting membrane-expressed IgE B cell receptor with an antibody to the M1 prime epitope reduces IgE production". Sci Transl Med. 6 (243): 243ra85. doi:10.1126/scitranslmed.3008961. PMID 24990880. S2CID 41593528.
- ↑ Licari A, Castagnoli R, Panfili E, Marseglia A, Brambilla I, Marseglia GL (March 2017). "An Update on Anti-IgE Therapy in Pediatric Respiratory Diseases". Curr Respir Med Rev. 13 (1): 22–29. doi:10.2174/1573398X13666170616110738. PMC 5735517 . PMID 29290750.
- ↑ Deza G, Ricketti PA, Giménez-Arnau AM, Casale TB (2018). "Emerging Biomarkers and Therapeutic Pipelines for Chronic Spontaneous Urticaria". J Allergy Clin Immunol Pract. 6 (4): 1108–1117. doi:10.1016/j.jaip.2018.02.024. PMID 30033912. S2CID 51710078.