
Antiviral drugs are a class of medication used for treating viral infections. Most antivirals target specific viruses, while a broad-spectrum antiviral is effective against a wide range of viruses. Antiviral drugs are one class of antimicrobials, a larger group which also includes antibiotic, antifungal and antiparasitic drugs, or antiviral drugs based on monoclonal antibodies. Most antivirals are considered relatively harmless to the host, and therefore can be used to treat infections. They should be distinguished from virucides, which are not medication but deactivate or destroy virus particles, either inside or outside the body. Natural virucides are produced by some plants such as eucalyptus and Australian tea trees.

Ribavirin, also known as tribavirin, is an antiviral medication used to treat RSV infection, hepatitis C and some viral hemorrhagic fevers. For hepatitis C, it is used in combination with other medications such as simeprevir, sofosbuvir, peginterferon alfa-2b or peginterferon alfa-2a. Among the viral hemorrhagic fevers it is used for Lassa fever, Crimean–Congo hemorrhagic fever, and Hantavirus infection but should not be used for Ebola or Marburg infections. Ribavirin is taken by mouth or inhaled.

Entecavir (ETV), sold under the brand name Baraclude, is an antiviral medication used in the treatment of hepatitis B virus (HBV) infection. In those with both HIV/AIDS and HBV antiretroviral medication should also be used. Entecavir is taken by mouth as a tablet or solution.

Milan Panić is a Serbian-American businessman, humanitarian and former politician. He served as the Prime Minister of the Federal Republic of Yugoslavia from 1992 to 1993. During and after his time as Prime Minister, he campaigned for peace and democracy in the Balkan region. He ran for President of Serbia in 1992, ultimately coming in second to Slobodan Milošević in an election marked by allegations of media and vote tampering by the ruling party. Panić became Prime Minister of Yugoslavia while an American citizen. The legality of retaining US citizenship while accepting this office has been questioned based on a Constitutional prohibition of a US citizen accepting office on behalf of a foreign nation. Panić is the first US citizen to occupy a high-level political position in a foreign country since Golda Meir. Panić is also the first civilian to serve as Defence Minister in the history of Serbia.

Umifenovir, sold under the brand name Arbidol, is an antiviral medication for the treatment of influenza and COVID infections used in Russia and China. The drug is manufactured by Pharmstandard. It is not approved by the U.S. Food and Drug Administration (FDA) for the treatment or prevention of influenza.

Buformin (1-butylbiguanide) is an oral antidiabetic drug of the biguanide class, chemically related to metformin and phenformin. Buformin was marketed by German pharmaceutical company Grünenthal as Silubin.

Telaprevir (VX-950), marketed under the brand names Incivek and Incivo, is a pharmaceutical drug for the treatment of hepatitis C co-developed by Vertex Pharmaceuticals and Johnson & Johnson. It is a member of a class of antiviral drugs known as protease inhibitors. Specifically, telaprevir inhibits the hepatitis C viral enzyme NS3/4A serine protease. Telaprevir is only indicated for use against hepatitis C genotype 1 viral infections and has not been proven to be safe or effective when used for other genotypes of the virus. The standard therapy of pegylated interferon and ribavirin is less effective than telaprevir in those with genotype 1.
CCR5 receptor antagonists are a class of small molecules that antagonize the CCR5 receptor. The C-C motif chemokine receptor CCR5 is involved in the process by which HIV, the virus that causes AIDS, enters cells. Hence antagonists of this receptor are entry inhibitors and have potential therapeutic applications in the treatment of HIV infections.

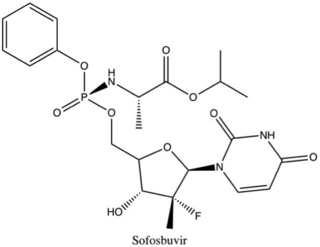
Sofosbuvir, sold under the brand name Sovaldi among others, is a medication used to treat hepatitis C. It is taken by mouth.

Daclatasvir, sold under the brand name Daklinza, is an antiviral medication used in combination with other medications to treat hepatitis C (HCV). The other medications used in combination include sofosbuvir, ribavirin, and interferon, vary depending on the virus type and whether the person has cirrhosis. It is taken by mouth.

Simeprevir, sold under the brand name Olysio among others, is a medication used in combination with other medications for the treatment of hepatitis C. It is specifically used for hepatitis C genotype 1 and 4. Medications it is used with include sofosbuvir or ribavirin and peginterferon-alfa. Cure rates are in 80s to 90s percent. It may be used in those who also have HIV/AIDS. It is taken by mouth once daily for typically 12 weeks.
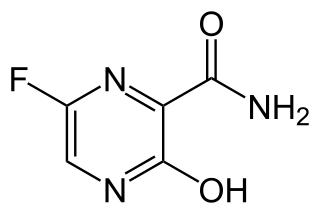
Favipiravir, sold under the brand name Avigan among others, is an antiviral medication used to treat influenza in Japan. It is also being studied to treat a number of other viral infections, including SARS-CoV-2. Like the experimental antiviral drugs T-1105 and T-1106, it is a pyrazinecarboxamide derivative.
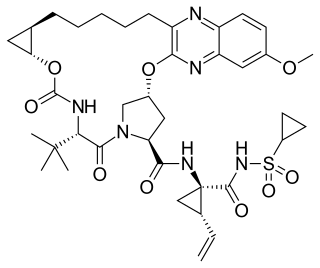
Grazoprevir is a drug approved for the treatment of hepatitis C. It was developed by Merck and completed Phase III trials, used in combination with the NS5A replication complex inhibitor elbasvir under the trade name Zepatier, either with or without ribavirin.
Samatasvir (IDX-719) is an experimental drug for the treatment of hepatitis C. It was originally developed by Idenix, and development has been continued by Merck & Co. following their acquisition of Idenix. Samatasvir has shown good results in Phase II trials.

Riamilovir is a broad-spectrum antiviral drug developed in Russia through a joint effort of Ural Federal University, Russian Academy of Sciences, Ural Center for Biopharma Technologies and Medsintez Pharmaceutical. It has a novel triazolotriazine core, which represents a new structural class of non-nucleoside antiviral drugs.

Daclatasvir/sofosbuvir is a two-drug combination for the treatment of hepatitis C. It is given as a single daily pill containing daclatasvir, a viral NS5A inhibitor, and sofosbuvir, a nucleotide inhibitor of the viral RNA polymerase NS5B.
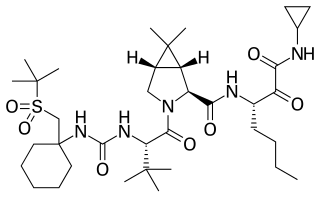
Narlaprevir, is an inhibitor of NS3/4A serine protease, intended for the treatment of chronic hepatitis C caused by genotype 1 virus in combination with other antiviral drugs.
Non-structural protein 5B (NS5B) inhibitors are a class of direct-acting antivirals widely used in the treatment of chronic hepatitis C. Depending on site of action and chemical composition, NS5B inhibitors may be categorized into three classes—nucleoside active site inhibitors (NIs), non-nucleoside allosteric inhibitors, and pyrophosphate analogues. Subsequently, all three classes are then subclassified. All inhibit RNA synthesis by NS5B but at different stages/sites resulting in inability of viral RNA replication. Expression of direct-acting NS5B inhibitors does not take place in cells that are not infected by hepatitis C virus, which seems to be beneficial for this class of drugs.

Lobucavir is an antiviral drug that shows broad-spectrum activity against herpesviruses, hepatitis B and other hepadnaviruses, HIV/AIDS and cytomegalovirus. It initially demonstrated positive results in human clinical trials against hepatitis B with minimal adverse effects but was discontinued from further development following the discovery of increased risk of cancer associated with long-term use in mice. Although this carcinogenic risk is present in other antiviral drugs, such as zidovudine and ganciclovir that have been approved for clinical use, development was halted by Bristol-Myers Squibb, its manufacturer.

Bemnifosbuvir is an antiviral drug invented by Atea Pharmaceuticals and licensed to Roche for clinical development, a novel nucleotide analog prodrug originally developed for the treatment of hepatitis C. Bemnifosbuvir is the orally bioavailable hemisulfate salt of AT-511, which is metabolized in several steps to the active nucleotide triphosphate AT-9010, acting as an RNA polymerase inhibitor and thereby interfering with viral replication. Bemnifosbuvir has been researched for the treatment of coronavirus diseases such as that produced by SARS-CoV-2. It showed good results in early clinical trials but had inconsistent results at later stages, so the planned Phase 3 trials are being redesigned and results are not expected until late 2022.



















