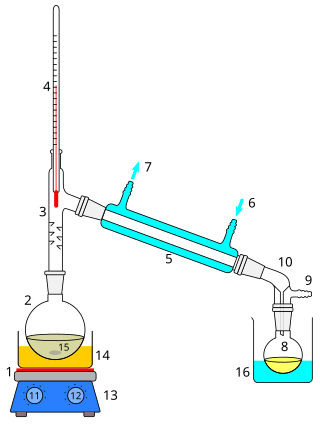
Distillation, also classical distillation, is the process of separating the component substances of a liquid mixture of two or more chemically discrete substances; the separation process is realized by way of the selective boiling of the mixture and the condensation of the vapors in a still.

A vacuum pump is a type of pump device that draws gas particles from a sealed volume in order to leave behind a partial vacuum. The first vacuum pump was invented in 1650 by Otto von Guericke, and was preceded by the suction pump, which dates to antiquity.
The following is a timeline of low-temperature technology and cryogenic technology. It also lists important milestones in thermometry, thermodynamics, statistical physics and calorimetry, that were crucial in development of low temperature systems.
A cryopump or a "cryogenic pump" is a vacuum pump that traps gases and vapours by condensing them on a cold surface, but are only effective on some gases. The effectiveness depends on the freezing and boiling points of the gas relative to the cryopump's temperature. They are sometimes used to block particular contaminants, for example in front of a diffusion pump to trap backstreaming oil, or in front of a McLeod gauge to keep out water. In this function, they are called a cryotrap, waterpump or cold trap, even though the physical mechanism is the same as for a cryopump.

A dehumidifier is an air conditioning device which reduces and maintains the level of humidity in the air. This is done usually for health or thermal comfort reasons or to eliminate musty odor and to prevent the growth of mildew by extracting water from the air. It can be used for household, commercial, or industrial applications. Large dehumidifiers are used in commercial buildings such as indoor ice rinks and swimming pools, as well as manufacturing plants or storage warehouses. Typical air conditioning systems combine dehumidification with cooling, by operating cooling coils below the dewpoint and draining away the water that condenses.

Liquid nitrogen (LN2) is nitrogen in a liquid state at low temperature. Liquid nitrogen has a boiling point of about −196 °C (−321 °F; 77 K). It is produced industrially by fractional distillation of liquid air. It is a colorless, mobile liquid whose viscosity is about one-tenth that of acetone (i.e. roughly one-thirtieth that of water at room temperature). Liquid nitrogen is widely used as a coolant.

A cryostat is a device used to maintain low cryogenic temperatures of samples or devices mounted within the cryostat. Low temperatures may be maintained within a cryostat by using various refrigeration methods, most commonly using cryogenic fluid bath such as liquid helium. Hence it is usually assembled into a vessel, similar in construction to a vacuum flask or Dewar. Cryostats have numerous applications within science, engineering, and medicine.

A McLeod gauge is a scientific instrument used to measure very low pressures, down to 10−6 Torr. It was invented in 1874 by Herbert McLeod (1841–1923). McLeod gauges were once commonly found attached to equipment that operates under vacuum, such as a lyophilizer. Today, however, these gauges have largely been replaced by electronic vacuum gauges.

A liquid-ring pump is a rotating positive-displacement gas pump, with liquid under centrifugal force acting as a seal.

A condensate pump is a specific type of pump used to pump the condensate (water) produced in an HVAC, refrigeration, condensing boiler furnace, or steam system.

A rotary vane pump is a type of positive-displacement pump that consists of vanes mounted to a rotor that rotates inside a cavity. In some cases, these vanes can have variable length and/or be tensioned to maintain contact with the walls as the pump rotates.
The sorption pump is a vacuum pump that creates a vacuum by adsorbing molecules on a very porous material like molecular sieve which is cooled by a cryogen, typically liquid nitrogen. The ultimate pressure is about 10−2 mbar. With special techniques this can be lowered till 10−7 mbar. The main advantages are the absence of oil or other contaminants, low cost and vibration free operation because there are no moving parts. The main disadvantages are that it cannot operate continuously and cannot effectively pump hydrogen, helium and neon, all gases with lower condensation temperature than liquid nitrogen. The main application is as a roughing pump for a sputter-ion pump in ultra-high vacuum experiments, for example in surface physics.

Vapour-compression refrigeration or vapor-compression refrigeration system (VCRS), in which the refrigerant undergoes phase changes, is one of the many refrigeration cycles and is the most widely used method for air conditioning of buildings and automobiles. It is also used in domestic and commercial refrigerators, large-scale warehouses for chilled or frozen storage of foods and meats, refrigerated trucks and railroad cars, and a host of other commercial and industrial services. Oil refineries, petrochemical and chemical processing plants, and natural gas processing plants are among the many types of industrial plants that often utilize large vapor-compression refrigeration systems. Cascade refrigeration systems may also be implemented using two compressors.

A turboexpander, also referred to as a turbo-expander or an expansion turbine, is a centrifugal or axial-flow turbine, through which a high-pressure gas is expanded to produce work that is often used to drive a compressor or generator.

The Schlenk line is a commonly used chemistry apparatus developed by Wilhelm Schlenk. It consists of a dual manifold with several ports. One manifold is connected to a source of purified inert gas, while the other is connected to a vacuum pump. The inert-gas line is vented through an oil bubbler, while solvent vapors and gaseous reaction products are prevented from contaminating the vacuum pump by a liquid-nitrogen or dry-ice/acetone cold trap. Special stopcocks or Teflon taps allow vacuum or inert gas to be selected without the need for placing the sample on a separate line.

Natural-gas processing is a range of industrial processes designed to purify raw natural gas by removing contaminants such as solids, water, carbon dioxide (CO2), hydrogen sulfide (H2S), mercury and higher molecular mass hydrocarbons (condensate) to produce pipeline quality dry natural gas for pipeline distribution and final use. Some of the substances which contaminate natural gas have economic value and are further processed or sold. Hydrocarbons that are liquid at ambient conditions: temperature and pressure (i.e., pentane and heavier) are called natural-gas condensate (sometimes also called natural gasoline or simply condensate).

In chemistry, a condenser is laboratory apparatus used to condense vapors – that is, turn them into liquids – by cooling them down.
An air separation plant separates atmospheric air into its primary components, typically nitrogen and oxygen, and sometimes also argon and other rare inert gases.
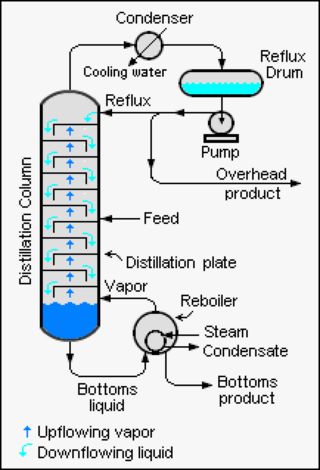
Reflux is a technique involving the condensation of vapors and the return of this condensate to the system from which it originated. It is used in industrial and laboratory distillations. It is also used in chemistry to supply energy to reactions over a long period of time.

A cryogenic gas plant is an industrial facility that creates molecular oxygen, molecular nitrogen, argon, krypton, helium, and xenon at relatively high purity. As air is made up of nitrogen, the most common gas in the atmosphere, at 78%, with oxygen at 19%, and argon at 1%, with trace gasses making up the rest, cryogenic gas plants separate air inside a distillation column at cryogenic temperatures to produce high purity gasses such as argon, nitrogen, oxygen, and many more with 1 ppm or less impurities. The process is based on the general theory of the Hampson-Linde cycle of air separation, which was invented by Carl von Linde in 1895.
















