In chemistry, particularly in biochemistry, a fatty acid is a carboxylic acid with an aliphatic chain, which is either saturated or unsaturated. Most naturally occurring fatty acids have an unbranched chain of an even number of carbon atoms, from 4 to 28. Fatty acids are a major component of the lipids in some species such as microalgae but in some other organisms are not found in their standalone form, but instead exist as three main classes of esters: triglycerides, phospholipids, and cholesteryl esters. In any of these forms, fatty acids are both important dietary sources of fuel for animals and important structural components for cells.

In nutrition, biology, and chemistry, fat usually means any ester of fatty acids, or a mixture of such compounds, most commonly those that occur in living beings or in food.

A triglyceride is an ester derived from glycerol and three fatty acids. Triglycerides are the main constituents of body fat in humans and other vertebrates, as well as vegetable fat. They are also present in the blood to enable the bidirectional transference of adipose fat and blood glucose from the liver, and are a major component of human skin oils.
Essential fatty acids, or EFAs, are fatty acids that humans and other animals must ingest because the body requires them for good health, but cannot synthesize them.

Margarine is a spread used for flavoring, baking, and cooking. It is most often used as a substitute for butter. Although originally made from animal fats, most margarine consumed today is made from vegetable oil. The spread was originally named oleomargarine from Latin for oleum and Greek margarite. The name was later shortened to margarine.

Coconut oil is an edible oil derived from the kernels, meat, and milk of the coconut palm fruit. Coconut oil is a white solid fat below around 25 °C (77 °F), and a clear thin liquid oil in warmer climates. Unrefined varieties have a distinct coconut aroma. Coconut oil is used as a food oil, and in industrial applications for cosmetics and detergent production. The oil is rich in medium-chain fatty acids.
A saturated fat is a type of fat in which the fatty acid chains have all single bonds between the carbon atoms. A fat known as a glyceride is made of two kinds of smaller molecules: a short glycerol backbone and fatty acids that each contain a long linear or branched chain of carbon (C) atoms. Along the chain, some carbon atoms are linked by single bonds (-C-C-) and others are linked by double bonds (-C=C-). A double bond along the carbon chain can react with a pair of hydrogen atoms to change into a single -C-C- bond, with each H atom now bonded to one of the two C atoms. Glyceride fats without any carbon chain double bonds are called saturated because they are "saturated with" hydrogen atoms, having no double bonds available to react with more hydrogen.
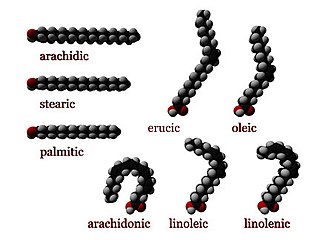
An unsaturated fat is a fat or fatty acid in which there is at least one double bond within the fatty acid chain. A fatty acid chain is monounsaturated if it contains one double bond, and polyunsaturated if it contains more than one double bond.

Stearic acid is a saturated fatty acid with an 18-carbon chain. The IUPAC name is octadecanoic acid. It is a soft waxy solid with the formula CH3(CH2)16CO2H. The triglyceride derived from three molecules of stearic acid is called stearin. Stearic acid is a prevalent fatty-acid in nature, found in many animal and vegetable fats, but is usually higher in animal fat than vegetable fat. It has a melting point of 69.4 °C (156.9 °F) °C and a pKa of 4.50.
In biochemistry and nutrition, a monounsaturated fat is a fat that contains a monounsaturated fatty acid (MUFA), a subclass of fatty acid characterized by having a double bond in the fatty acid chain with all of the remaining carbon atoms being single-bonded. By contrast, polyunsaturated fatty acids (PUFAs) have more than one double bond.

Saponification value or saponification number represents the number of milligrams of potassium hydroxide (KOH) or sodium hydroxide (NaOH) required to saponify one gram of fat under the conditions specified. It is a measure of the average molecular weight of all the fatty acids present in the sample in form of triglycerides. The higher the saponification value, the lower the fatty acids average length, the lighter the mean molecular weight of triglycerides and vice versa. Practically, fats or oils with high saponification value are more suitable for soap making.
Lauric acid, systematically dodecanoic acid, is a saturated fatty acid with a 12-carbon atom chain, thus having many properties of medium-chain fatty acids. It is a bright white, powdery solid with a faint odor of bay oil or soap. The salts and esters of lauric acid are known as laurates.
Palm kernel oil is an edible plant oil derived from the kernel of the oil palm tree Elaeis guineensis. It is related to two other edible oils: palm oil, extracted from the fruit pulp of the oil palm, and coconut oil, extracted from the kernel of the coconut.

Retinyl palmitate, or vitamin A palmitate, is the ester of retinol (vitamin A) and palmitic acid, with formula C36H60O2. It is the most abundant form of vitamin A storage in animals.

In biochemistry and nutrition, a polyunsaturated fat is a fat that contains a polyunsaturated fatty acid, which is a subclass of fatty acid characterized by a backbone with two or more carbon–carbon double bonds. Some polyunsaturated fatty acids are essentials. Polyunsaturated fatty acids are precursors to and are derived from polyunsaturated fats, which include drying oils.

A medium-chain triglyceride (MCT) is a triglyceride with two or three fatty acids having an aliphatic tail of 6–12 carbon atoms, i.e. a medium-chain fatty acid (MCFA). Rich food sources for commercial extraction of MCTs include palm kernel oil and coconut oil.
Oleochemistry is the study of vegetable oils and animal oils and fats, and oleochemicals derived from these fats and oils. The resulting product can be called oleochemicals (from Latin: oleum "olive oil"). The major product of this industry is soap, approximately 8.9×106 tons of which were produced in 1990. Other major oleochemicals include fatty acids, fatty acid methyl esters, fatty alcohols and fatty amines. Glycerol is a side product of all of these processes. Intermediate chemical substances produced from these basic oleochemical substances include alcohol ethoxylates, alcohol sulfates, alcohol ether sulfates, quaternary ammonium salts, monoacylglycerols (MAG), diacylglycerols (DAG), structured triacylglycerols (TAG), sugar esters, and other oleochemical products.
SN2 Palmitate is a structured triglyceride where palmitic acid is bonded to the middle position (sn-2) of the glycerol backbone. Structured triglycerides are achieved through an enzymatic process using vegetable oils. Current usage of structured triglycerides is mainly for infant formula providing a human milk fat substitute.

Cooking oil is a plant or animal liquid fat used in frying, baking, and other types of cooking. Oil allows higher cooking temperatures than water, making cooking faster and more flavorful, while likewise distributing heat, reducing burning and uneven cooking. It sometimes imparts its own flavor. Cooking oil is also used in food preparation and flavoring not involving heat, such as salad dressings and bread dips.

Trans fat, also called trans-unsaturated fatty acids, or trans fatty acids, is a type of unsaturated fat that occurs in foods. Trace concentrations of trans fats occur naturally, but large amounts are found in some processed foods. Since consumption of trans fats is unhealthy, artificial trans fats are highly regulated or banned in many nations. However, they are still widely consumed in developing nations, resulting in hundreds of thousands of deaths each year. The World Health Organization (WHO) had set a goal to make the world free from industrially produced trans fat by the end of 2023. The goal was not met, and the WHO announced another goal "for accelerated action till 2025 to complete this effort" along with associated support on 1 February 2024.


 [2]
[2] 











