
In chemistry, a carbide is a compound composed of carbon and a less electronegative element. Carbides can be generally classified by the chemical bonds type as follows: (i) salt-like, (ii) covalent compounds, (iii) interstitial compounds, and (iv) "intermediate" transition metal carbides. Examples include calcium carbide (CaC2), silicon carbide (SiC), tungsten carbide (WC; often called, simply, carbide when referring to machine tooling), and cementite (Fe3C), each used in key industrial applications. The naming of ionic carbides is not systematic.
A coordinate covalent bond, also known as a dative bond or coordinate bond is a kind of 2-center, 2-electron covalent bond in which the two electrons derive from the same atom. The bonding of metal ions to ligands involves this kind of interaction. This type of interaction is central to Lewis acid-base theory.

Boranes is the name given to the class of synthetic hydrides of boron with generic formula BxHy. In the past, borane molecules were often labeled " electron-deficient" because of their multicenter bonding (in which a pair of bonding electrons links more than two atoms, as in 3-center-2-electron bonds); this was done in order to distinguish such molecules from hydrocarbons and other classically bonded compounds. However, this usage is incorrect, as most boranes and related clusters such as carboranes are actually electron-precise, not electron-deficient. For example, the extremely stable icosahedral B12H122- dianion, whose 26 cluster valence electrons exactly fill the 13 bonding molecular orbitals, is in no actual sense deficient in electrons; indeed it is thermodynamically far more stable than benzene.

Diborane(6), generally known as diborane is the chemical compound consisting of boron and hydrogen with the formula B2H6. It is a colorless, pyrophoric gas with a repulsively sweet odor. Synonyms include boroethane, boron hydride, and diboron hexahydride. Diborane is a key boron compound with a variety of applications. It has attracted wide attention for its electronic structure. Its derivatives are useful reagents.
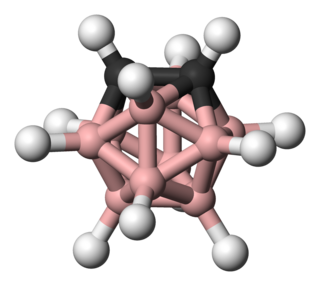
Carboranes are electron-delocalized clusters composed of boron, carbon and hydrogen atoms that may also contain other nonmetallic and nonmetallic elements in the cluster framework. Like many of the related boron hydrides, these clusters are polyhedra or fragments of polyhedra, and are similarly classified as closo-, nido-, arachno-, hypho-, etc. based on whether they represent a complete (closo-) polyhedron, or a polyhedron that is missing one (nido-), two (arachno-), three (hypho-), or more vertices. Carboranes are a notable example of heteroboranes.

A borabenzene is a heteroaromatic compound that has a boron atom instead of the carbon atom of a benzene molecule. A free borabenzene, which has no donor ligand on the boron atom, has not yet been isolated despite its simple structure and the chemical robustness of boron-carbon bonds. The instability of this molecule results from the high Lewis acidity of the boron atom due to its electron deficiency. All attempts to create a free borabenzene have resulted in formations of complexes with anionic or neutral ligands. However, the complexes with anionic ligands, called boratabenzenes, show rich chemistry including the coordination chemistry as anionic π-type ligands. Borabenzene also forms stable adducts with electron donor molecules such as pyridine or triphenylphosphine.
Boron trifluoride is the inorganic compound with the formula BF3. This pungent colourless toxic gas forms white fumes in moist air. It is a useful Lewis acid and a versatile building block for other boron compounds.
A boride is a compound between boron and a less electronegative element, for example silicon boride (SiB3 and SiB6). The borides are a very large group of compounds that are generally high melting and are covalent more than ionic in nature. Some borides exhibit very useful physical properties. The term boride is also loosely applied to compounds such as B12As2 (N.B. Arsenic has an electronegativity higher than boron) that is often referred to as icosahedral boride.

Organoborane or organoboron compounds are chemical compounds of boron and carbon that are organic derivatives of BH3, for example trialkyl boranes. Organoboron chemistry or organoborane chemistry is the chemistry of these compounds. Organoboron compounds are important reagents in organic chemistry enabling many chemical transformations, the most important one called hydroboration.
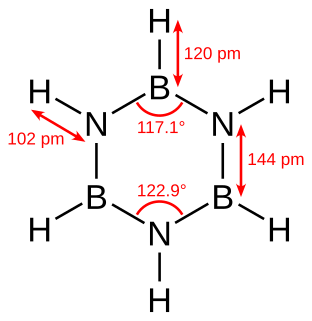
Borazine is an inorganic compound with the chemical formula B3H6N3. In this cyclic compound, the three BH units and three NH units alternate. The compound is isoelectronic and isostructural with benzene. For this reason borazine is sometimes referred to as “inorganic benzene”. Like benzene, borazine is a colourless liquid.
Aziridines are organic compounds containing the aziridine functional group, a three-membered heterocycle with one amine group (-NH-) and two methylene bridges. The parent compound is aziridine, with molecular formula C
2H
5N.
1,2,3-Triazole is one of a pair of isomeric chemical compounds with molecular formula C2H3N3, called triazoles, which have a five-membered ring of two carbon atoms and three nitrogen atoms. 1,2,3-Triazole is a basic aromatic heterocycle.
Azirines are three membered heterocyclic unsaturated compounds containing a nitrogen atom and related to the saturated analogue aziridine. They are highly reactive yet have reported in a few natural products such as Dysidazirine. There are two isomers of azirine: 1H-azirine with a carbon-carbon double bond are not stable and rearrange to the tautomeric 2H-azirine, a compound with a carbon-nitrogen double bond. 2H-Azirines can be considered strained imines and are isolable.

Aluminium diboride (AlB2) is a chemical compound made from the metal aluminium and the metalloid boron. It is one of two compounds of aluminium and boron, the other being AlB12, which are both commonly referred to as aluminium boride.
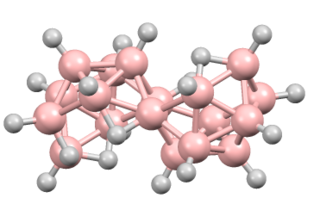
Octadecaborane is an inorganic compound, a borane with chemical formula B18H22. It is a colorless flammable solid, like many higher boron hydrides. Although the compound has no practical applications, its structure is of theoretical and pedagogical interest.
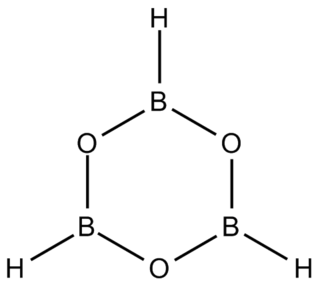
Boroxine (B3H3O3) is a 6-membered, heterocyclic compound composed of alternating oxygen and singly-hydrogenated boron atoms. Boroxine derivatives (boronic anhydrides) such as trimethylboroxine and triphenylboroxine also make up a broader class of compounds called boroxines. These compounds are solids that are usually in equilibrium with their respective boronic acids at room temperature. Beside being used in theoretical studies, boroxine is primarily used in the production of optics.
Boron monofluoride or fluoroborylene is a chemical compound with formula BF, one atom of boron and one of fluorine. It was discovered as an unstable gas and only in 2009 found to be a stable ligand combining with transition metals, in the same way as carbon monoxide. It is a subhalide, containing fewer than the normal number of fluorine atoms, compared with boron trifluoride. It can also be called a borylene, as it contains boron with two unshared electrons. BF is isoelectronic with carbon monoxide and dinitrogen and each molecule has 14 electrons.
In chemistry, an oxoborane is any chemical compound containing a boron atom with a terminal oxygen atom. The compound class is of some relevance to academic research. The parent compound, HBO, itself called "oxoborane", together with derivatives FBO (fluorine), ClBO chlorine, BrBO bromine, HOBO (hydroxyl) and MeBO (methyl) have been detected in matrix isolation or in the gasphase at high temperature. In these compounds the boron and oxygen form a triple bond prone to cyclotrimerization to boroxines.

Diborane(4) is a transient inorganic compound with the chemical formula B
2H
4. Stable derivatives are known.
Oximide is an unstable chemical compound, the cyclic imide of oxalic acid. Other names for this are the systematic name 2,3-Aziridinedione or oxalimide. The chemical formula is C2HNO2. Its core is a three member heterocycle, aziridine.














