
Delta waves are high amplitude neural oscillations with a frequency between 0.5 and 4 hertz. Delta waves, like other brain waves, can be recorded with electroencephalography (EEG) and are usually associated with the deep stage 3 of NREM sleep, also known as slow-wave sleep (SWS), and aid in characterizing the depth of sleep. Suppression of delta waves leads to inability of body rejuvenation, brain revitalization and poor sleep.

A brain–computer interface (BCI), sometimes called a brain–machine interface (BMI), is a direct communication pathway between the brain's electrical activity and an external device, most commonly a computer or robotic limb. BCIs are often directed at researching, mapping, assisting, augmenting, or repairing human cognitive or sensory-motor functions. Implementations of BCIs range from non-invasive and partially invasive to invasive, based on how close electrodes get to brain tissue.
A mirror neuron is a neuron that fires both when an animal acts and when the animal observes the same action performed by another. Thus, the neuron "mirrors" the behavior of the other, as though the observer were itself acting. Such neurons have been directly observed in human and primate species, and in birds.

The reticular formation is a set of interconnected nuclei that are located throughout the brainstem. It is not anatomically well defined, because it includes neurons located in different parts of the brain. The neurons of the reticular formation make up a complex set of networks in the core of the brainstem that extend from the upper part of the midbrain to the lower part of the medulla oblongata. The reticular formation includes ascending pathways to the cortex in the ascending reticular activating system (ARAS) and descending pathways to the spinal cord via the reticulospinal tracts.
A gamma wave or gamma Rhythm is a pattern of neural oscillation in humans with a frequency between 25 and 140 Hz, the 40-Hz point being of particular interest. Gamma rhythms are correlated with large scale brain network activity and cognitive phenomena such as working memory, attention, and perceptual grouping, and can be increased in amplitude via meditation or neurostimulation. Altered gamma activity has been observed in many mood and cognitive disorders such as Alzheimer's disease, epilepsy, and schizophrenia.
The sensorimotor rhythm (SMR) is a brain wave. It is an oscillatory idle rhythm of synchronized electric brain activity. It appears in spindles in recordings of EEG, MEG, and ECoG over the sensorimotor cortex. For most individuals, the frequency of the SMR is in the range of 13 to 15 Hz.
Beta waves, or beta rhythm, are a neural oscillation (brainwave) in the brain with a frequency range of between 12.5 and 30 Hz. Beta waves can be split into three sections: Low Beta Waves ; Beta Waves ; and High Beta Waves. Beta states are the states associated with normal waking consciousness.
Alpha waves, or the alpha rhythm, are neural oscillations in the frequency range of 8–12 Hz likely originating from the synchronous and coherent electrical activity of thalamic pacemaker cells in humans. Historically, they are also called "Berger's waves" after Hans Berger, who first described them when he invented the EEG in 1924.
Brainwave entrainment, also referred to as brainwave synchronization or neural entrainment, refers to the observation that brainwaves will naturally synchronize to the rhythm of periodic external stimuli, such as flickering lights, speech, music, or tactile stimuli.

Neural oscillations, or brainwaves, are rhythmic or repetitive patterns of neural activity in the central nervous system. Neural tissue can generate oscillatory activity in many ways, driven either by mechanisms within individual neurons or by interactions between neurons. In individual neurons, oscillations can appear either as oscillations in membrane potential or as rhythmic patterns of action potentials, which then produce oscillatory activation of post-synaptic neurons. At the level of neural ensembles, synchronized activity of large numbers of neurons can give rise to macroscopic oscillations, which can be observed in an electroencephalogram. Oscillatory activity in groups of neurons generally arises from feedback connections between the neurons that result in the synchronization of their firing patterns. The interaction between neurons can give rise to oscillations at a different frequency than the firing frequency of individual neurons. A well-known example of macroscopic neural oscillations is alpha activity.
Theta waves generate the theta rhythm, a neural oscillation in the brain that underlies various aspects of cognition and behavior, including learning, memory, and spatial navigation in many animals. It can be recorded using various electrophysiological methods, such as electroencephalogram (EEG), recorded either from inside the brain or from electrodes attached to the scalp.
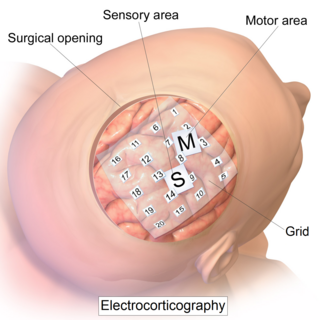
Electrocorticography (ECoG), or intracranial electroencephalography (iEEG), is a type of electrophysiological monitoring that uses electrodes placed directly on the exposed surface of the brain to record electrical activity from the cerebral cortex. In contrast, conventional electroencephalography (EEG) electrodes monitor this activity from outside the skull. ECoG may be performed either in the operating room during surgery or outside of surgery. Because a craniotomy is required to implant the electrode grid, ECoG is an invasive procedure.
The phi complex is a brain rhythm in the awake human brain that appears to serve various social functions. Phi is one of several brain rhythms in the awake human brain that coordinate human behavior. "Phi" operates in the 10-Hz band, and is located above the right centro-parietal cortex. It consists of two components, one favoring independent behaviors, and the other favoring interpersonal coordination between people.
Recurrent thalamo-cortical resonance is an observed phenomenon of oscillatory neural activity between the thalamus and various cortical regions of the brain. It is proposed by Rodolfo Llinas and others as a theory for the integration of sensory information into the whole of perception in the brain. Thalamocortical oscillation is proposed to be a mechanism of synchronization between different cortical regions of the brain, a process known as temporal binding. This is possible through the existence of thalamocortical networks, groupings of thalamic and cortical cells that exhibit oscillatory properties.
Ponto-geniculo-occipital waves or PGO waves are distinctive wave forms of propagating activity between three key brain regions: the pons, lateral geniculate nucleus, and occipital lobe; specifically, they are phasic field potentials. These waves can be recorded from any of these three structures during and immediately before REM sleep. The waves begin as electrical pulses from the pons, then move to the lateral geniculate nucleus residing in the thalamus, and end in the primary visual cortex of the occipital lobe. The appearances of these waves are most prominent in the period right before REM sleep, albeit they have been recorded during wakefulness as well. They are theorized to be intricately involved with eye movement of both wake and sleep cycles in many different animals.

Electroencephalography (EEG) is a method to record an electrogram of the electrical activity on the scalp that has been shown to represent the macroscopic activity of the surface layer of the brain underneath. It is typically non-invasive, with the electrodes placed along the scalp. Electrocorticography, involving invasive electrodes, is sometimes called "intracranial EEG".
The neuroscience of music is the scientific study of brain-based mechanisms involved in the cognitive processes underlying music. These behaviours include music listening, performing, composing, reading, writing, and ancillary activities. It also is increasingly concerned with the brain basis for musical aesthetics and musical emotion. Scientists working in this field may have training in cognitive neuroscience, neurology, neuroanatomy, psychology, music theory, computer science, and other relevant fields.

Meditation and its effect on brain activity and the central nervous system became a focus of collaborative research in neuroscience, psychology and neurobiology during the latter half of the 20th century. Research on meditation sought to define and characterize various practices. Meditation's effect on the brain can be broken up into two categories: state changes and trait changes, respectively alterations in brain activities during the act of meditating and changes that are the outcome of long-term practice.
Pain empathy is a specific variety of empathy that involves recognizing and understanding another person's pain.

Fetal electroencephalography, also known as prenatal EEG includes any recording of electrical fluctuations arising from the brain of a fetus. Doctors and scientists use EEGs to detect and characterize brain activity, such as sleep states, potential seizures, or levels of a coma. EEG captures the electrical activity in the vicinity of the recording electrodes. The majority of the neural electrical activity arises from the flow of current from the cell bodies of pyramidal neurons to their apical dendrites, which become depolarized by excitatory inputs from other neurons. To record the most accurate signals, scientists try to minimize the distance between the recording electrode and the neural activity that they want to detect. Given the difficulty of attaching electrodes to a fetus inside a uterus, doctors and scientists use a variety of techniques to record fetal brain activity.









