
Photosynthesis is a system of biological processes by which photosynthetic organisms, such as most plants, algae, and cyanobacteria, convert light energy, typically from sunlight, into the chemical energy necessary to fuel their activities. Photosynthetic organisms use intracellular organic compounds to store the chemical energy they produce in photosynthesis within organic compounds like sugars, glycogen, cellulose and starches. Photosynthesis is usually used to refer to oxygenic photosynthesis, a process that produces oxygen. To use this stored chemical energy, the organisms' cells metabolize the organic compounds through another process called cellular respiration. Photosynthesis plays a critical role in producing and maintaining the oxygen content of the Earth's atmosphere, and it supplies most of the biological energy necessary for complex life on Earth.
Phycoerythrin (PE) is a red protein-pigment complex from the light-harvesting phycobiliprotein family, present in cyanobacteria, red algae and cryptophytes, accessory to the main chlorophyll pigments responsible for photosynthesis.The red pigment is due to the prosthetic group, phycoerythrobilin, which gives phycoerythrin its red color.

Photosystems are functional and structural units of protein complexes involved in photosynthesis. Together they carry out the primary photochemistry of photosynthesis: the absorption of light and the transfer of energy and electrons. Photosystems are found in the thylakoid membranes of plants, algae, and cyanobacteria. These membranes are located inside the chloroplasts of plants and algae, and in the cytoplasmic membrane of photosynthetic bacteria. There are two kinds of photosystems: PSI and PSII.

Photosystem I is one of two photosystems in the photosynthetic light reactions of algae, plants, and cyanobacteria. Photosystem I is an integral membrane protein complex that uses light energy to catalyze the transfer of electrons across the thylakoid membrane from plastocyanin to ferredoxin. Ultimately, the electrons that are transferred by Photosystem I are used to produce the moderate-energy hydrogen carrier NADPH. The photon energy absorbed by Photosystem I also produces a proton-motive force that is used to generate ATP. PSI is composed of more than 110 cofactors, significantly more than Photosystem II.

Purple bacteria or purple photosynthetic bacteria are Gram-negative proteobacteria that are phototrophic, capable of producing their own food via photosynthesis. They are pigmented with bacteriochlorophyll a or b, together with various carotenoids, which give them colours ranging between purple, red, brown, and orange. They may be divided into two groups – purple sulfur bacteria and purple non-sulfur bacteria. Purple bacteria are anoxygenic phototrophs widely spread in nature, but especially in aquatic environments, where there are anoxic conditions that favor the synthesis of their pigments.
Photodissociation, photolysis, photodecomposition, or photofragmentation is a chemical reaction in which molecules of a chemical compound are broken down by photons. It is defined as the interaction of one or more photons with one target molecule.

A photosynthetic reaction center is a complex of several proteins, pigments, and other co-factors that together execute the primary energy conversion reactions of photosynthesis. Molecular excitations, either originating directly from sunlight or transferred as excitation energy via light-harvesting antenna systems, give rise to electron transfer reactions along the path of a series of protein-bound co-factors. These co-factors are light-absorbing molecules (also named chromophores or pigments) such as chlorophyll and pheophytin, as well as quinones. The energy of the photon is used to excite an electron of a pigment. The free energy created is then used, via a chain of nearby electron acceptors, for a transfer of hydrogen atoms (as protons and electrons) from H2O or hydrogen sulfide towards carbon dioxide, eventually producing glucose. These electron transfer steps ultimately result in the conversion of the energy of photons to chemical energy.

A chlorosome is a photosynthetic antenna complex found in green sulfur bacteria (GSB) and many green non-sulfur bacteria (GNsB), together known as green bacteria. They differ from other antenna complexes by their large size and lack of protein matrix supporting the photosynthetic pigments. Green sulfur bacteria are a group of organisms that generally live in extremely low-light environments, such as at depths of 100 metres in the Black Sea. The ability to capture light energy and rapidly deliver it to where it needs to go is essential to these bacteria, some of which see only a few photons of light per chlorophyll per day. To achieve this, the bacteria contain chlorosome structures, which contain up to 250,000 chlorophyll molecules. Chlorosomes are ellipsoidal bodies, in GSB their length varies from 100 to 200 nm, width of 50-100 nm and height of 15 – 30 nm, in GNsB the chlorosomes are somewhat smaller.
Photoprotection is the biochemical process that helps organisms cope with molecular damage caused by sunlight. Plants and other oxygenic phototrophs have developed a suite of photoprotective mechanisms to prevent photoinhibition and oxidative stress caused by excess or fluctuating light conditions. Humans and other animals have also developed photoprotective mechanisms to avoid UV photodamage to the skin, prevent DNA damage, and minimize the downstream effects of oxidative stress.

Biological pigments, also known simply as pigments or biochromes, are substances produced by living organisms that have a color resulting from selective color absorption. Biological pigments include plant pigments and flower pigments. Many biological structures, such as skin, eyes, feathers, fur and hair contain pigments such as melanin in specialized cells called chromatophores. In some species, pigments accrue over very long periods during an individual's lifespan.

A retinalophototroph is one of two different types of phototrophs, and are named for retinal-binding proteins they utilize for cell signaling and converting light into energy. Like all phototrophs, retinalophototrophs absorb photons to initiate their cellular processes. In contrast with chlorophototrophs, retinalophototrophs do not use chlorophyll or an electron transport chain to power their chemical reactions. This means retinalophototrophs are incapable of traditional carbon fixation, a fundamental photosynthetic process that transforms inorganic carbon into organic compounds. For this reason, experts consider them to be less efficient than their chlorophyll-using counterparts, chlorophototrophs.
Quantum biology is the study of applications of quantum mechanics and theoretical chemistry to aspects of biology that cannot be accurately described by the classical laws of physics. An understanding of fundamental quantum interactions is important because they determine the properties of the next level of organization in biological systems.

The antenna complex in purple photosynthetic bacteria are protein complexes responsible for the transfer of solar energy to the photosynthetic reaction centre. Purple bacteria, particularly Rhodopseudomonas acidophila of purple non-sulfur bacteria, have been one of the main groups of organisms used to study bacterial antenna complexes so much is known about this group's photosynthetic components. It is one of the many independent types of light-harvesting complex used by various photosynthetic organisms.

Light-dependent reactions are certain photochemical reactions involved in photosynthesis, the main process by which plants acquire energy. There are two light dependent reactions: the first occurs at photosystem II (PSII) and the second occurs at photosystem I (PSI).
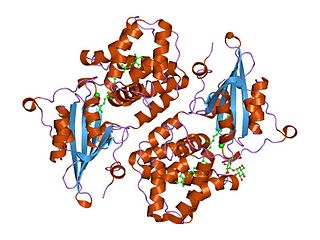
Orange carotenoid protein (OCP) is a water-soluble protein which plays a role in photoprotection in diverse cyanobacteria. It is the only photoactive protein known to use a carotenoid as the photoresponsive chromophore. The protein consists of two domains, with a single keto-carotenoid molecule non-covalently bound between the two domains. It is a very efficient quencher of excitation energy absorbed by the primary light-harvesting antenna complexes of cyanobacteria, the phycobilisomes. The quenching is induced by blue-green light. It is also capable of preventing oxidative damage by directly scavenging singlet oxygen (1O2).

The peridinin-chlorophyll-protein complex is a soluble molecular complex consisting of the peridinin-chlorophyll a-protein bound to peridinin, chlorophyll, and lipids. The peridinin molecules absorb light in the blue-green wavelengths and transfer energy to the chlorophyll molecules with extremely high efficiency. PCP complexes are found in many photosynthetic dinoflagellates, in which they may be the primary light-harvesting complexes.
Light harvesting materials harvest solar energy that can then be converted into chemical energy through photochemical processes. Synthetic light harvesting materials are inspired by photosynthetic biological systems such as light harvesting complexes and pigments that are present in plants and some photosynthetic bacteria. The dynamic and efficient antenna complexes that are present in photosynthetic organisms has inspired the design of synthetic light harvesting materials that mimic light harvesting machinery in biological systems. Examples of synthetic light harvesting materials are dendrimers, porphyrin arrays and assemblies, organic gels, biosynthetic and synthetic peptides, organic-inorganic hybrid materials, and semiconductor materials. Synthetic and biosynthetic light harvesting materials have applications in photovoltaics, photocatalysis, and photopolymerization.
Alexander Glazer was a professor of the Graduate School in the Department of Molecular and Cell Biology at the University of California, Berkeley. He had a passion for protein chemistry and structure function relationships. He also had a longstanding interest in light-harvesting complexes in cyanobacteria and red algae called phycobilisomes. He had also spent more than 10 years working on the human genome project where he has investigated methods for DNA detection and sequencing which most notably includes the development of fluorescent reagents involved in cell labeling. Most recently, he had focused his studies on issues in environmental sciences. He died on July 18, 2021, in Orinda, California
Professor Richard Cogdell is plant molecular biologist and holds the Hooker Chair of Botany at the University of Glasgow. Cogdell is the director of Glasgow Biomedical Research Centre, with a principal research interest in the structure and function of purple bacterial photosynthetic membrane proteins. Cogdell has authored over 250 peer-reviewed journal articles, and was a member of the Council of the BBSRC from 2014 to 2018.
Spheroidene is a carotenoid pigment. It is a component of the photosynthetic reaction center of certain purple bacteria of the Rhodospirillaceae family, including Rhodobacter sphaeroides and Rhodopseudomonas sphaeroides. Like other carotenoids, it is a tetraterpenoid. In purified form, it is a brick-red solid soluble in benzene.









