
Magnesium is a chemical element; it has symbol Mg and atomic number 12. It is a shiny gray metal having a low density, low melting point and high chemical reactivity. Like the other alkaline earth metals it occurs naturally only in combination with other elements and it almost always has an oxidation state of +2. It reacts readily with air to form a thin passivation coating of magnesium oxide that inhibits further corrosion of the metal. The free metal burns with a brilliant-white light. The metal is obtained mainly by electrolysis of magnesium salts obtained from brine. It is less dense than aluminium and is used primarily as a component in strong and lightweight alloys that contain aluminium.

In crystallography, crystal structure is a description of the ordered arrangement of atoms, ions, or molecules in a crystalline material. Ordered structures occur from the intrinsic nature of the constituent particles to form symmetric patterns that repeat along the principal directions of three-dimensional space in matter.
Aluminium–silicon alloys or Silumin is a general name for a group of lightweight, high-strength aluminium alloys based on an aluminum–silicon system (AlSi) that consist predominantly of aluminum - with silicon as the quantitatively most important alloying element. Pure AlSi alloys cannot be hardened, the commonly used alloys AlSiCu and AlSiMg can be hardened. The hardening mechanism corresponds to that of AlCu and AlMgSi.

In crystallography, the cubiccrystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals.

An intermetallic is a type of metallic alloy that forms an ordered solid-state compound between two or more metallic elements. Intermetallics are generally hard and brittle, with good high-temperature mechanical properties. They can be classified as stoichiometric or nonstoichiometic intermetallic compounds.

Raney nickel, also called spongy nickel, is a fine-grained solid composed mostly of nickel derived from a nickel–aluminium alloy. Several grades are known, of which most are gray solids. Some are pyrophoric, but most are used as air-stable slurries. Raney nickel is used as a reagent and as a catalyst in organic chemistry. It was developed in 1926 by American engineer Murray Raney for the hydrogenation of vegetable oils. Raney is a registered trademark of W. R. Grace and Company. Other major producers are Evonik and Johnson Matthey.
A solid solution, a term popularly used for metals, is a homogeneous mixture of two different kinds of atoms in solid state and having a single crystal structure. Many examples can be found in metallurgy, geology, and solid-state chemistry. The word "solution" is used to describe the intimate mixing of components at the atomic level and distinguishes these homogeneous materials from physical mixtures of components. Two terms are mainly associated with solid solutions – solvents and solutes, depending on the relative abundance of the atomic species.

A lattice constant or lattice parameter is one of the physical dimensions and angles that determine the geometry of the unit cells in a crystal lattice, and is proportional to the distance between atoms in the crystal. A simple cubic crystal has only one lattice constant, the distance between atoms, but in general lattices in three dimensions have six lattice constants: the lengths a, b, and c of the three cell edges meeting at a vertex, and the angles α, β, and γ between those edges.
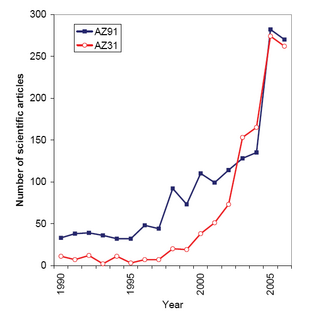
Magnesium alloys are mixtures of magnesium with other metals, often aluminium, zinc, manganese, silicon, copper, rare earths and zirconium. Magnesium alloys have a hexagonal lattice structure, which affects the fundamental properties of these alloys. Plastic deformation of the hexagonal lattice is more complicated than in cubic latticed metals like aluminium, copper and steel; therefore, magnesium alloys are typically used as cast alloys, but research of wrought alloys has been more extensive since 2003. Cast magnesium alloys are used for many components of modern automobiles and have been used in some high-performance vehicles; die-cast magnesium is also used for camera bodies and components in lenses.

Titanium hydride normally refers to the inorganic compound TiH2 and related nonstoichiometric materials. It is commercially available as a stable grey/black powder, which is used as an additive in the production of Alnico sintered magnets, in the sintering of powdered metals, the production of metal foam, the production of powdered titanium metal and in pyrotechnics.
Complex metallic alloys (CMAs) or complex intermetallics (CIMs) are intermetallic compounds characterized by the following structural features:
- large unit cells, comprising some tens up to thousands of atoms,
- the presence of well-defined atom clusters, frequently of icosahedral point group symmetry,
- the occurrence of inherent disorder in the ideal structure.
Nickel aluminide refers to either of two widely used intermetallic compounds, Ni3Al or NiAl, but the term is sometimes used to refer to any nickel–aluminium alloy. These alloys are widely used because of their high strength even at high temperature, low density, corrosion resistance, and ease of production. Ni3Al is of specific interest as a precipitate in nickel-based superalloys, where it is called the γ' (gamma prime) phase. It gives these alloys high strength and creep resistance up to 0.7–0.8 of its melting temperature. Meanwhile, NiAl displays excellent properties such as lower density and higher melting temperature than those of Ni3Al, and good thermal conductivity and oxidation resistance. These properties make it attractive for special high-temperature applications like coatings on blades in gas turbines and jet engines. However, both these alloys have the disadvantage of being quite brittle at room temperature, with Ni3Al remaining brittle at high temperatures as well. To address this problem, has been shown that Ni3Al can be made ductile when manufactured in single-crystal form rather than in polycrystalline form.

Wadsleyite is an orthorhombic mineral with the formula β-(Mg,Fe)2SiO4. It was first found in nature in the Peace River meteorite from Alberta, Canada. It is formed by a phase transformation from olivine (α-(Mg,Fe)2SiO4) under increasing pressure and eventually transforms into spinel-structured ringwoodite (γ-(Mg,Fe)2SiO4) as pressure increases further. The structure can take up a limited amount of other bivalent cations instead of magnesium, but contrary to the α and γ structures, a β structure with the sum formula Fe2SiO4 is not thermodynamically stable. Its cell parameters are approximately a = 5.7 Å, b = 11.71 Å and c = 8.24 Å.

Aluminium diboride (AlB2) is a chemical compound made from the metal aluminium and the metalloid boron. It is one of two compounds of aluminium and boron, the other being AlB12, which are both commonly referred to as aluminium boride.

Magnesium hydride is the chemical compound with the molecular formula MgH2. It contains 7.66% by weight of hydrogen and has been studied as a potential hydrogen storage medium.
Aluminium magnesium boride or Al3Mg3B56, colloquially known as BAM, is a chemical compound of aluminium, magnesium and boron. Whereas its nominal formula is AlMgB14, the chemical composition is closer to Al0.75Mg0.75B14. It is a ceramic alloy that is highly resistive to wear and has an extremely low coefficient of sliding friction, reaching a record value of 0.04 in unlubricated and 0.02 in lubricated AlMgB14−TiB2 composites. First reported in 1970, BAM has an orthorhombic structure with four icosahedral B12 units per unit cell. This ultrahard material has a coefficient of thermal expansion comparable to that of other widely used materials such as steel and concrete.
Chromium hydrides are compounds of chromium and hydrogen, and possibly other elements. Intermetallic compounds with not-quite-stoichometric quantities of hydrogen exist, as well as highly reactive molecules. When present at low concentrations, hydrogen and certain other elements alloyed with chromium act as softening agents that enables the movement of dislocations that otherwise not occur in the crystal lattices of chromium atoms.

Iron–hydrogen alloy, also known as iron hydride, is an alloy of iron and hydrogen and other elements. Because of its lability when removed from a hydrogen atmosphere, it has no uses as a structural material.
Iron aluminides are intermetallic compounds of iron and aluminium - they typically contain ~18% Al or more.
Aluminium–magnesium alloys (AlMg) – standardised in the 5000 series – are aluminium alloys that are mainly made of aluminium and contain magnesium as the main alloy element. Most standardised alloys also contain small additives of manganese (AlMg(Mn)). Pure AlMg alloys and the AlMg(Mn) alloys belong to the medium-strength, natural (not hardened by heat treatment) alloys. Other AlMg alloys are aluminium–magnesium–copper alloys (AlMgCu) and aluminium–magnesium–silicon alloys (AlMgSi, 6000 series).












