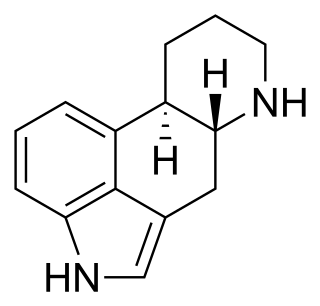
Ergoline is a chemical compound whose structural skeleton is contained in a variety of alkaloids, referred to as ergoline derivatives or ergoline alkaloids. Ergoline alkaloids, one being ergine, were initially characterized in ergot. Some of these are implicated in the condition ergotism, which can take a convulsive form or a gangrenous form. Even so, many ergoline alkaloids have been found to be clinically useful. Annual world production of ergot alkaloids has been estimated at 5,000–8,000 kg of all ergopeptines and 10,000–15,000 kg of lysergic acid, used primarily in the manufacture of semi-synthetic derivatives.

A dopamine agonist(DA) is a compound that activates dopamine receptors. There are two families of dopamine receptors, D1-like and D2-like. They are all G protein-coupled receptors. D1- and D5-receptors belong to the D1-like family and the D2-like family includes D2, D3 and D4 receptors. Dopamine agonists are primarily used in the treatment of Parkinson's disease, and to a lesser extent, in hyperprolactinemia and restless legs syndrome. They are also used off-label in the treatment of clinical depression. The use of dopamine agonists is associated with impulse control disorders and dopamine agonist withdrawal syndrome (DAWS).

PB-28 is an agonist of the sigma-2 receptor.
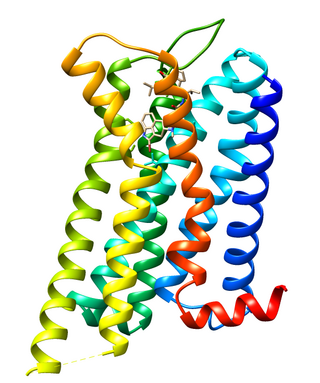
Dopamine receptor D2, also known as D2R, is a protein that, in humans, is encoded by the DRD2 gene. After work from Paul Greengard's lab had suggested that dopamine receptors were the site of action of antipsychotic drugs, several groups, including those of Solomon Snyder and Philip Seeman used a radiolabeled antipsychotic drug to identify what is now known as the dopamine D2 receptor. The dopamine D2 receptor is the main receptor for most antipsychotic drugs. The structure of DRD2 in complex with the atypical antipsychotic risperidone has been determined.

Dopamine receptor D1, also known as DRD1. It is one of the two types of D1-like receptor family — receptors D1 and D5. It is a protein that in humans is encoded by the DRD1 gene.

8-OH-DPAT is a research chemical of the aminotetralin chemical class which was developed in the 1980s and has been widely used to study the function of the 5-HT1A receptor. It was one of the first major 5-HT1A receptor full agonists to have been discovered.

Dopamine receptor D3 is a protein that in humans is encoded by the DRD3 gene.

N-n-Propylnorapomorphine (NPA) is an aporphine derivative dopamine agonist closely related to apomorphine. In rodents it has been shown to produce hyperactivity, stereotypy, hypothermia, antinociception, and penile erection, among other effects. Notably, its effects on locomotion are biphasic, with low doses producing inhibition and catalepsy and high doses resulting in enhancement of activity. This is likely due to preferential activation of D2/D3 autoreceptors versus postsynaptic receptors, the latter of which overcomes the former to increase postsynaptic dopaminergic signaling only with high doses.

UH-232 ((+)-UH232) is a drug which acts as a subtype selective mixed agonist-antagonist for dopamine receptors, acting as a weak partial agonist at the D3 subtype, and an antagonist at D2Sh autoreceptors on dopaminergic nerve terminals. It causes dopamine release in the brain and has a stimulant effect, as well as blocking the behavioural effects of cocaine. It may also serve as a 5-HT2A receptor agonist, based on animal studies. It was investigated in clinical trials for the treatment of schizophrenia, but unexpectedly caused symptoms to become worse.

SKF-83,959 is a synthetic benzazepine derivative used in scientific research which acts as an agonist at the D1–D2 dopamine receptor heteromer. It behaves as a full agonist at the D1 protomer and a high-affinity partial agonist at the D2 protomer. It was further shown to act as an allosteric modulator of the sigma-1 receptor. SKF-83,959 additionally inhibits sodium channels as well as delayed rectifier potassium channels. SKF-83,959 is a racemate that consists of the R-(+)- and S-(−)-enantiomers MCL-202 and MCL-201, respectively.

6-Br-APB is a synthetic compound that acts as a selective D1 agonist, with the (R)-enantiomer being a potent full agonist, while the (S) enantiomer retains its D1 selectivity but is a weak partial agonist. (R)-6-Br-APB and similar D1-selective full agonists like SKF-81,297 and SKF-82,958 produce characteristic anorectic effects, stereotyped behaviour and self-administration in animals, with a similar but not identical profile to that of dopaminergic stimulants such as amphetamine.

7-OH-DPAT is a synthetic compound that acts as a dopamine receptor agonist with reasonable selectivity for the D3 receptor subtype, and low affinity for serotonin receptors, unlike its structural isomer 8-OH-DPAT. 7-OH-DPAT is self-administered in several animal models, and is used to study its addiction effects to cocaine.

A-86929 is a synthetic compound that acts as a selective dopamine receptor D1 agonist. It was developed as a possible treatment for Parkinson's disease, as well as for other applications such as treatment of cocaine addiction, but while it had reasonable efficacy in humans it also caused dyskinesias and has not been continued. It has mainly been used as its diacetate ester prodrug adrogolide (ABT-431), which has better bioavailability.

2-OH-NPA is a drug used in scientific research which acts as a potent and selective agonist for the dopamine D2 receptor.

PD-128,907 is a drug used in scientific research which acts as a potent and selective agonist for the dopamine D2 and D3 receptors. It is used for studying the role of these receptors in the brain, in roles such as inhibitory autoreceptors that act to limit further dopamine release, as well as release of other neurotransmitters. In animal studies, it has been shown to reduce toxicity from cocaine overdose.
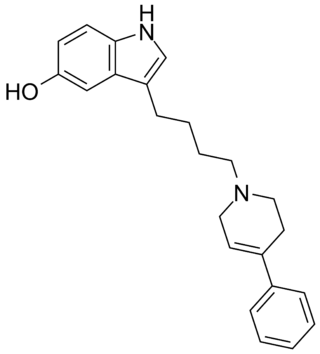
Roxindole (EMD-49,980) is a dopaminergic and serotonergic drug which was originally developed by Merck KGaA for the treatment of schizophrenia. In clinical trials its antipsychotic efficacy was only modest but it was unexpectedly found to produce potent and rapid antidepressant and anxiolytic effects. As a result, roxindole was further researched for the treatment of depression instead. It has also been investigated as a therapy for Parkinson's disease and prolactinoma.
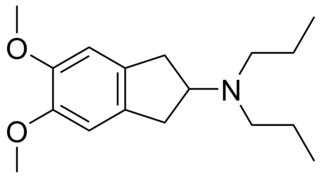
PNU-99,194(A) (or U-99,194(A)) is a drug which acts as a moderately selective D3 receptor antagonist with ~15-30-fold preference for D3 over the D2 subtype. Though it has substantially greater preference for D3 over D2, the latter receptor does still play some role in its effects, as evidenced by the fact that PNU-99,194 weakly stimulates both prolactin secretion and striatal dopamine synthesis, actions it does not share with the more selective (100-fold) D3 receptor antagonists S-14,297 and GR-103,691.
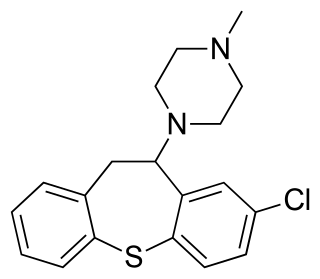
Clorotepine, also known as octoclothepin or octoclothepine, is an antipsychotic of the tricyclic group which was derived from perathiepin in 1965 and marketed in the Czech Republic by Spofa in or around 1971 for the treatment of schizophrenic psychosis.
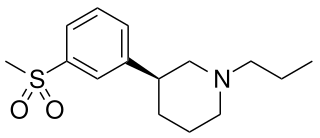
OSU-6162 (PNU-96391) is a compound which acts as a partial agonist at both dopamine D2 receptors and 5-HT2A receptors. It acts as a dopamine stabilizer in a similar manner to the closely related drug pridopidine, and has antipsychotic, anti-addictive and anti-Parkinsonian effects in animal studies. Both enantiomers show similar activity but with different ratios of effects, with the (S) enantiomer (–)-OSU-6162 that is more commonly used in research, having higher binding affinity to D2 but is a weaker partial agonist at 5-HT2A, while the (R) enantiomer (+)-OSU-6162 has higher efficacy at 5-HT2A but lower D2 affinity.

3-PPP (N-n-propyl-3-(3-hydroxyphenyl)piperidine) is a mixed sigma σ1 and σ2 receptor agonist (with similar affinity for both subtypes, though slightly higher affinity for the latter) and D2 receptor partial agonist which is used in scientific research. It shows stereoselectivity in its pharmacodynamics. (+)-3-PPP is the enantiomer that acts as an agonist of the sigma receptors; it is also an agonist of both D2 presynaptic and postsynaptic receptors. Conversely, (−)-3-PPP, also known as preclamol (INNTooltip International Nonproprietary Name), acts as an agonist of presynaptic D2 receptors but as an antagonist of postsynaptic D2 receptors, and has antipsychotic effects. 3-PPP has also been reported to be a monoamine reuptake inhibitor and possibly to act at adrenergic receptors or some other non-sigma receptor.




















