Related Research Articles

A lithium-ion or Li-ion battery is a type of rechargeable battery that uses the reversible intercalation of Li+ ions into electronically conducting solids to store energy. In comparison with other commercial rechargeable batteries, Li-ion batteries are characterized by higher specific energy, higher energy density, higher energy efficiency, a longer cycle life, and a longer calendar life. Also noteworthy is a dramatic improvement in lithium-ion battery properties after their market introduction in 1991: within the next 30 years, their volumetric energy density increased threefold while their cost dropped tenfold.
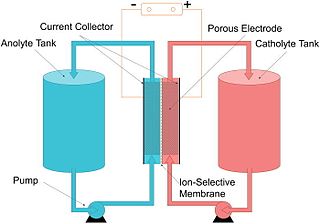
The vanadium redox battery (VRB), also known as the vanadium flow battery (VFB) or vanadium redox flow battery (VRFB), is a type of rechargeable flow battery. It employs vanadium ions as charge carriers. The battery uses vanadium's ability to exist in a solution in four different oxidation states to make a battery with a single electroactive element instead of two. For several reasons, including their relative bulkiness, vanadium batteries are typically used for grid energy storage, i.e., attached to power plants/electrical grids.

A flow battery, or redox flow battery, is a type of electrochemical cell where chemical energy is provided by two chemical components dissolved in liquids that are pumped through the system on separate sides of a membrane. Ion transfer inside the cell occurs through the membrane while both liquids circulate in their own respective space. Cell voltage is chemically determined by the Nernst equation and ranges, in practical applications, from 1.0 to 2.43 volts. The energy capacity is a function of the electrolyte volume and the power is a function of the surface area of the electrodes.

Lithium iron phosphate or lithium ferro-phosphate (LFP) is an inorganic compound with the formula LiFePO
4. It is a gray, red-grey, brown or black solid that is insoluble in water. The material has attracted attention as a component of lithium iron phosphate batteries, a type of Li-ion battery. This battery chemistry is targeted for use in power tools, electric vehicles, solar energy installations and more recently large grid-scale energy storage.
The lithium–air battery (Li–air) is a metal–air electrochemical cell or battery chemistry that uses oxidation of lithium at the anode and reduction of oxygen at the cathode to induce a current flow.
A metal–air electrochemical cell is an electrochemical cell that uses an anode made from pure metal and an external cathode of ambient air, typically with an aqueous or aprotic electrolyte.
A lithium ion manganese oxide battery (LMO) is a lithium-ion cell that uses manganese dioxide, MnO
2, as the cathode material. They function through the same intercalation/de-intercalation mechanism as other commercialized secondary battery technologies, such as LiCoO
2. Cathodes based on manganese-oxide components are earth-abundant, inexpensive, non-toxic, and provide better thermal stability.
Lithium–silicon batteries are lithium-ion battery that employ a silicon-based anode and lithium ions as the charge carriers. Silicon based materials generally have a much larger specific capacity, for example 3600 mAh/g for pristine silicon, relative to the standard anode material graphite, which is limited to a maximum theoretical capacity of 372 mAh/g for the fully lithiated state LiC6.

Kuzhikalail M. Abraham is an American scientist, a recognized expert on lithium-ion and lithium-ion polymer batteries and is the inventor of the ultrahigh energy density lithium–air battery. Abraham is the principal of E-KEM Sciences in Needham, Massachusetts and a professor at the Northeastern University Center for Renewable Energy Technologies, Northeastern University, in Boston, Massachusetts.
Sakti3, a wholly owned subsidiary of Dyson Ltd., is a solid-state battery company based in Ann Arbor, Michigan. Dyson once stated that it was “committed to investing £1B in battery technology over the coming years, and Sakti3 is an essential and exciting part of that program.” However, Dyson later wrote off much of its investment in Sakti3, indicating that it could no longer be valued at the $93 million price that Dyson had paid. Dyson eventually also cancelled its plans to develop an electric vehicle.
Yang Shao-Horn is a Chinese American scholar, Professor of Mechanical Engineering and Materials Science and Engineering and a member of Research Laboratory of Electronics at the Massachusetts Institute of Technology. She is known for research on understanding and controlling of processes for storing electrons in chemical bonds towards zero-carbon energy and chemicals.

A solid dispersion redox flow battery is a type of redox flow battery using dispersed solid active materials as the energy storage media. The solid suspensions are stored in energy storage tanks and pumped through electrochemical cells while charging or discharging. In comparison with a conventional redox flow battery where active species are dissolved in aqueous or organic electrolyte, the active materials in a solid dispersion redox flow battery maintain the solid form and are suspended in the electrolyte. Further development expanded the applicable active materials. The solid active materials, especially with active materials from lithium-ion battery, can help the suspensions achieve much higher energy densities than conventional redox flow batteries. This concept is similar to semi-solid flow batteries in which slurries of active materials accompanied by conductive carbon additives to facilitate electrons conducting are stored in energy storage tanks and pumped through the electrochemical reaction cells. Based upon this technique, an analytical method was developed to measure the electrochemical performance of lithium-ion battery active materials, named dispersed particle resistance (DPR).
Dispersed particle resistance (DPR) is a measured parameter to characterize battery active materials. It is seen as an indicator of lithium-ion battery active material rate capability. It is the slope of voltage-current linear fit for active material samples in suspensions. It can be obtained by applying different voltages on a suspension and measuring the currents, after which the data points are plotted. The slope of the plot is referred to as dispersed particle resistance. It can also be done in the opposite way where different currents are applied and voltages are measured. The key advantage of this dispersed particle resistance technique is fast and accurate comparing with the conventional characterization method for which batteries need to be fabricated and tested for a long time.

Doron Aurbach is an Israeli electrochemist, materials and surface scientist.
Scanning vibrating electrode technique (SVET), also known as vibrating probe within the field of biology, is a scanning probe microscopy (SPM) technique which visualizes electrochemical processes at a sample. It was originally introduced in 1974 by Jaffe and Nuccitelli to investigate the electrical current densities near living cells. Starting in the 1980s Hugh Isaacs began to apply SVET to a number of different corrosion studies. SVET measures local current density distributions in the solution above the sample of interest, to map electrochemical processes in situ as they occur. It utilizes a probe, vibrating perpendicular to the sample of interest, to enhance the measured signal. It is related to scanning ion-selective electrode technique (SIET), which can be used with SVET in corrosion studies, and scanning reference electrode technique (SRET), which is a precursor to SVET.
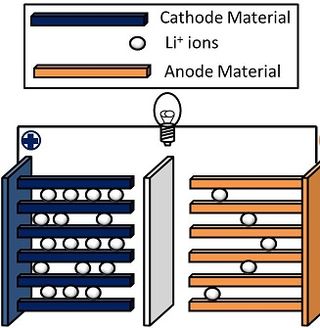
Lithium nickel manganese cobalt oxides (abbreviated NMC, Li-NMC, LNMC, or NCM) are mixed metal oxides of lithium, nickel, manganese and cobalt with the general formula LiNixMnyCo1-x-yO2. These materials are commonly used in lithium-ion batteries for mobile devices and electric vehicles, acting as the positively charged cathode.

Electrochemical quartz crystal microbalance (EQCM) is the combination of electrochemistry and quartz crystal microbalance, which was generated in the eighties. Typically, an EQCM device contains an electrochemical cells part and a QCM part. Two electrodes on both sides of the quartz crystal serve two purposes. Firstly, an alternating electric field is generated between the two electrodes for making up the oscillator. Secondly, the electrode contacting electrolyte is used as a working electrode (WE), together with a counter electrode (CE) and a reference electrode (RE), in the potentiostatic circuit constituting the electrochemistry cell. Thus, the working electrode of electrochemistry cell is the sensor of QCM.

This is a history of the lithium-ion battery.
Fluoride-ion batteries are rechargeable battery technology based on the shuttle of fluoride ions as ionic charge carriers.
The Iron Redox Flow Battery (IRFB), also known as Iron Salt Battery (ISB), stores and releases energy through the electrochemical reaction of iron salt. This type of battery belongs to the class of redox-flow batteries (RFB), which are alternative solutions to Lithium-Ion Batteries (LIB) for stationary applications. The IRFB can achieve up to 70% round trip energy efficiency. In comparison, other long duration storage technologies such as pumped hydro energy storage provide around 80% round trip energy efficiency.
References
- ↑ LeVine, Steve (December 15, 2017). "Battery exec leaves Dyson two years after $90 million buyout". Axios.com .
- 1 2 3 4 5 Desmond, Kevin (2016). Innovators in battery technology : profiles of 93 influential electrochemists. McFarland & Company. ISBN 9780786499335.[ permanent dead link ]
- ↑ Berhan, L.; Sastry, A. M. (April 30, 2007). "Modeling percolation in high-aspect-ratio fiber systems. I. Soft-core versus hard-core models". Physical Review E. 75 (4): 041120. Bibcode:2007PhRvE..75d1120B. doi:10.1103/PhysRevE.75.041120. PMID 17500878. S2CID 6380421.
- ↑ Berhan, L.; Sastry, A. M. (April 30, 2007). "Modeling percolation in high-aspect-ratio fiber systems. II. The effect of waviness on the percolation onset". Physical Review E. 75 (4): 041121. Bibcode:2007PhRvE..75d1121B. doi:10.1103/PhysRevE.75.041121. PMID 17500879. S2CID 13999078.
- ↑ Yi, Y.-B.; Sastry, A. M. (August 8, 2004). "Analytical approximation of the percolation threshold for overlapping ellipsoids of revolution". Proceedings of the Royal Society of London A: Mathematical, Physical and Engineering Sciences. 460 (2048): 2353–2380. Bibcode:2004RSPSA.460.2353Y. doi:10.1098/rspa.2004.1279. ISSN 1364-5021. S2CID 2475482.
- ↑ Vincent, Andrea M.; Perrone, Lorena; Sullivan, Kelli A.; Backus, Carey; Sastry, Ann Marie; Lastoskie, Christian; Feldman, Eva L. (February 1, 2007). "Receptor for Advanced Glycation End Products Activation Injures Primary Sensory Neurons via Oxidative Stress". Endocrinology. 148 (2): 548–558. doi: 10.1210/en.2006-0073 . ISSN 0013-7227. PMID 17095586.
- ↑ Zhang, Xiangchun; Shyy, Wei; Sastry, Ann Marie (October 1, 2007). "Numerical Simulation of Intercalation-Induced Stress in Li-Ion Battery Electrode Particles". Journal of the Electrochemical Society. 154 (10): A910–A916. Bibcode:2007JElS..154A.910Z. doi:10.1149/1.2759840. ISSN 0013-4651. S2CID 15540771.
- ↑ Wang, Chia-Wei; Sastry, Ann Marie (November 1, 2007). "Mesoscale Modeling of a Li-Ion Polymer Cell". Journal of the Electrochemical Society. 154 (11): A1035–A1047. Bibcode:2007JElS..154A1035W. doi:10.1149/1.2778285. ISSN 0013-4651. S2CID 10130595.
- ↑ Kim, Hyon C; Sastry, Ann M (November 1, 2012). "Effects of carbon fiber electrode deformation in multifunctional structural lithium ion batteries". Journal of Intelligent Material Systems and Structures. 23 (16): 1787–1797. doi:10.1177/1045389X12449914. ISSN 1045-389X. S2CID 108505799.
- ↑ Chung, M. D.; Seo, J. H.; Zhang, X. C.; Sastry, A. M. (April 1, 2011). "Implementing Realistic Geometry and Measured Diffusion Coefficients into Single Particle Electrode Modeling Based on Experiments with Single LiMn2O4 Spinel Particles". Journal of the Electrochemical Society. 158 (4): A371–A378. CiteSeerX 10.1.1.686.6838 . doi:10.1149/1.3549161. ISSN 0013-4651.
- ↑ Chen, Y. -H.; Wang, C. -W.; Zhang, X.; Sastry, A. M. (May 1, 2010). "Porous cathode optimization for lithium cells: Ionic and electronic conductivity, capacity, and selection of materials". Journal of Power Sources. 195 (9): 2851–2862. Bibcode:2010JPS...195.2851C. doi:10.1016/j.jpowsour.2009.11.044.
- ↑ "Ann Marie Sastry - Google Scholar Citations". scholar.google.com. Retrieved August 3, 2017.
- ↑ "Sakti3 Raises $7 Million in Series B Financing". www.businesswire.com. April 1, 2010. Retrieved November 27, 2020.
- ↑ LeVine, Steve (October 21, 2015). "Sakti3's quest for a better battery: Hype, funding, promises, and then a surprise sale". Quartz. Retrieved November 27, 2020.
- ↑ Charlton, Alistair (April 3, 2017). "Out with the old, in with the new: Dyson drops old battery patents in solid state cell push". International Business Times UK. Retrieved August 3, 2017.
- ↑ "Dyson acquires Sakti3 for $90M to help commercialize 'breakthrough' solid-state battery tech". VentureBeat. October 19, 2015. Retrieved November 27, 2020.
- ↑ LeVine, Steve (December 15, 2017). "Battery exec leaves Dyson two years after $90 million buyout". Axios. Retrieved November 27, 2020.
- ↑ "Dyson has scrapped its electric car project". BBC News. October 11, 2019. Retrieved November 27, 2020.
- 1 2 3 LeVine, Steve (October 21, 2015). "Sakti3's quest for a better battery: Hype, funding, promises, and then a surprise sale". Quartz. Retrieved June 24, 2021.
- ↑ Walker, L.; Robertson, D.; Basco, J.; Prezas, P.; Bloom, I. "Electrochemical Performance Testing" (PDF). Argonne National Laboratory. Retrieved July 20, 2021.
- ↑ "The Presidential Early Career Award for Scientists and Engineers: Recipient Search Results | NSF – National Science Foundation". www.nsf.gov. Retrieved August 3, 2017.
- ↑ "Frank Kreith Award of the ASME" . Retrieved August 3, 2017.
- ↑ "ASME Fellows" (PDF). Retrieved August 3, 2017.