Tooth decay, also known as cavities or caries, is the breakdown of teeth due to acids produced by bacteria. The cavities may be a number of different colors from yellow to black. Symptoms may include pain and difficulty with eating. Complications may include inflammation of the tissue around the tooth, tooth loss and infection or abscess formation.
Dental products are specially fabricated materials, designed for use in dentistry. There are many different types of dental products, and their characteristics vary according to their intended purpose.
Dental restoration, dental fillings, or simply fillings are treatments used to restore the function, integrity, and morphology of missing tooth structure resulting from caries or external trauma as well as to the replacement of such structure supported by dental implants. They are of two broad types—direct and indirect—and are further classified by location and size. A root canal filling, for example, is a restorative technique used to fill the space where the dental pulp normally resides.
Dental sealants are a dental treatment intended to prevent tooth decay. Teeth have recesses on their biting surfaces; the back teeth have fissures (grooves) and some front teeth have cingulum pits. It is these pits and fissures that are most vulnerable to tooth decay because food and bacteria stick in them and because they are hard-to-clean areas. Dental sealants are materials placed in these pits and fissures to fill them in, creating a smooth surface which is easy to clean. Dental sealants are mainly used in children who are at higher risk of tooth decay, and are usually placed as soon as the adult molar teeth come through.

Dental composite resins are dental cements made of synthetic resins. Synthetic resins evolved as restorative materials since they were insoluble, of good tooth-like appearance, insensitive to dehydration, easy to manipulate and inexpensive. Composite resins are most commonly composed of Bis-GMA and other dimethacrylate monomers, a filler material such as silica and in most applications, a photoinitiator. Dimethylglyoxime is also commonly added to achieve certain physical properties such as flow-ability. Further tailoring of physical properties is achieved by formulating unique concentrations of each constituent.

Abrasion is the non-carious, mechanical wear of tooth from interaction with objects other than tooth-tooth contact. It most commonly affects the premolars and canines, usually along the cervical margins. Based on clinical surveys, studies have shown that abrasion is the most common but not the sole aetiological factor for development of non-carious cervical lesions (NCCL) and is most frequently caused by incorrect toothbrushing technique.

In dentistry, inlays and onlays are used to fill cavities, and then cemented in place in the tooth. This is an alternative to a direct restoration, made out of composite, amalgam or glass ionomer, that is built up within the mouth.

Also known as a "bonderizer" bonding agents are resin materials used to make a dental composite filling material adhere to both dentin and enamel.
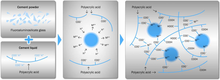
A luting agent is a dental cement connecting the underlying tooth structure to a fixed prosthesis. To lute means to glue two different structures together. There are two major purposes of luting agents in dentistry – to secure a cast restoration in fixed prosthodontics, and to keep orthodontic bands and appliances in situ.

In dentistry, amalgam is a liquid mercury and metal alloy mixture used to fill cavities caused by tooth decay.

Fluoride varnish is a highly concentrated form of fluoride which is applied to the tooth's surface, by a dentist, dental hygienist or other health care professional, as a type of topical fluoride therapy. It is not a permanent varnish but due to its adherent nature it is able to stay in contact with the tooth surface for several hours. It may be applied to the enamel, dentine or cementum of the tooth and can be used to help prevent decay, remineralise the tooth surface and to treat dentine hypersensitivity. There are more than 30 fluoride-containing varnish products on the market today, and they have varying compositions and delivery systems. These compositional differences lead to widely variable pharmacokinetics, the effects of which remain largely untested clinically.
Dental cements have a wide range of dental and orthodontic applications. Common uses include temporary restoration of teeth, cavity linings to provide pulpal protection, sedation or insulation and cementing fixed prosthodontic appliances. Recent uses of dental cement also include two-photon calcium imaging of neuronal activity in brains of animal models in basic experimental neuroscience.
Minimal intervention dentistry is a modern dental practice designed around the principal aim of preservation of as much of the natural tooth structure as possible. It uses a disease-centric philosophy that directs attention to first control and management of the disease that causes tooth decay—dental caries—and then to relief of the residual symptoms it has left behind—the decayed teeth. The approach uses similar principles for prevention of future caries, and is intended to be a complete management solution for tooth decay.

Enamel hypoplasia is a defect of the teeth in which the enamel is deficient in quantity, caused by defective enamel matrix formation during enamel development, as a result of inherited and acquired systemic condition(s). It can be identified as missing tooth structure and may manifest as pits or grooves in the crown of the affected teeth, and in extreme cases, some portions of the crown of the tooth may have no enamel, exposing the dentin. It may be generalized across the dentition or localized to a few teeth. Defects are categorized by shape or location. Common categories are pit-form, plane-form, linear-form, and localised enamel hypoplasia. Hypoplastic lesions are found in areas of the teeth where the enamel was being actively formed during a systemic or local disturbance. Since the formation of enamel extends over a long period of time, defects may be confined to one well-defined area of the affected teeth. Knowledge of chronological development of deciduous and permanent teeth makes it possible to determine the approximate time at which the developmental disturbance occurred. Enamel hypoplasia varies substantially among populations and can be used to infer health and behavioural impacts from the past. Defects have also been found in a variety of non-human animals.
Dental compomers, also known as polyacid-modified resin composite, are used in dentistry as a filling material. They were introduced in the early 1990s as a hybrid of two other dental materials, dental composites and glass ionomer cement, in an effort to combine their desirable properties: aesthetics for dental composites and the fluoride releasing ability for glass ionomer cements.

Pulp capping is a technique used in dental restorations to prevent the dental pulp from necrosis, after being exposed, or nearly exposed during a cavity preparation, from a traumatic injury, or by a deep cavity that reaches the center of the tooth causing the pulp to die. When dental caries is removed from a tooth, all or most of the infected and softened enamel and dentin are removed. This can lead to the pulp of the tooth either being exposed or nearly exposed which causes pulpitis (inflammation). Pulpitis, in turn, can become irreversible, leading to pain and pulp necrosis, and necessitating either root canal treatment or extraction. The ultimate goal of pulp capping or stepwise caries removal is to protect a healthy dental pulp and avoid the need for root canal therapy.
Silver diammine fluoride (SDF), also known as silver diamine fluoride in most of the dental literature, is a topical medication used to treat and prevent dental caries and relieve dentinal hypersensitivity. It is a colorless or blue-tinted, odourless liquid composed of silver, ammonium and fluoride ions at a pH of 10.4 or 13. Ammonia compounds reduce the oxidative potential of SDF, increase its stability and helps to maintain a constant concentration over a period of time, rendering it safe for use in the mouth. Silver and fluoride ions possess antimicrobial properties and are used in the remineralization of enamel and dentin on teeth for preventing and arresting dental caries.

Molar incisor hypomineralisation (MIH) is a type of enamel defect affecting, as the name suggests, the first molars and incisors in the permanent dentition. MIH is considered a worldwide problem with a global prevalence of 12.9% and is usually identified in children under 10 years old. This developmental condition is caused by the lack of mineralisation of enamel during its maturation phase, due to interruption to the function of ameloblasts. Peri- and post-natal factors including premature birth, certain medical conditions, fever and antibiotic use have been found to be associated with development of MIH. Recent studies have suggested the role of genetics and/or epigenetic changes to be contributors of MIH development. However, further studies on the aetiology of MIH are required because it is believed to be multifactorial.

Non-carious cervical lesions (NCCLs) are a group of lesions that are characterised by a loss of hard dental tissue at the cementoenamel junction (CEJ) region at the neck of the tooth, without the action of microorganisms or inflammatory processes. These lesions vary in shape from regular depressions that look like a dome or a cup, to deep wedge-shaped defects with the apex pointing inwards. NCCLs can occur either above or below the level of the gum, at any of the surfaces of the teeth.
Atraumatic restorative treatment (ART) is a method for cleaning out tooth decay from teeth using only hand instruments and placing a filling. It does not use rotary dental instruments to prepare the tooth and can be performed in settings with no access to dental equipment. No drilling or local anaesthetic injections are required. ART is considered a conservative approach, not only because it removes the decayed tissue with hand instruments, avoiding removing more tissue necessary which preserves as much tooth structure as possible, but also because it avoids pulp irritation and minimises patient discomfort. ART can be used for small, medium and deep cavities caused by dental caries.