Related Research Articles

Bradykinin (BK) (Greek brady-, slow; -kinin, kīn(eîn) to move) is a peptide that promotes inflammation. It causes arterioles to dilate (enlarge) via the release of prostacyclin, nitric oxide, and endothelium-derived hyperpolarizing factor and makes veins constrict, via prostaglandin F2, thereby leading to leakage into capillary beds, due to the increased pressure in the capillaries. Bradykinin consists of nine amino acids, and is a physiologically and pharmacologically active peptide of the kinin group of proteins.
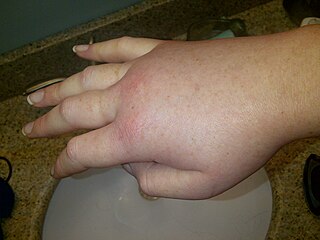
Hereditary angioedema (HAE) is a disorder that results in recurrent attacks of severe swelling. The swelling most commonly affects the arms, legs, face, intestinal tract, and airway. If the intestinal tract is affected, abdominal pain and vomiting may occur. Swelling of the airway can result in its obstruction and trouble breathing. Without preventive treatment, attacks typically occur every two weeks and last for a few days.

Angioedema is an area of swelling (edema) of the lower layer of skin and tissue just under the skin or mucous membranes. The swelling may occur in the face, tongue, larynx, abdomen, or arms and legs. Often it is associated with hives, which are swelling within the upper skin. Onset is typically over minutes to hours.
The kinin–kallikrein system or simply kinin system is a poorly understood hormonal system with limited available research. It consists of blood proteins that play a role in inflammation, blood pressure control, coagulation and pain. Its important mediators bradykinin and kallidin are vasodilators and act on many cell types. Clinical symptoms include marked weakness, tachycardia, fever, leukocytosis and acceleration of ESR.
Kallikreins are a subgroup of serine proteases, enzymes capable of cleaving peptide bonds in proteins. In humans, plasma kallikrein has no known paralogue, while tissue kallikrein-related peptidases (KLKs) encode a family of fifteen closely related serine proteases. These genes are localised to chromosome 19q13, forming the largest contiguous cluster of proteases within the human genome. Kallikreins are responsible for the coordination of various physiological functions including blood pressure, semen liquefaction and skin desquamation.
Kininogens are precursor proteins for kinins, biologically active polypeptides involved in blood coagulation, vasodilation, smooth muscle contraction, inflammatory regulation, and the regulation of the cardiovascular and renal systems.
Mepolizumab, sold under the brand name Nucala by GlaxoSmithKline, is a humanized monoclonal antibody used for the treatment of severe eosinophilic asthma, eosinophilic granulomatosis, and hypereosinophilic syndrome (HES). It recognizes and blocks interleukin-5 (IL-5), a signalling protein of the immune system.

Icatibant, sold under the brand name Firazyr, is a medication for the symptomatic treatment of acute attacks of hereditary angioedema (HAE) in adults with C1-esterase-inhibitor deficiency. It is not effective in angioedema caused by medication from the ACE inhibitor class.
Ecallantide is a drug used for the treatment of hereditary angioedema (HAE) and in the prevention of blood loss in cardiothoracic surgery. It is an inhibitor of the protein kallikrein and a 60-amino acid polypeptide which was developed from a Kunitz domain through phage display to mimic antibodies inhibiting kallikrein.
BioCryst Pharmaceuticals, Inc. is an American pharmaceutical company headquartered in Durham, North Carolina. The company is a late stage biotech company that focuses on oral drugs for rare and serious diseases. BioCryst's antiviral drug peramivir (Rapivab) was approved by FDA in December 2014. It has also been approved in Japan, Korea, and China.
Ixekizumab, sold under the brand name Taltz, is an injectable medication for the treatment of autoimmune diseases. Chemically, it is a form of a humanized monoclonal antibody. The substance acts by binding interleukin 17A and neutralizing it, reducing inflammation.

Baricitinib, sold under the brand name Olumiant among others, is an immunomodulatory medication used for the treatment of rheumatoid arthritis, alopecia areata, and COVID-19. It acts as an inhibitor of janus kinase (JAK), blocking the subtypes JAK1 and JAK2.
Risankizumab, sold under the brand name Skyrizi, is a humanized monoclonal antibody used for the treatment of plaque psoriasis, psoriatic arthritis, and Crohn's disease. It is designed to target interleukin 23A (IL-23A). It is given by subcutaneous injection.

Upadacitinib, sold under the brand name Rinvoq, is a medication used for the treatment of rheumatoid arthritis, psoriatic arthritis, atopic dermatitis, ulcerative colitis, Crohn's disease, ankylosing spondylitis, and axial spondyloarthritis. Upadacitinib is a Janus kinase (JAK) inhibitor that works by blocking the action of enzymes called Janus kinases. These enzymes are involved in setting up processes that lead to inflammation, and blocking their effect brings inflammation in the joints under control.
Lecanemab, sold under the brand name Leqembi, is a monoclonal antibody medication used for the treatment of Alzheimer's disease. Lecanemab is an amyloid beta-directed antibody. It is given via intravenous infusion. The most common side effects of lecanemab include headache, infusion-related reactions and amyloid-related imaging abnormalities, a side effect known to occur with the class of antibodies targeting amyloid.
Lumasiran, sold under the brand name Oxlumo, is a medication for the treatment of primary hyperoxaluria type 1 (PH1).

Berotralstat, sold under the brand name Orladeyo, is a medication used to prevent attacks of hereditary angioedema (HAE) in people aged twelve years and older.
Dasiglucagon, sold under the brand name Zegalogue, is a medication used to treat severe hypoglycemia in people with diabetes.

Tirzepatide, sold under the brand name Mounjaro among others, is an antidiabetic medication used for the treatment of type 2 diabetes and for weight loss. Tirzepatide is administered through subcutaneous injection.
Vutrisiran, previously known as (ALN-TTRSC02), sold under the brand name Amvuttra, is a medication used for the treatment of the polyneuropathy of hereditary transthyretin-mediated (hATTR) amyloidosis in adults. It is a double stranded small interfering RNA (siRNA) that interferes with the expression of the transthyretin (TTR) gene. Transthyretin is a serum protein made in the liver whose major function is transport of vitamin A and thyroxine. Rare mutations in the transthyretin gene result in accumulation of large amyloid deposits of misfolded transthyretin molecules most prominently in peripheral nerves and the heart. Patients with hATTR typically present with polyneuropathy or autonomic dysfunction followed by cardiomyopathy which, if untreated, is fatal within 5 to 10 years.
References
- ↑ "Lanadelumab (Takhzyro) Use During Pregnancy". Drugs.com. 19 September 2018. Archived from the original on 20 October 2020. Retrieved 4 September 2020.
- ↑ "Takhzyro Product information". Health Canada. 25 April 2012. Archived from the original on 30 May 2022. Retrieved 29 May 2022.
- ↑ "Takhzyro 300 mg solution for injection in pre-filled syringe - Patient Information Leaflet (PIL)". (emc). 1 July 2022. Archived from the original on 19 April 2021. Retrieved 1 July 2022.
- 1 2 3 "Takhzyro- lanadelumab-flyo solution Takhzyro- lanadelumab-flyo injection, solution". DailyMed. 17 February 2022. Archived from the original on 20 January 2022. Retrieved 30 May 2022.
- ↑ "Takhzyro EPAR". European Medicines Agency. 17 October 2018. Archived from the original on 19 October 2021. Retrieved 30 May 2022.
- ↑ Kenniston JA, Faucette RR, Martik D, Comeau SR, Lindberg AP, Kopacz KJ, et al. (August 2014). "Inhibition of plasma kallikrein by a highly specific active site blocking antibody". The Journal of Biological Chemistry. 289 (34): 23596–23608. doi: 10.1074/jbc.M114.569061 . PMC 4156074 . PMID 24970892.
- 1 2 3 4 5 6 7 Banerji A, Busse P, Shennak M, Lumry W, Davis-Lorton M, Wedner HJ, et al. (February 2017). "Inhibiting Plasma Kallikrein for Hereditary Angioedema Prophylaxis" (PDF). The New England Journal of Medicine. 376 (8): 717–728. doi:10.1056/NEJMoa1605767. hdl: 2434/550555 . PMID 28225674. S2CID 54486011. Archived (PDF) from the original on 31 October 2021. Retrieved 2 September 2019.
- ↑ Statement On A Nonproprietary Name Adopted By The USAN Council - Lanadelumab Archived 2 April 2016 at the Wayback Machine , American Medical Association .
- ↑ World Health Organization (2015). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 114" (PDF). WHO Drug Information. 29 (4). Archived (PDF) from the original on 7 August 2020. Retrieved 5 October 2020.
- 1 2 3 4 "FDA approves new treatment for rare hereditary disease". U.S. Food and Drug Administration. 30 September 2021. Archived from the original on 19 January 2021. Retrieved 15 March 2021.
 This article incorporates text from this source, which is in the public domain .
This article incorporates text from this source, which is in the public domain . - 1 2 3 "www.takhzyro.com". Archived from the original on 3 August 2021. Retrieved 1 July 2022.
- 1 2 3 "FDA approves new treatment for rare hereditary disease". U.S. Food and Drug Administration (Press release). 30 September 2021. Archived from the original on 7 February 2023. Retrieved 10 February 2023.
 This article incorporates text from this source, which is in the public domain .
This article incorporates text from this source, which is in the public domain . - ↑ "Drug Trials Snapshots: Takhzyro". U.S. Food and Drug Administration (FDA). 9 February 2019. Archived from the original on 30 September 2022. Retrieved 10 February 2023.
- ↑ "U.S. FDA Approves Takeda's Takhzyro (lanadelumab-flyo) to Prevent Hereditary Angioedema (HAE) Attacks in Children 2 Years of Age and Older" (Press release). Takeda. 4 February 2023. Archived from the original on 6 February 2023. Retrieved 10 February 2023.
- 1 2 3 4 5 6 "Efficacy and Safety Study of DX-2930 to Prevent Acute Angioedema Attacks in Patients With Type I and Type II HAE". 13 May 2021. Archived from the original on 19 April 2021. Retrieved 24 March 2017.
- ↑ Banerji A, Riedl MA, Bernstein JA, Cicardi M, Longhurst HJ, Zuraw BL, et al. (November 2018). "Effect of Lanadelumab Compared With Placebo on Prevention of Hereditary Angioedema Attacks: A Randomized Clinical Trial". JAMA. 320 (20): 2108–2121. doi:10.1001/jama.2018.16773. PMC 6583584 . PMID 30480729. S2CID 53757796. Archived from the original on 7 August 2020. Retrieved 15 March 2021.
- 1 2 3 4 5 "Clinical Pharmacology Database". Archived from the original on 31 October 2021. Retrieved 1 July 2022.
- ↑ Chyung Y, Vince B, Iarrobino R, Sexton D, Kenniston J, Faucette R, et al. (October 2014). "A phase 1 study investigating DX-2930 in healthy subjects". Annals of Allergy, Asthma & Immunology. 113 (4): 460–6.e2. doi: 10.1016/j.anai.2014.05.028 . PMID 24980392.
- ↑ Clinical trial number NCT01923207 for "A Single Increasing Dose Study to Assess Safety and Tolerability of DX-2930 in Healthy Subjects" at ClinicalTrials.gov
- ↑ Clinical trial number NCT02093923 for "Double-Blind, Multiple Ascending Dose Study to Assess Safety, Tolerability and Pharmacokinetics of DX-2930 in Hereditary Angioedema (HAE) Subjects" at ClinicalTrials.gov
- ↑ "Dyax Corp. Receives FDA Breakthrough Therapy Designation for DX-2930 for Prevention of Attacks of Hereditary Angioedema". www.businesswire.com. 7 July 2015. Archived from the original on 25 March 2017. Retrieved 24 March 2017.
- 1 2 3 4 5 6 "Lanadelumab - Takeda". Adis Insight. Springer Nature Switzerland AG. Archived from the original on 25 March 2017. Retrieved 24 March 2017.