
Lyme disease, also known as Lyme borreliosis, is a tick-borne disease caused by species of Borrelia bacteria, transmitted by blood-feeding ticks in the genus Ixodes. The most common sign of infection is an expanding red rash, known as erythema migrans (EM), which appears at the site of the tick bite about a week afterwards. The rash is typically neither itchy nor painful. Approximately 70–80% of infected people develop a rash. Early diagnosis can be difficult. Other early symptoms may include fever, headaches and tiredness. If untreated, symptoms may include loss of the ability to move one or both sides of the face, joint pains, severe headaches with neck stiffness or heart palpitations. Months to years later, repeated episodes of joint pain and swelling may occur. Occasionally, shooting pains or tingling in the arms and legs may develop. Despite appropriate treatment, about 10 to 20% of those affected develop joint pains, memory problems, and tiredness for six months or more; this constellation of symptoms is termed post-treatment Lyme disease syndrome (PTLDS).
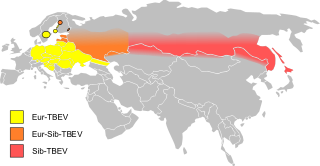
Tick-borne encephalitis (TBE) is a viral infectious disease involving the central nervous system. The disease most often manifests as meningitis, encephalitis or meningoencephalitis. Myelitis and spinal paralysis also occurs. In about one third of cases sequelae, predominantly cognitive dysfunction, persist for a year or more.

Borrelia burgdorferi is a bacterial species of the spirochete class in the genus Borrelia, and is one of the causative agents of Lyme disease in humans. Along with a few similar genospecies, some of which also cause Lyme disease, it makes up the species complex of Borrelia burgdorferi sensu lato. The complex currently comprises 20 accepted and 3 proposed genospecies. B. burgdorferi sensu stricto exists in North America and Eurasia and until 2016 was the only known cause of Lyme disease in North America. Borrelia species are Gram-negative.

Dermacentor variabilis, also known as the American dog tick or wood tick, is a species of tick that is known to carry bacteria responsible for several diseases in humans, including Rocky Mountain spotted fever and tularemia. It is one of the best-known hard ticks. Diseases are spread when it sucks blood from the host. It may take several days for the host to experience symptoms.
Relapsing fever is a vector-borne disease caused by infection with certain bacteria in the genus Borrelia, which is transmitted through the bites of lice, soft-bodied ticks, or hard-bodied ticks.
Powassan virus (POWV) is a Flavivirus transmitted by ticks, found in North America and in the Russian Far East. It is named after the town of Powassan, Ontario, where it was identified in a young boy who eventually died from it. It can cause encephalitis, inflammation of the brain. No approved vaccine or antiviral drug exists. Prevention of tick bites is the best precaution.
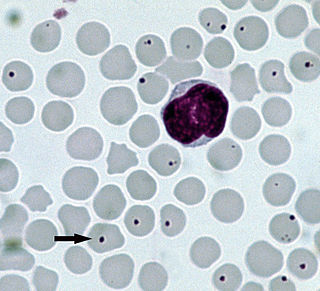
Anaplasmosis is a tick-borne disease affecting ruminants, dogs, and horses, and is caused by Anaplasma bacteria. Anaplasmosis is an infectious but not contagious disease. Anaplasmosis can be transmitted through mechanical and biological vector processes. Anaplasmosis can also be referred to as "yellow bag" or "yellow fever" because the infected animal can develop a jaundiced look. Other signs of infection include weight loss, diarrhea, paleness of the skin, aggressive behavior, and high fever.
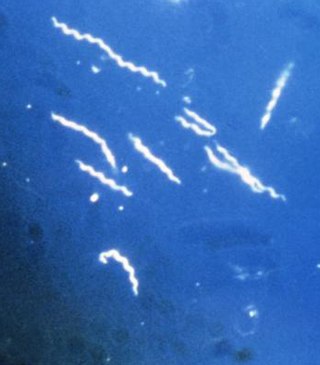
Lyme disease, or borreliosis, is caused by spirochetal bacteria from the genus Borrelia, which has 52 known species. Three main species are the main causative agents of the disease in humans, while a number of others have been implicated as possibly pathogenic. Borrelia species in the species complex known to cause Lyme disease are collectively called Borrelia burgdorferisensu lato (s.l.) not to be confused with the single species in that complex Borrelia burgdorferi sensu stricto which is responsible for nearly all cases of Lyme disease in North America.

Ixodes scapularis is commonly known as the deer tick or black-legged tick, and in some parts of the US as the bear tick. It was also named Ixodes dammini until it was shown to be the same species in 1993. It is a hard-bodied tick found in the eastern and northern Midwest of the United States as well as in southeastern Canada. It is a vector for several diseases of animals, including humans and is known as the deer tick owing to its habit of parasitizing the white-tailed deer. It is also known to parasitize mice, lizards, migratory birds, etc. especially while the tick is in the larval or nymphal stage.

Ixodes ricinus, the castor bean tick, is a chiefly European species of hard-bodied tick. It may reach a length of 11 mm (0.43 in) when engorged with a blood meal, and can transmit both bacterial and viral pathogens such as the causative agents of Lyme disease and tick-borne encephalitis.

Amblyomma americanum, also known as the lone star tick, the northeastern water tick, or the turkey tick, is a type of tick indigenous to much of the eastern United States and Mexico, that bites painlessly and commonly goes unnoticed, remaining attached to its host for as long as seven days until it is fully engorged with blood. It is a member of the phylum Arthropoda, class Arachnida. The adult lone star tick is sexually dimorphic, named for a silvery-white, star-shaped spot or "lone star" present near the center of the posterior portion of the adult female shield (scutum); adult males conversely have varied white streaks or spots around the margins of their shields.
Powassan encephalitis, caused by the Powassan virus (POWV), a flavivirus also known as the deer tick virus, is a form of arbovirus infection that results from tick bites. It can occur as a co-infection with Lyme disease, as both are transmitted to humans by the same species of tick. Over the last decade, there has been a surge in the number of cases and an expansion of its geographic range. In the United States, cases have been documented primarily in the northeast. The disease was first isolated from the brain of a boy who died of encephalitis in Powassan, Ontario, in 1958. This disease is classified as a zoonosis, originating in animals, often found in rodents and ticks, with subsequent transmission to humans. The virus shares antigenic similarities with the Far Eastern tick-borne encephalitis viruses.

Human granulocytic anaplasmosis (HGA) is a tick-borne, infectious disease caused by Anaplasma phagocytophilum, an obligate intracellular bacterium that is typically transmitted to humans by ticks of the Ixodes ricinus species complex, including Ixodes scapularis and Ixodes pacificus in North America. These ticks also transmit Lyme disease and other tick-borne diseases.
Ticks are insects known for attaching to and sucking blood from land-dwelling animals. Ticks fall under the category of 'arthropod', and while they are often thought of in the context of disease transmission, they are also known to cause direct harm to hosts through bites, toxin release, and infestation. Infestation can cause symptoms ranging from mild to severe and may even cause death. Hosts can include any number of vertebrates, though humans and livestock are more likely to be the interest of researchers.

Deer tick virus (DTV) is a virus in the genus Flavivirus spread via ticks that causes encephalitis.
Rickettsia australis is a bacterium that causes a medical condition called Queensland tick typhus. The probable vectors are the tick species, Ixodes holocyclus and Ixodes tasmani. Small marsupials are suspected reservoirs of this bacterium.
Borrelia miyamotoi is a bacterium of the spirochete phylum in the genus Borrelia. A zoonotic organism, B. miyamotoi can infect humans through the bite of several species of hard-shell Ixodes ticks, the same kind of ticks that spread B. burgdorferi, the causative bacterium of Lyme disease. Ixodes ticks are also the primary vector in the spread of babesiosis and anaplasmosis.
Tibovirus is a term often used to describe viruses that are transmitted by tick vectors. The word tibovirus is an acronym. This falls within the superorder arthropod thus tibovirus is classified under Arthropod Borne virus (Arborvirus). For a person to acquire infection the tick must bite and feed for a sufficient period of time. The tiboviruses that affect humans are limited to within 3 families: Flaviviridae, Reoviridae, and Bunyaviridae.
Borrelia mayonii is a Gram-negative, host-associated spirochete that is capable of causing Lyme disease. This organism can infect various vertebrate hosts such as humans via the bite of a black legged tick.

Global climate change has increased the occurrence of some infectious diseases. Infectious diseases whose transmission is impacted by climate change include, for example, vector-borne diseases like dengue fever, malaria, tick-borne diseases, leishmaniasis, zika fever, chikungunya and Ebola. One mechanism contributing to increased disease transmission is that climate change is altering the geographic range and seasonality of the insects that can carry the diseases. Scientists stated a clear observation in 2022: "the occurrence of climate-related food-borne and waterborne diseases has increased ."











