
Staphylococcus aureus is a gram-positive spherically shaped bacterium, a member of the Bacillota, and is a usual member of the microbiota of the body, frequently found in the upper respiratory tract and on the skin. It is often positive for catalase and nitrate reduction and is a facultative anaerobe, meaning that it can grow without oxygen. Although S. aureus usually acts as a commensal of the human microbiota, it can also become an opportunistic pathogen, being a common cause of skin infections including abscesses, respiratory infections such as sinusitis, and food poisoning. Pathogenic strains often promote infections by producing virulence factors such as potent protein toxins, and the expression of a cell-surface protein that binds and inactivates antibodies. S. aureus is one of the leading pathogens for deaths associated with antimicrobial resistance and the emergence of antibiotic-resistant strains, such as methicillin-resistant S. aureus (MRSA). The bacterium is a worldwide problem in clinical medicine. Despite much research and development, no vaccine for S. aureus has been approved.
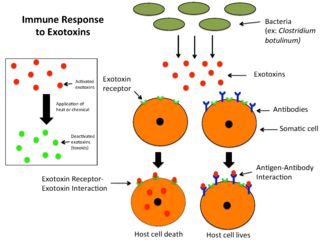
An exotoxin is a toxin secreted by bacteria. An exotoxin can cause damage to the host by destroying cells or disrupting normal cellular metabolism. They are highly potent and can cause major damage to the host. Exotoxins may be secreted, or, similar to endotoxins, may be released during lysis of the cell. Gram negative pathogens may secrete outer membrane vesicles containing lipopolysaccharide endotoxin and some virulence proteins in the bounding membrane along with some other toxins as intra-vesicular contents, thus adding a previously unforeseen dimension to the well-known eukaryote process of membrane vesicle trafficking, which is quite active at the host–pathogen interface.
Pathogenicity islands (PAIs), as termed in 1990, are a distinct class of genomic islands acquired by microorganisms through horizontal gene transfer. Pathogenicity islands are found in both animal and plant pathogens. Additionally, PAIs are found in both gram-positive and gram-negative bacteria. They are transferred through horizontal gene transfer events such as transfer by a plasmid, phage, or conjugative transposon. Although the general makeup of pathogenicity islands (PAIs) might vary throughout bacterial pathogen strains, all PAIs are known to have characteristics with all genomic islands, which includes virulence genes, functional mobility elements, and areas of homology to tRNA genes and direct repeats. Therefore, PAIs enables microorganisms to induce disease and also contribute to microorganisms' ability to evolve. The spread of antibiotic resistance and, more generally, the conversion of non-pathogenic strains in natural environments to strains that infect animal and plant hosts with disease are two examples of the evolutionary and ecological changes brought about by the transmission and acquisition of PAIs among bacterial species. However, It is impossible to overlook their impact on bacterial evolution, though, since if a PAI is acquired and is stably absorbed, it can irreversibly change the bacterial genome.

An enterotoxin is a protein exotoxin released by a microorganism that targets the intestines. They can be chromosomally or plasmid encoded. They are heat labile, of low molecular weight and water-soluble. Enterotoxins are frequently cytotoxic and kill cells by altering the apical membrane permeability of the mucosal (epithelial) cells of the intestinal wall. They are mostly pore-forming toxins, secreted by bacteria, that assemble to form pores in cell membranes. This causes the cells to die.
Virulence factors are cellular structures, molecules and regulatory systems that enable microbial pathogens to achieve the following:

Protein A is a 42 kDa surface protein originally found in the cell wall of the bacteria Staphylococcus aureus. It is encoded by the spa gene and its regulation is controlled by DNA topology, cellular osmolarity, and a two-component system called ArlS-ArlR. It has found use in biochemical research because of its ability to bind immunoglobulins. It is composed of five homologous Ig-binding domains that fold into a three-helix bundle. Each domain is able to bind proteins from many mammalian species, most notably IgGs. It binds the heavy chain within the Fc region of most immunoglobulins and also within the Fab region in the case of the human VH3 family. Through these interactions in serum, where IgG molecules are bound in the wrong orientation, the bacteria disrupts opsonization and phagocytosis.
Adenylate cyclase toxin is a virulence factor produced by some members of the genus Bordetella. Together with the pertussis toxin it is the most important virulence factor of the causative agent of whooping cough, Bordetella pertussis. Bordetella bronchiseptica and Bordetella parapertussis, also able to cause pertussis-like symptoms, also produce adenylate cyclase toxin. It is a toxin secreted by the bacteria to influence the host immune system.
Anthrax toxin is a three-protein exotoxin secreted by virulent strains of the bacterium, Bacillus anthracis—the causative agent of anthrax. The toxin was first discovered by Harry Smith in 1954. Anthrax toxin is composed of a cell-binding protein, known as protective antigen (PA), and two enzyme components, called edema factor (EF) and lethal factor (LF). These three protein components act together to impart their physiological effects. Assembled complexes containing the toxin components are endocytosed. In the endosome, the enzymatic components of the toxin translocate into the cytoplasm of a target cell. Once in the cytosol, the enzymatic components of the toxin disrupt various immune cell functions, namely cellular signaling and cell migration. The toxin may even induce cell lysis, as is observed for macrophage cells. Anthrax toxin allows the bacteria to evade the immune system, proliferate, and ultimately kill the host animal. Research on anthrax toxin also provides insight into the generation of macromolecular assemblies, and on protein translocation, pore formation, endocytosis, and other biochemical processes.

Panton–Valentine leukocidin (PVL) is a cytotoxin—one of the β-pore-forming toxins. The presence of PVL is associated with increased virulence of certain strains (isolates) of Staphylococcus aureus. It is present in the majority of community-associated methicillin-resistant Staphylococcus aureus (CA-MRSA) isolates studied and is the cause of necrotic lesions involving the skin or mucosa, including necrotic hemorrhagic pneumonia. PVL creates pores in the membranes of infected cells. PVL is produced from the genetic material of a bacteriophage that infects Staphylococcus aureus, making it more virulent.
Cytolysin refers to the substance secreted by microorganisms, plants or animals that is specifically toxic to individual cells, in many cases causing their dissolution through lysis. Cytolysins that have a specific action for certain cells are named accordingly. For instance, the cytolysins responsible for the destruction of red blood cells, thereby liberating hemoglobins, are named hemolysins, and so on. Cytolysins may be involved in immunity as well as in venoms.

Pore-forming proteins are usually produced by bacteria, and include a number of protein exotoxins but may also be produced by other organisms such as apple snails that produce perivitellin-2 or earthworms, who produce lysenin. They are frequently cytotoxic, as they create unregulated pores in the membrane of targeted cells.

Hemolysins or haemolysins are lipids and proteins that cause lysis of red blood cells by disrupting the cell membrane. Although the lytic activity of some microbe-derived hemolysins on red blood cells may be of great importance for nutrient acquisition, many hemolysins produced by pathogens do not cause significant destruction of red blood cells during infection. However, hemolysins are often capable of lysing red blood cells in vitro.
Phenol-soluble modulins (PSMs) are a family of small proteins, that carry out a variety of functions, including acting as toxins, assisting in biofilm formation, and colony spreading. PSMs are produced by Staphylococcus bacteria including Methicillin-resistant Staphylococcus aureus (MRSA), and Staphylococcus epidermidis. Many PSMs are encoded within the core genome and can play an important virulence factor. PSMs were first discovered in S. epidermidis by Seymour Klebanoff via hot-phenol extraction and were described as a pro-inflammatory complex of three peptides. Since their initial discovery, numerous roles of PSMs have been identified. However, due in part to the small size of many PSMs, they have largely gone unnoticed until recent years.
Staphylococcus aureus delta toxin is a toxin produced by Staphylococcus aureus. It has a wide spectrum of cytolytic activity.
Microbial toxins are toxins produced by micro-organisms, including bacteria, fungi, protozoa, dinoflagellates, and viruses. Many microbial toxins promote infection and disease by directly damaging host tissues and by disabling the immune system. Endotoxins most commonly refer to the lipopolysaccharide (LPS) or lipooligosaccharide (LOS) that are in the outer plasma membrane of Gram-negative bacteria. The botulinum toxin, which is primarily produced by Clostridium botulinum and less frequently by other Clostridium species, is the most toxic substance known in the world. However, microbial toxins also have important uses in medical science and research. Currently, new methods of detecting bacterial toxins are being developed to better isolate and understand these toxins. Potential applications of toxin research include combating microbial virulence, the development of novel anticancer drugs and other medicines, and the use of toxins as tools in neurobiology and cellular biology.
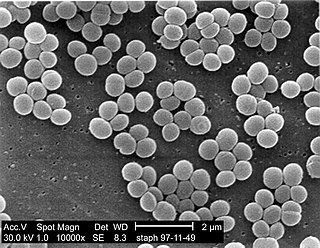
Staphylococcus is a genus of Gram-positive bacteria in the family Staphylococcaceae from the order Bacillales. Under the microscope, they appear spherical (cocci), and form in grape-like clusters. Staphylococcus species are facultative anaerobic organisms.
Clostridium perfringens beta toxin is one of the four major lethal protein toxins produced by Clostridium perfringens Type B and Type C strains. It is a necrotizing agent and it induces hypertension by release of catecholamine. It has been shown to cause necrotic enteritis in mammals and induces necrotizing intestinal lesions in the rabbit ileal loop model. C. perfringens beta toxin is susceptible to breakdown by proteolytic enzymes, particularly trypsin. Beta toxin is therefore highly lethal to infant mammals because of trypsin inhibitors present in the colostrum.
Staphylococcus pseudintermedius is a gram-positive spherically shaped bacterium of the genus Staphylococcus found worldwide. It is primarily a pathogen for domestic animals, but has been known to affect humans as well. S. pseudintermedius is an opportunistic pathogen that secretes immune-modulating virulence factors, has many adhesion factors, and the potential to create biofilms, all of which help to determine the pathogenicity of the bacterium. Diagnoses of S. pseudintermedius have traditionally been made using cytology, plating, and biochemical tests. More recently, molecular technologies like MALDI-TOF, DNA hybridization and PCR have become preferred over biochemical tests for their more rapid and accurate identifications. This includes the identification and diagnosis of antibiotic resistant strains.

Cry6Aa is a toxic crystal protein generated by the bacterial family Bacillus thuringiensis during sporulation. This protein is a member of the alpha pore forming toxins family, which gives it insecticidal qualities advantageous in agricultural pest control. Each Cry protein has some level of target specificity; Cry6Aa has specific toxic action against coleopteran insects and nematodes. The corresponding B. thuringiensis gene, cry6aa, is located on bacterial plasmids. Along with several other Cry protein genes, cry6aa can be genetically recombined in Bt corn and Bt cotton so the plants produce specific toxins. Insects are developing resistance to the most commonly inserted proteins like Cry1Ac. Since Cry6Aa proteins function differently than other Cry proteins, they are combined with other proteins to decrease the development of pest resistance. Recent studies suggest this protein functions better in combination with other virulence factors such as other Cry proteins and metalloproteinases.>
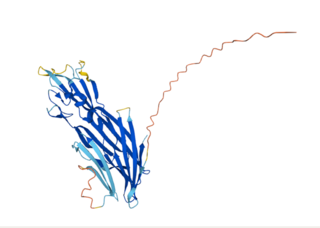
Cytotoxin-K (CytK) is a protein toxin produced by the gram-positive bacteria Bacillus cereus. It was first discovered in a certain Bacillus cereus strain which was isolated from a food poisoning epidemic that occurred in a French nursing home in 1998. There were six cases of bloody diarrhea, three of which were fatal. None of the known enterotoxins from B. cereus could be detected at this time. Later, this B. cereus strain and its relatives were classified as a brand-new species called Bacillus cytotoxicus, which is the thermo-tolerant member of the B. cereus genus. The cytotoxin-K gene is present in approximately 50% of Bacillus cereus isolates, and its expression is regulated by several factors, including temperature and nutrient availability.










