Related Research Articles
Combinatorial chemistry comprises chemical synthetic methods that make it possible to prepare a large number of compounds in a single process. These compound libraries can be made as mixtures, sets of individual compounds or chemical structures generated by computer software. Combinatorial chemistry can be used for the synthesis of small molecules and for peptides.

In chemistry, a reagent or analytical reagent is a substance or compound added to a system to cause a chemical reaction, or test if one occurs. The terms reactant and reagent are often used interchangeably, but reactant specifies a substance consumed in the course of a chemical reaction. Solvents, though involved in the reaction mechanism, are usually not called reactants. Similarly, catalysts are not consumed by the reaction, so they are not reactants. In biochemistry, especially in connection with enzyme-catalyzed reactions, the reactants are commonly called substrates.
Cytotoxicity is the quality of being toxic to cells. Examples of toxic agents are toxic metals, toxic chemicals, microbe neurotoxins, radiation particles and even specific neurotransmitters when the system is out of balance. Also some types of venom, e.g. from the puff adder or brown recluse spider are toxic to cells.
An assay is an investigative (analytic) procedure in laboratory medicine, mining, pharmacology, environmental biology and molecular biology for qualitatively assessing or quantitatively measuring the presence, amount, or functional activity of a target entity. The measured entity is often called the analyte, the measurand, or the target of the assay. The analyte can be a drug, biochemical substance, chemical element or compound, or cell in an organism or organic sample. An assay usually aims to measure an analyte's intensive property and express it in the relevant measurement unit.
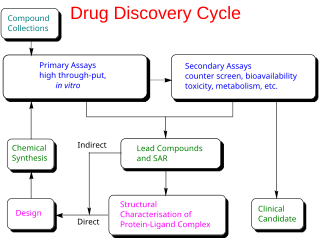
In the fields of medicine, biotechnology and pharmacology, drug discovery is the process by which new candidate medications are discovered.
Cheminformatics refers to the use of physical chemistry theory with computer and information science techniques—so called "in silico" techniques—in application to a range of descriptive and prescriptive problems in the field of chemistry, including in its applications to biology and related molecular fields. Such in silico techniques are used, for example, by pharmaceutical companies and in academic settings to aid and inform the process of drug discovery, for instance in the design of well-defined combinatorial libraries of synthetic compounds, or to assist in structure-based drug design. The methods can also be used in chemical and allied industries, and such fields as environmental science and pharmacology, where chemical processes are involved or studied.

Laboratory robotics is the act of using robots in biology, chemistry or engineering labs. For example, pharmaceutical companies employ robots to move biological or chemical samples around to synthesize novel chemical entities or to test pharmaceutical value of existing chemical matter. Advanced laboratory robotics can be used to completely automate the process of science, as in the Robot Scientist project.

High-throughput screening (HTS) is a method for scientific discovery especially used in drug discovery and relevant to the fields of biology, materials science and chemistry. Using robotics, data processing/control software, liquid handling devices, and sensitive detectors, high-throughput screening allows a researcher to quickly conduct millions of chemical, genetic, or pharmacological tests. Through this process one can quickly recognize active compounds, antibodies, or genes that modulate a particular biomolecular pathway. The results of these experiments provide starting points for drug design and for understanding the noninteraction or role of a particular location.

Medicinal or pharmaceutical chemistry is a scientific discipline at the intersection of chemistry and pharmacy involved with designing and developing pharmaceutical drugs. Medicinal chemistry involves the identification, synthesis and development of new chemical entities suitable for therapeutic use. It also includes the study of existing drugs, their biological properties, and their quantitative structure-activity relationships (QSAR).
A chemical compound microarray is a collection of organic chemical compounds spotted on a solid surface, such as glass and plastic. This microarray format is very similar to DNA microarray, protein microarray and antibody microarray. In chemical genetics research, they are routinely used for searching proteins that bind with specific chemical compounds, and in general drug discovery research, they provide a multiplex way to search potential drugs for therapeutic targets.
High-content screening (HCS), also known as high-content analysis (HCA) or cellomics, is a method that is used in biological research and drug discovery to identify substances such as small molecules, peptides, or RNAi that alter the phenotype of a cell in a desired manner. Hence high content screening is a type of phenotypic screen conducted in cells involving the analysis of whole cells or components of cells with simultaneous readout of several parameters. HCS is related to high-throughput screening (HTS), in which thousands of compounds are tested in parallel for their activity in one or more biological assays, but involves assays of more complex cellular phenotypes as outputs. Phenotypic changes may include increases or decreases in the production of cellular products such as proteins and/or changes in the morphology of the cell. Hence HCA typically involves automated microscopy and image analysis. Unlike high-content analysis, high-content screening implies a level of throughput which is why the term "screening" differentiates HCS from HCA, which may be high in content but low in throughput.
Hit to lead (H2L) also known as lead generation is a stage in early drug discovery where small molecule hits from a high throughput screen (HTS) are evaluated and undergo limited optimization to identify promising lead compounds. These lead compounds undergo more extensive optimization in a subsequent step of drug discovery called lead optimization (LO). The drug discovery process generally follows the following path that includes a hit to lead stage:
A chemical library or compound library is a collection of stored chemicals usually used ultimately in high-throughput screening or industrial manufacture. The chemical library can consist in simple terms of a series of stored chemicals. Each chemical has associated information stored in some kind of database with information such as the chemical structure, purity, quantity, and physiochemical characteristics of the compound.
High throughput biology is the use of automation equipment with classical cell biology techniques to address biological questions that are otherwise unattainable using conventional methods. It may incorporate techniques from optics, chemistry, biology or image analysis to permit rapid, highly parallel research into how cells function, interact with each other and how pathogens exploit them in disease.
Chemoproteomics entails a broad array of techniques used to identify and interrogate protein-small molecule interactions. Chemoproteomics complements phenotypic drug discovery, a paradigm that aims to discover lead compounds on the basis of alleviating a disease phenotype, as opposed to target-based drug discovery, in which lead compounds are designed to interact with predetermined disease-driving biological targets. As phenotypic drug discovery assays do not provide confirmation of a compound's mechanism of action, chemoproteomics provides valuable follow-up strategies to narrow down potential targets and eventually validate a molecule's mechanism of action. Chemoproteomics also attempts to address the inherent challenge of drug promiscuity in small molecule drug discovery by analyzing protein-small molecule interactions on a proteome-wide scale. A major goal of chemoproteomics is to characterize the interactome of drug candidates to gain insight into mechanisms of off-target toxicity and polypharmacology.
Pan-assay interference compounds (PAINS) are chemical compounds that often give false positive results in high-throughput screens. PAINS tend to react nonspecifically with numerous biological targets rather than specifically affecting one desired target. A number of disruptive functional groups are shared by many PAINS.

Aurora Biosciences was a biotechnology company founded in 1995 in San Diego to commercialize fluorescence assays based on Roger Y. Tsien's discoveries concerning green fluorescent protein and its uses in basic research - work for which Tsien eventually won the 2008 Nobel Prize in chemistry along with two other chemists.
The European Lead Factory is a public-private partnership that aims to accelerate early drug discovery in Europe. The European Lead Factory is funded by the Innovative Medicines Initiative and consists of a pan-European consortium that includes 7 pharmaceutical companies as well as partners from academia and small and medium-sized enterprises (SMEs).

Fluorescence polarization immunoassay (FPIA) is a class of in vitro biochemical test used for rapid detection of antibody or antigen in sample. FPIA is a competitive homogenous assay, that consists of a simple prepare and read method, without the requirement of separation or washing steps.

James Inglese is an American biochemist, the director of the Assay Development and Screening Technology laboratory at the National Center for Advancing Translational Sciences, a Center within the National Institutes of Health. His specialty is small molecule high throughput screening. Inglese's laboratory develops methods and strategies in molecular pharmacology with drug discovery applications. The work of his research group and collaborators focuses on genetic and infectious disease-associated biology.
References
- ↑ Archer JR (December 2004). "History, evolution, and trends in compound management for high throughput screening". Assay and Drug Development Technologies. 2 (6): 675–681. doi:10.1089/adt.2004.2.675. PMID 15674025.
- ↑ Yates I (March 2003). "Compound management comes of age" (PDF). Drug Discov World. 2: 35–42.
- ↑ Handen JS (2005). "Compound Management". Industrialization of Drug Discovery. CRC Press. ISBN 9780824723910.
- ↑ Wedin R (January–February 1999). "Taming the monster haystack: The challenge of compound management". Modern Drug Discovery. 2: 47–53.
- ↑ Yasgar A, Shinn P, Jadhav A, Auld D, Michael S, Zheng W, et al. (April 2008). "Compound Management for Quantitative High-Throughput Screening". Jala. 13 (2): 79–89. doi:10.1016/j.jala.2007.12.004. PMC 2390859 . PMID 18496600.
- ↑ "Compound Management & Integrity 2009". Chemistry Conferences. Archived from the original on 25 December 2008.