Related Research Articles
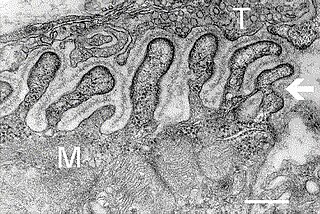
A neuromuscular junction is a chemical synapse between a motor neuron and a muscle fiber.

Snake venom is a highly toxic saliva containing zootoxins that facilitates in the immobilization and digestion of prey. This also provides defense against threats. Snake venom is injected by unique fangs during a bite, whereas some species are also able to spit venom.

The enzyme phospholipase A2 (EC 3.1.1.4, PLA2, systematic name phosphatidylcholine 2-acylhydrolase) catalyse the cleavage of fatty acids in position 2 of phospholipids, hydrolyzing the bond between the second fatty acid “tail” and the glycerol molecule:

Crotalus scutulatus, also known commonly as the Mojave rattlesnake, the Mojave green rattlesnake, and the Mohave rattlesnake, is a species of highly venomous pit viper in the family Viperidae. The species is native to the deserts of the southwestern United States and adjacent central Mexico. It is perhaps best known for its potent neurotoxic-hemotoxic venom. Two subspecies are recognized as being valid, including the nominate subspecies described here.
A latrotoxin is a high-molecular mass neurotoxin found in the venom of spiders of the genus Latrodectus as well as at least one species of another genus in the same family, Steatoda nobilis. Latrotoxins are the main active components of the venom and are responsible for the symptoms of latrodectism.

Convulxin is a snake venom toxin found in a tropical rattlesnake known as Crotalus durissus terrificus. It belongs to the family of hemotoxins, which destroy red blood cells or, as is the case with convulxin, induce blood coagulation.

Myotoxins are small, basic peptides found in snake venoms and lizard venoms. This involves a non-enzymatic mechanism that leads to severe muscle necrosis. These peptides act very quickly, causing instantaneous paralysis to prevent prey from escaping and eventually death due to diaphragmatic paralysis.
Taicatoxin (TCX) is a snake toxin that blocks voltage-dependent L-type calcium channels and small conductance Ca2+-activated K+ channels. The name taicatoxin (TAIpan + CAlcium + TOXIN) is derived from its natural source, the taipan snake, the site of its action, calcium channels, and from its function as a toxin. Taicatoxin was isolated from the venom of Australian taipan snake, Oxyuranus scutellatus scutellatus. TCX is a secreted protein, produced in the venom gland of the snake.

Crotalus durissus, known as the South American rattlesnake, tropical rattlesnake, and by other names, is a highly venomous pit viper species found in South America. It is the most widely distributed member of its genus. Currently, seven subspecies are recognized.
Taipoxin is a potent myo- and neurotoxin that was isolated from the venom of the coastal taipan Oxyuranus scutellatus or also known as the common taipan. Taipoxin like many other pre-synaptic neurotoxins are phospholipase A2 (PLA2) toxins, which inhibit/complete block the release of the motor transmitter acetylcholine and lead to death by paralysis of the respiratory muscles (asphyxia). It is the most lethal neurotoxin isolated from any snake venom to date.
Neuromuscular junction disease is a medical condition where the normal conduction through the neuromuscular junction fails to function correctly.
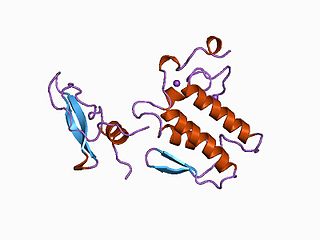
β-Bungarotoxin is a form of bungarotoxin that is fairly common in Krait venoms. It is the prototypic class of snake β-neurotoxins. There are at least five isoforms, coded β1 to β5, assembled from different combinations of A and Bchains.
Imperatoxin I (IpTx) is a peptide toxin derived from the venom of the African scorpion Pandinus imperator.
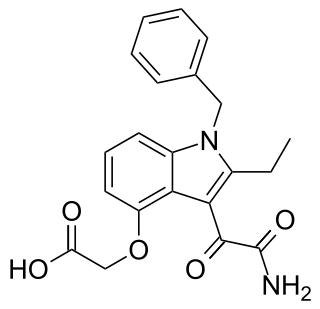
Varespladib is an inhibitor of the IIa, V, and X isoforms of secretory phospholipase A2 (sPLA2). The molecule acts as an anti-inflammatory agent by disrupting the first step of the arachidonic acid pathway of inflammation. From 2006 to 2012, varespladib was under active investigation by Anthera Pharmaceuticals as a potential therapy for several inflammatory diseases, including acute coronary syndrome and acute chest syndrome. The trial was halted in March 2012 due to inadequate efficacy. The selective sPLA2 inhibitor varespladib (IC50 value 0.009 μM in chromogenic assay, mole fraction 7.3X10-6) was studied in the VISTA-16 randomized clinical trial (clinicaltrials.gov Identifier: NCT01130246) and the results were published in 2014. The sPLA2 inhibition by varespladib in this setting seemed to be potentially harmful, and thus not a useful strategy for reducing adverse cardiovascular outcomes from acute coronary syndrome. Since 2016, scientific research has focused on the use of Varespladib as an inhibitor of snake venom toxins using various types of in vitro and in vivo models. Varespladib showed a significant inhibitory effect to snake venom PLA2 which makes it a potential first-line drug candidate in snakebite envenomation therapy. In 2019, the U.S. Food and Drug Administration (FDA) granted varespladib orphan drug status for its potential to treat snakebite.
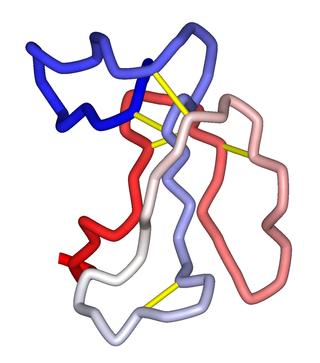
α-Neurotoxins are a group of neurotoxic peptides found in the venom of snakes in the families Elapidae and Hydrophiidae. They can cause paralysis, respiratory failure, and death. Members of the three-finger toxin protein family, they are antagonists of post-synaptic nicotinic acetylcholine receptors (nAChRs) in the neuromuscular synapse that bind competitively and irreversibly, preventing synaptic acetylcholine (ACh) from opening the ion channel. Over 100 α-neurotoxins have been identified and sequenced.

Venom in snakes and some lizards is a form of saliva that has been modified into venom over its evolutionary history. In snakes, venom has evolved to kill or subdue prey, as well as to perform other diet-related functions. While snakes occasionally use their venom in self defense, this is not believed to have had a strong effect on venom evolution. The evolution of venom is thought to be responsible for the enormous expansion of snakes across the globe.

Three-finger toxins are a protein superfamily of small toxin proteins found in the venom of snakes. Three-finger toxins are in turn members of a larger superfamily of three-finger protein domains which includes non-toxic proteins that share a similar protein fold. The group is named for its common structure consisting of three beta strand loops connected to a central core containing four conserved disulfide bonds. The 3FP protein domain has no enzymatic activity and is typically between 60-74 amino acid residues long. Despite their conserved structure, three-finger toxin proteins have a wide range of pharmacological effects. Most members of the family are neurotoxins that act on cholinergic intercellular signaling; the alpha-neurotoxin family interacts with muscle nicotinic acetylcholine receptors (nAChRs), the kappa-bungarotoxin family with neuronal nAChRs, and muscarinic toxins with muscarinic acetylcholine receptors (mAChRs).

Three-finger proteins or three-finger protein domains are a protein superfamily consisting of small, roughly 60-80 amino acid residue protein domains with a common tertiary structure: three beta strand loops extended from a hydrophobic core stabilized by disulfide bonds. The family is named for the outstretched "fingers" of the three loops. Members of the family have no enzymatic activity, but are capable of forming protein-protein interactions with high specificity and affinity. The founding members of the family, also the best characterized by structure, are the three-finger toxins found in snake venom, which have a variety of pharmacological effects, most typically by disruption of cholinergic signaling. The family is also represented in non-toxic proteins, which have a wide taxonomic distribution; 3FP domains occur in the extracellular domains of some cell-surface receptors as well as in GPI-anchored and secreted globular proteins, usually involved in signaling.
Notexin is a toxin produced by the tiger snake (Notechis scutatus). It is a myotoxic and presynaptic, neurotoxic phospholipase A2 (PLA2s). These are enzymes that hydrolyze the bond between a fatty acid tail and glycerol in fatty acids on the 2-position.
MiDCA1, short for Micrurus dumerili carinicauda 1, is a β-neurotoxin primarily affecting presynaptic synapses, where it interferes with the release of neurotransmitters by inhibiting potassium (K+) channels. This toxin belongs to the phospholipase A2 (PLA2) family but distinguishes itself by existing as a monomer, unlike some other PLA2 toxins. It occurs naturally in the venom of the coral snake Micrurus dumerili carinicauda.
References
- 1 2 Slotta, KH, Fraenkel-Conrat H. "Two Active Proteins from Rattlesnake Venom", Nature , 142, 213 (1938). doi:10.1038/142213a0.
- ↑ Rübsamen K, Breithaupt H, Habermann E (1971). "Biochemistry and Pharmacology of the Crotoxin Complex: I. Subfractionation and Recombination of the Crotoxin Complex", Naunyn-Schmiedebergs Archiv für Pharmakologie , 270, 274-288.
- 1 2 3 4 Fernandes C, Pazin W, Dreyer T et al. "Biophysical studies suggest a new structural arrangement of crotoxin and provide insights into its toxic mechanism", Scientific Reports , 7, 43885 (2017). doi:10.1038/srep43885.
- 1 2 Sampaio SC, Hyslop S, Fontes MRM, Prado-Franceschi J, Zambelli VO, Magro AJ, Brigatte P, Gutierrez VP, Cury Y (2010). "Crotoxin: Novel activities for a classic β-neurotoxin", Toxicon , 55(6), 1045-1060. doi:10.1016/j.toxicon.2010.01.011.
- ↑ Vital Brazil et al (1966). "Pharmacology of crystalline crotoxin - toxicity", Memórias do Instituto Butantan, 33, 973-980.
- ↑ Pereanez JA, Gómez ID, Patino AC (2012). "Relationship between the structure and the enzymatic activity of crotoxin complex and its phospholipase A2 subunit: An in silico approach", Journal of Molecular Graphics and Modelling , 35, 36-42. doi:10.1016/j.jmgm.2012.01.004.
- ↑ Faure G, Xu H, Saul FA (2011). "Crystal Structure of Crotoxin Reveals Key Residues Involved in the Stability and Toxicity of This Potent Heterodimeric β-Neurotoxin", J. Mol. Biol. , 412(2), 176-191. doi:10.1016/j.jmb.2011.07.027.
- ↑ Faure G, Harvey AL, Thomson E, Saliou B, Radvanyi F, Bon C (1993). "Comparison of crotoxin isoforms reveals that stability of the complex plays a major role in its pharmacological action", European Journal of Biochemistry , 214(2), 491-496. doi:10.1111/j.1432-1033.1993.tb17946.x.
- ↑ Choumet V, Bouchier C, Délot E, Faure G, Saliou B, Bon C (1996). "Structure and Function Relationship of Crotoxin, a Heterodimeric Neurotoxin Phospholipase A2 from the Venom of a South-American Rattlesnake", In: Singh B.R., Tu A.T. (eds), Natural Toxins 2, Advances in Experimental Medicine and Biology, vol 391. Springer, Boston, MA.
- ↑ Vulfius, Catherine A., et al (2017). "Pancreatic and Snake Venom Presynaptically Active Phospholipases A2 Inhibit Nicotinic Acetylcholine Receptors", PLOS One , 12(10), doi:10.1371/journal.pone.0186206.
- 1 2 Bon, Cassian, et al (1979). "Postsynaptic Effects of Crotoxin and of Its Isolated Subunits", European Journal of Biochemistry , 99(3), 471–482. doi:10.1111/j.1432-1033.1979.tb13278.x.
- ↑ Cavalcante, Walter L.g., et al (2017). "Neuromuscular Paralysis by the Basic Phospholipase A 2 Subunit of Crotoxin from Crotalus Durissus Terrificus Snake Venom Needs Its Acid Chaperone to Concurrently Inhibit Acetylcholine Release and Produce Muscle Blockage", Toxicology and Applied Pharmacology , 334, 8–17. doi:10.1016/j.taap.2017.08.021.
- ↑ Montecucco, Cesare, et al (2009). "Different Mechanisms of Inhibition of Nerve Terminals by Botulinum and Snake Presynaptic Neurotoxins", Toxicon , 54(5), 561–564. doi:10.1016/j.toxicon.2008.12.012.
- ↑ Kini, R Manjunatha (2003). "Excitement Ahead: Structure, Function and Mechanism of Snake Venom Phospholipase A2 Enzymes", Toxicon , 42(8), 827–840. doi:10.1016/j.toxicon.2003.11.002.
- ↑ Rigoni, Michela, et al (2007). "Calcium Influx and Mitochondrial Alterations at Synapses Exposed to Snake Neurotoxins or Their Phospholipid Hydrolysis Products", Journal of Biological Chemistry , 282(15), 11238–11245. doi:10.1074/jbc.m610176200.
- ↑ Laustsen AH (2016). "Toxin synergism in snake venoms", Toxin Reviews , 35(3-4), 165-170. doi:10.1080/15569543.2016.1220397.
- ↑ Chiung Chang C, Dong Lee J (1977). "Crotoxin, the Neurotoxin of South American Rattlesnake Venom, is a Presynaptic Toxin Acting Like β-Bungarotoxin", Arch. Pharmacol. , 296, 159-168.
- 1 2 Amaral CFS, Campolina D, Dias MB, Bueno CM, Chávez-Olortegui C, Penaforte CL, Diniz CR, Rezende NA (1997). "Time factor in the detection of circulating whole venom and crotoxin and efficacy of antivenom therapy in patients envenomed by Crotalus durissus", Toxicon , 35(5), 699-704.
- 1 2 3 Cura JE, Blanzaco DP, Brisson C, Cura MA, Cabrol R, Larrateguy L, Mendez C, Sechi JC, Silveira JS, Theiller E, Roodt AR de, Vidal JC (2002). "Phase I and Pharmacokinetics Study of Crotoxin (cytotoxin PLA2, NSC-624244) in Patients with Advanced Cancer", Clinical Cancer Research , 8, 1033-1041.
- ↑ Magalhaes T, Proietti Viotti A, Teperino Gomes R, Viana de Freitas T (2001). "Effect of membrane composition and of co-encapsulation of immunostimulants in a liposome-entrapped crotoxin", Biotechnol. Appl. Biochem. , 33, 61-64.
- ↑ de Araújo Pimenta L, de Almeida MES, Bretones ML et al. "Crotoxin promotes macrophage reprogramming towards an antiangiogenic phenotype", Scientific Reports , 9, 4281 (2019). doi:10.1038/s41598-019-40903-0.
- ↑ de Andrade CM, Rey FM, Cintra ACO, Sampaio SV, Torqueti MR (2019). "Effects of crotoxin, a neurotoxin from Crotalus durissus terrificus snake venom, on human endothelial cells", International Journal of Biological Macromolecules , 134(1), 613-621. doi:10.1016/j.ijbiomac.2019.05.019.
- 1 2 Barros Ribeiro G de, Almeida HC de, Toledo Velarde D (2012). "Crotoxin in humans: analysis of the effects on extraocular and facial muscles", Arquivos Brasileiros de Oftalmologia, 75(6). doi:10.1590/S0004-27492012000600002.
- ↑ International Application Published Under the Patent Cooperation Treaty (PCT). "International Publication Number: WO 2009/018643 A2", Published: 6 August 2008.
- ↑ Sartim MA, Menaldo DL, Sampaio SV (2018). "Immunotherapeutic potential of Crotoxin: anti-inflammatory and immunosuppressive properties", Journal of Venomous Animals and Toxins including Tropical Diseases , 24(39). doi:10.1186/s40409-018-0178-3.
- ↑ National Cancer Institute. "Clinical Trials Using Crotoxin", retrieved on 16 March 2020.