
MN 18 is an indazole-based synthetic cannabinoid that is an agonist for the cannabinoid receptors, with Ki values of 45.72 nM at CB1 and 11.098 nM at CB2 and EC50 values of 2.028 nM at CB1 and 1.233 nM at CB2, and has been sold online as a designer drug. It is the indazole core analogue of NNE1. Given the known metabolic liberation (and presence as an impurity) of amantadine in the related compound APINACA, it is suspected that metabolic hydrolysis of the amide group of MN-18 may release 1-naphthylamine, a known carcinogen. MN-18 metabolism has been described in literature.

AB-CHMINACA is an indazole-based synthetic cannabinoid. It is a potent agonist of the CB1 receptor (Ki = 0.78 nM) and CB2 receptor (Ki = 0.45 nM) and fully substitutes for Δ9-THC in rat discrimination studies, while being 16x more potent. Continuing the trend seen in other cannabinoids of this generation, such as AB-FUBINACA and AB-PINACA, it contains a valine amino acid amide residue as part of its structure, where older cannabinoids contained a naphthyl or adamantane residue.
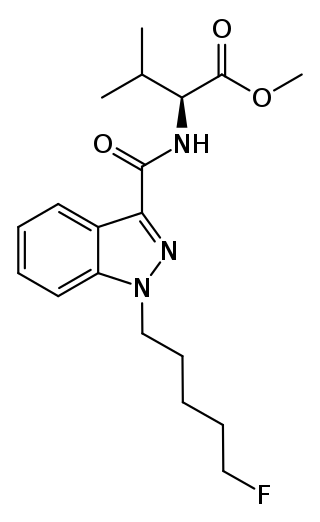
5F-AMB (also known as 5F-MMB-PINACA and 5F-AMB-PINACA) is an indazole-based synthetic cannabinoid from the indazole-3-carboxamide family, which has been used as an active ingredient in synthetic cannabis products. It was first identified in Japan in early 2014. Although only very little pharmacological information about 5F-AMB itself exists, its 4-cyanobutyl analogue (instead of 5-fluoropentyl) has been reported to be a potent agonist for the CB1 receptor (KI = 0.7 nM).
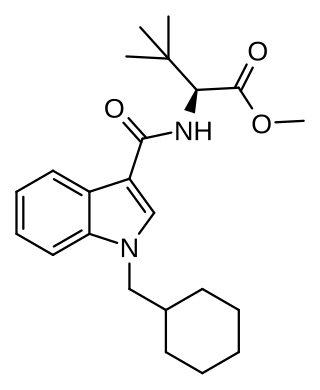
MDMB-CHMICA is an indole-based synthetic cannabinoid that is a potent agonist of the CB1 receptor and has been sold online as a designer drug. While MDMB-CHMICA was initially sold under the name "MMB-CHMINACA", the compound corresponding to this code name (i.e. the isopropyl instead of t-butyl analogue of MDMB-CHMINACA) has been identified on the designer drug market in 2015 as AMB-CHMINACA.
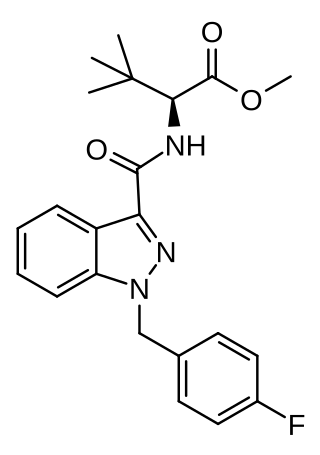
MDMB-FUBINACA (also known as MDMB(N)-Bz-F and FUB-MDMB) is an indazole-based synthetic cannabinoid that is a potent agonist for the cannabinoid receptors, with Ki values of 1.14 nM at CB1 and 0.1228 nM at CB2 and EC50 values of 0.2668 nM at CB1 and 0.1411 nM at CB2, and has been sold online as a designer drug. Its benzyl analogue (instead of 4-fluorobenzyl) has been reported to be a potent agonist for the CB1 receptor (Ki = 0.14 nM, EC50 = 2.42 nM). The structure of MDMB-FUBINACA contains the amino acid, 3-methylvaline or tert-leucine methyl ester.
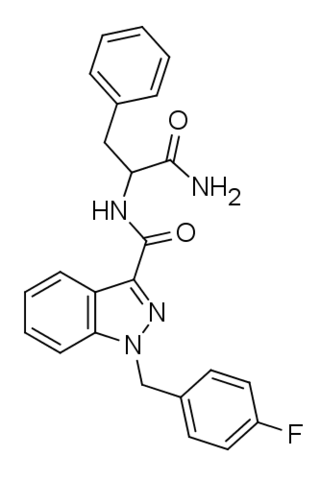
APP-FUBINACA is an indazole-based synthetic cannabinoid that has been sold online as a designer drug. Pharmacological testing showed APP-FUBINACA to have only moderate affinity for the CB1 receptor, with a Ki of 708 nM, while its EC50 was not tested. It contains a phenylalanine amino acid residue in its structure.

AMB-FUBINACA (also known as FUB-AMB and MMB-FUBINACA) is an indazole-based synthetic cannabinoid that is a potent agonist for the cannabinoid receptors, with Ki values of 10.04 nM at CB1 and 0.786 nM at CB2 and EC50 values of 0.5433 nM at CB1 and 0.1278 nM at CB2, and has been sold online as a designer drug. It was originally developed by Pfizer which described the compound in a patent in 2009, but was later abandoned and never tested on humans. AMB-FUBINACA was the most common synthetic cannabinoid identified in drug seizures by the Drug Enforcement Administration in 2017 and the first half of 2018.
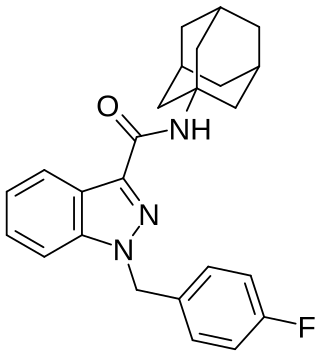
FUB-APINACA (also known as A-FUBINACA according to the EMCCDA framework for naming synthetic cannabinoids and FUB-AKB48) is an indazole-based synthetic cannabinoid that is presumed to be a potent agonist of the CB1 receptor and has been sold online as a designer drug. It is an analog of APINACA and 5F-APINACA where the pentyl chain has been replaced with fluorobenzyl.

5F-AB-FUPPYCA (also known as AZ-037) is a pyrazole-based synthetic cannabinoid that is presumed to be an agonist of the CB1 receptor and has been sold online as a designer drug. It was first detected by the EMCDDA as part of a seizure of 540 g white powder in France in February 2015.
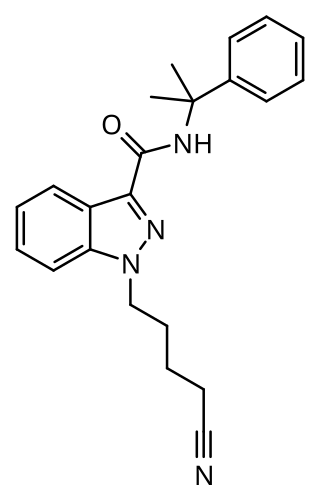
CUMYL-4CN-BINACA (also known as CUMYL-CYBINACA or SGT-78) is an indazole-3-carboxamide based synthetic cannabinoid that has been sold online as a designer drug. It is a potent agonist for cannabinoid receptors CB1 and CB2, with in vitro EC50 values of 0.58 nM and 6.12 nM, respectively. In mice, CUMYL-4CN-BINACA produces hypothermic and pro-convulsant effects via the CB1 receptor, and anecdotal reports suggest it has an active dose of around 0.1 mg in humans.

5F-CUMYL-P7AICA is a pyrrolo[2,3-b]pyridine-3-carboxamide based synthetic cannabinoid that has been sold as a designer drug. It was first identified by the EMCDDA in February 2015.

4F-MDMB-BINACA (also known as 4F-MDMB-BUTINACA or 4F-ADB) is an indazole-based synthetic cannabinoid from the indazole-3-carboxamide family. It has been used as an active ingredient in synthetic cannabis products and sold as a designer drug since late 2018. 4F-MDMB-BINACA is an agonist of the CB1 receptor (EC50 = 7.39 nM), though it is unclear whether it is selective for this target. In December 2019, the UNODC announced scheduling recommendations placing 4F-MDMB-BINACA into Schedule II throughout the world.

CUMYL-CBMICA (SGT-280) is an indole-3-carboxamide based synthetic cannabinoid receptor agonist which has been sold as a designer drug, first being identified in Germany in August 2019. Since the structure fell outside the German drug analogue law provisions at the time, an amendment was made to the law to expand the relevant definition, which came into effect in April 2020. It has been shown to act as a CB1 receptor agonist with an EC50 of 62.9nM.

ADB-BINACA is a cannabinoid designer drug that has been found as an ingredient in some synthetic cannabis products. It was originally developed by Pfizer as a potential analgesic, and is a potent agonist of the CB1 receptor with a binding affinity (Ki) of 0.33 nM and an EC50 of 14.7 nM.

ADB-BUTINACA is a synthetic cannabinoid compound which has been sold as a designer drug. It is a potent CB1 agonist, with a binding affinity of 0.29nM for CB1 and 0.91nM for CB2, and an EC50 of 6.36 nM for CB1.

ADB-HEXINACA is a cannabinoid designer drug that has been found as an ingredient in some synthetic cannabis products, first appearing in early 2021. It is a longer chain homologue of previously encountered synthetic cannabinoid compounds such as ADB-BUTINACA and ADB-PINACA.

ADB-4en-PINACA is a cannabinoid designer drug that has been found as an ingredient in some synthetic cannabis products, first appearing in early 2021. It is a reasonably potent cannabinoid agonist in vitro but has not been so widely sold as related compounds such as ADB-PINACA and MDMB-4en-PINACA.

MDMB-5Br-INACA is an indazole-3-carboxamide derivative which has been sold as a designer drug. Surprisingly it appears to produce psychoactive activity despite the lack of a "tail" group at the indazole 1-position, but is of relatively low potency and has been encountered being misrepresented as other illicit drugs such as MDMA.


















