Related Research Articles
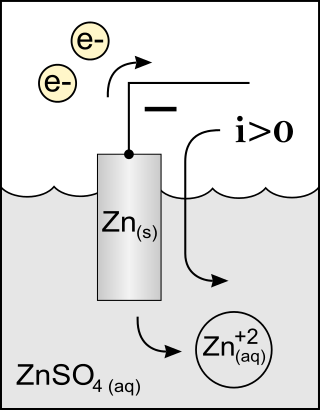
An anode is an electrode of a polarized electrical device through which conventional current enters the device. This contrasts with a cathode, an electrode of the device through which conventional current leaves the device. A common mnemonic is ACID, for "anode current into device". The direction of conventional current in a circuit is opposite to the direction of electron flow, so electrons flow out the anode of a galvanic cell, into an outside or external circuit connected to the cell. For example, the end of a household battery marked with a "-" (minus) is the anode.
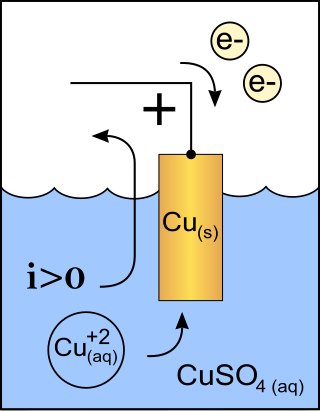
A cathode is the electrode from which a conventional current leaves a polarized electrical device. This definition can be recalled by using the mnemonic CCD for Cathode Current Departs. A conventional current describes the direction in which positive charges move. Electrons have a negative electrical charge, so the movement of electrons is opposite to that of the conventional current flow. Consequently, the mnemonic cathode current departs also means that electrons flow into the device's cathode from the external circuit. For example, the end of a household battery marked with a + (plus) is the cathode.
Electrochemistry is the branch of physical chemistry concerned with the relationship between electrical potential difference, as a measurable and quantitative phenomenon, and identifiable chemical change, with the potential difference as an outcome of a particular chemical change, or vice versa. These reactions involve electrons moving via an electronically-conducting phase between electrodes separated by an ionically conducting and electronically insulating electrolyte.

Rust is an iron oxide, a usually reddish-brown oxide formed by the reaction of iron and oxygen in the catalytic presence of water or air moisture. Rust consists of hydrous iron(III) oxides (Fe2O3·nH2O) and iron(III) oxide-hydroxide (FeO(OH), Fe(OH)3), and is typically associated with the corrosion of refined iron.
In electrochemistry, electrode potential is the electromotive force of a galvanic cell built from a standard reference electrode and another electrode to be characterized. By convention, the reference electrode is the standard hydrogen electrode (SHE). It is defined to have a potential of zero volts. It may also be defined as the potential difference between the charged metallic rods and salt solution.

Electroplating, also known as electrochemical deposition or electrodeposition, is a process for producing a metal coating on a solid substrate through the reduction of cations of that metal by means of a direct electric current. The part to be coated acts as the cathode of an electrolytic cell; the electrolyte is a solution of a salt of the metal to be coated; and the anode is usually either a block of that metal, or of some inert conductive material. The current is provided by an external power supply.

Corrosion is a natural process that converts a refined metal into a more chemically stable oxide. It is the gradual deterioration of materials by chemical or electrochemical reaction with their environment. Corrosion engineering is the field dedicated to controlling and preventing corrosion.

A galvanic anode, or sacrificial anode, is the main component of a galvanic cathodic protection system used to protect buried or submerged metal structures from corrosion.
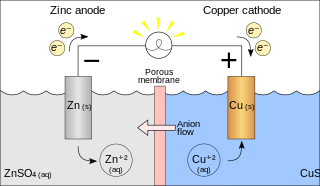
A galvanic cell or voltaic cell, named after the scientists Luigi Galvani and Alessandro Volta, respectively, is an electrochemical cell in which an electric current is generated from spontaneous Oxidation-Reduction reactions. A common apparatus generally consists of two different metals, each immersed in separate beakers containing their respective metal ions in solution that are connected by a salt bridge or separated by a porous membrane.

Cathodic protection is a technique used to control the corrosion of a metal surface by making it the cathode of an electrochemical cell. A simple method of protection connects the metal to be protected to a more easily corroded "sacrificial metal" to act as the anode. The sacrificial metal then corrodes instead of the protected metal. For structures such as long pipelines, where passive galvanic cathodic protection is not adequate, an external DC electrical power source is used to provide sufficient current.

A mercury-arc valve or mercury-vapor rectifier or (UK) mercury-arc rectifier is a type of electrical rectifier used for converting high-voltage or high-current alternating current (AC) into direct current (DC). It is a type of cold cathode gas-filled tube, but is unusual in that the cathode, instead of being solid, is made from a pool of liquid mercury and is therefore self-restoring. As a result mercury-arc valves, when used as intended, are far more robust and durable and can carry much higher currents than most other types of gas discharge tube. Some examples have been in continuous service, rectifying 50 ampere currents for decades.
A silver chloride electrode is a type of reference electrode, commonly used in electrochemical measurements. For environmental reasons it has widely replaced the saturated calomel electrode. For example, it is usually the internal reference electrode in pH meters and it is often used as reference in reduction potential measurements. As an example of the latter, the silver chloride electrode is the most commonly used reference electrode for testing cathodic protection corrosion control systems in sea water environments.
The galvanic series determines the nobility of metals and semi-metals. When two metals are submerged in an electrolyte, while also electrically connected by some external conductor, the less noble (base) will experience galvanic corrosion. The rate of corrosion is determined by the electrolyte, the difference in nobility, and the relative areas of the anode and cathode exposed to the electrolyte. The difference can be measured as a difference in voltage potential: the less noble metal is the one with a lower electrode potential than the nobler one, and will function as the anode within the electrolyte device functioning as described above. Galvanic reaction is the principle upon which batteries are based.
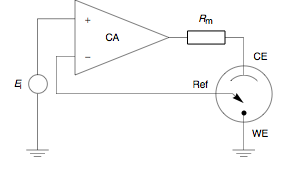
A potentiostat is the electronic hardware required to control a three electrode cell and run most electroanalytical experiments. A Bipotentiostat and polypotentiostat are potentiostats capable of controlling two working electrodes and more than two working electrodes, respectively.

Voltammetry is a category of electroanalytical methods used in analytical chemistry and various industrial processes. In voltammetry, information about an analyte is obtained by measuring the current as the potential is varied. The analytical data for a voltammetric experiment comes in the form of a voltammogram which plots the current produced by the analyte versus the potential of the working electrode.
In battery technology, a concentration cell is a limited form of a galvanic cell that has two equivalent half-cells of the same composition differing only in concentrations. One can calculate the potential developed by such a cell using the Nernst equation. A concentration cell produces a small voltage as it attempts to reach chemical equilibrium, which occurs when the concentration of reactant in both half-cells are equal. Because an order of magnitude concentration difference produces less than 60 millivolts at room temperature, concentration cells are not typically used for energy storage.
Electrogalvanizing is a process in which a layer of zinc is bonded to steel in order to protect against corrosion. The process involves electroplating, running a current of electricity through a saline/zinc solution with a zinc anode and steel conductor. Such Zinc electroplating or Zinc alloy electroplating maintains a dominant position among other electroplating process options, based upon electroplated tonnage per annum. According to the International Zinc Association, more than 5 million tons are used yearly for both hot dip galvanizing and electroplating. The plating of zinc was developed at the beginning of the 20th century. At that time, the electrolyte was cyanide based. A significant innovation occurred in the 1960s, with the introduction of the first acid chloride based electrolyte. The 1980s saw a return to alkaline electrolytes, only this time, without the use of cyanide. The most commonly used electrogalvanized cold rolled steel is SECC, acronym of "Steel, Electrogalvanized, Cold-rolled, Commercial quality". Compared to hot dip galvanizing, electroplated zinc offers these significant advantages:
Corrosion engineering is an engineering specialty that applies scientific, technical, engineering skills, and knowledge of natural laws and physical resources to design and implement materials, structures, devices, systems, and procedures to manage corrosion. From a holistic perspective, corrosion is the phenomenon of metals returning to the state they are found in nature. The driving force that causes metals to corrode is a consequence of their temporary existence in metallic form. To produce metals starting from naturally occurring minerals and ores, it is necessary to provide a certain amount of energy, e.g. Iron ore in a blast furnace. It is therefore thermodynamically inevitable that these metals when exposed to various environments would revert to their state found in nature. Corrosion and corrosion engineering thus involves a study of chemical kinetics, thermodynamics, electrochemistry and materials science.

Bipolar electrochemistry is a phenomenon in electrochemistry based on the polarization of conducting objects in electric fields. Indeed, this polarization generates a potential difference between the two extremities of the substrate that is equal to the electric field value multiplied by the size of the object. If this potential difference is important enough, then redox reactions can be generated at the extremities of the object, oxidations will occur at one extremity coupled simultaneously to reductions at the other extremity. In a simple experimental setup consisting of a platinum wire in a weighing boat containing a pH indicator solution, a 30 V voltage across two electrodes will cause water reduction at one end of the wire and a pH increase and water oxidation at the anodic end and a pH decrease. The poles of the bipolar electrode also align themselves with the applied electric field.

Galvanic corrosion is an electrochemical process in which one metal corrodes preferentially when it is in electrical contact with another, in the presence of an electrolyte. A similar galvanic reaction is exploited in primary cells to generate a useful electrical voltage to power portable devices. This phenomenon is named after Italian physician Luigi Galvani (1737-1798).
References
- ↑ Néstor Pérez " Electrochemistry and corrosion science", Kluwer Academic Publishers, 2004 (google books).
- ↑ Anodic protection : theory and practice in the prevention of corrosion, by Olen L. Riggs, Jr. and Carl E. Locke, Plenum Press, New York, 1981
- ↑ CORRWIKI Archived 5 December 2010 at the Wayback Machine , retrieved 8 September 2010