Related Research Articles

Proteolysis is the breakdown of proteins into smaller polypeptides or amino acids. Uncatalysed, the hydrolysis of peptide bonds is extremely slow, taking hundreds of years. Proteolysis is typically catalysed by cellular enzymes called proteases, but may also occur by intra-molecular digestion. Low pH or high temperatures can also cause proteolysis non-enzymatically.

A protease is an enzyme that catalyzes proteolysis, the breakdown of proteins into smaller polypeptides or single amino acids. They do this by cleaving the peptide bonds within proteins by hydrolysis, a reaction where water breaks bonds. Proteases are involved in many biological functions, including digestion of eaten proteins, protein catabolism, and cell signalling.

Phagocytosis is the process by which a cell uses its plasma membrane to engulf a large particle, giving rise to an internal compartment called the phagosome. It is one type of endocytosis.

An exoenzyme, or extracellular enzyme, is an enzyme that is secreted by a cell and functions outside of that cell. Exoenzymes are produced by both prokaryotic and eukaryotic cells and have been shown to be a crucial component of many biological processes. Most often these enzymes are involved in the breakdown of larger macromolecules. The breakdown of these larger macromolecules is critical for allowing their constituents to pass through the cell membrane and enter into the cell. For humans and other complex organisms, this process is best characterized by the digestive system which breaks down solid food via exoenzymes. The small molecules, generated by the exoenzyme activity, enter into cells and are utilized for various cellular functions. Bacteria and fungi also produce exoenzymes to digest nutrients in their environment, and these organisms can be used to conduct laboratory assays to identify the presence and function of such exoenzymes. Some pathogenic species also use exoenzymes as virulence factors to assist in the spread of these disease causing microorganisms. In addition to the integral roles in biological systems, different classes of microbial exoenzymes have been used by humans since pre-historic times for such diverse purposes as food production, biofuels, textile production and in the paper industry. Another important role that microbial exoenzymes serve is in the natural ecology and bioremediation of terrestrial and marine environments.
Hydrolase is a class of enzyme that commonly perform as biochemical catalysts that use water to break a chemical bond, which typically results in dividing a larger molecule to smaller molecules. Some common examples of hydrolase enzymes are esterases including lipases, phosphatases, glycosidases, peptidases, and nucleosidases.
Digestive enzymes are a group of enzymes that break down polymeric macromolecules into their smaller building blocks, in order to facilitate their absorption by the body. Digestive enzymes are found in the digestive tracts of animals and in the tracts of carnivorous plants, where they aid in the digestion of food, as well as inside cells, especially in their lysosomes, where they function to maintain cellular survival. Digestive enzymes of diverse specificities are found in the saliva secreted by the salivary glands, in the secretions of cells lining the stomach, in the pancreatic juice secreted by pancreatic exocrine cells, and in the secretions of cells lining the small and large intestines.

Cathepsins are proteases found in all animals as well as other organisms. There are approximately a dozen members of this family, which are distinguished by their structure, catalytic mechanism, and which proteins they cleave. Most of the members become activated at the low pH found in lysosomes. Thus, the activity of this family lies almost entirely within those organelles. There are, however, exceptions such as cathepsin K, which works extracellularly after secretion by osteoclasts in bone resorption. Cathepsins have a vital role in mammalian cellular turnover.

In cell biology, a phagosome is a vesicle formed around a particle engulfed by a phagocyte via phagocytosis. Professional phagocytes include macrophages, neutrophils, and dendritic cells (DCs). A phagosome is formed by the fusion of the cell membrane around a microorganism, a senescent cell or an apoptotic cell. Phagosomes have membrane-bound proteins to recruit and fuse with lysosomes to form mature phagolysosomes. The lysosomes contain hydrolytic enzymes and reactive oxygen species (ROS) which kill and digest the pathogens. Phagosomes can also form in non-professional phagocytes, but they can only engulf a smaller range of particles, and do not contain ROS. The useful materials from the digested particles are moved into the cytosol, and waste is removed by exocytosis. Phagosome formation is crucial for tissue homeostasis and both innate and adaptive host defense against pathogens.

A catalytic triad is a set of three coordinated amino acids that can be found in the active site of some enzymes. Catalytic triads are most commonly found in hydrolase and transferase enzymes. An Acid-Base-Nucleophile triad is a common motif for generating a nucleophilic residue for covalent catalysis. The residues form a charge-relay network to polarise and activate the nucleophile, which attacks the substrate, forming a covalent intermediate which is then hydrolysed to release the product and regenerate free enzyme. The nucleophile is most commonly a serine or cysteine amino acid, but occasionally threonine or even selenocysteine. The 3D structure of the enzyme brings together the triad residues in a precise orientation, even though they may be far apart in the sequence.
Cysteine proteases, also known as thiol proteases, are enzymes that degrade proteins. These proteases share a common catalytic mechanism that involves a nucleophilic cysteine thiol in a catalytic triad or dyad.
Exocrine pancreatic insufficiency (EPI) is the inability to properly digest food due to a lack of digestive enzymes made by the pancreas. EPI is found in humans afflicted with cystic fibrosis and Shwachman–Diamond syndrome, and is common in dogs. EPI is caused by a progressive loss of the pancreatic cells that make digestive enzymes; loss of digestive enzymes leads to maldigestion and malabsorption of nutrients from normal digestive processes.

2-Arachidonoylglycerol (2-AG) is an endocannabinoid, an endogenous agonist of the CB1 receptor and the primary endogenous ligand for the CB2 receptor. It is an ester formed from the omega-6 fatty acid arachidonic acid and glycerol. It is present at relatively high levels in the central nervous system, with cannabinoid neuromodulatory effects. It has been found in maternal bovine and human milk. The chemical was first described in 1994-1995, although it had been discovered some time before that. The activities of phospholipase C (PLC) and diacylglycerol lipase (DAGL) mediate its formation. 2-AG is synthesized from arachidonic acid-containing diacylglycerol (DAG).
Serine hydrolases are one of the largest known enzyme classes comprising approximately ~200 enzymes or 1% of the genes in the human proteome. A defining characteristic of these enzymes is the presence of a nucleophilic serine in their active site, which is used for the hydrolysis of substrates. Catalysis proceeds by the formation of an acyl-enzyme intermediate through this serine, followed by water/hydroxide-induced saponification of the intermediate and regeneration of the enzyme. Unlike other non-catalytic serines, the nucleophilic serine of these hydrolases is typically activated by a proton relay involving a catalytic triad consisting of the serine, an acidic residue and a basic residue, although variations on this mechanism exist.

Insulin-degrading enzyme, also known as IDE, is an enzyme.

Gastric lipase, also known as LIPF, is an enzymatic protein that, in humans, is encoded by the LIPF gene.

In molecular biology Proteinase K is a broad-spectrum serine protease. The enzyme was discovered in 1974 in extracts of the fungus Engyodontium album. Proteinase K is able to digest hair (keratin), hence, the name "Proteinase K". The predominant site of cleavage is the peptide bond adjacent to the carboxyl group of aliphatic and aromatic amino acids with blocked alpha amino groups. It is commonly used for its broad specificity. This enzyme belongs to Peptidase family S8 (subtilisin). The molecular weight of Proteinase K is 28,900 daltons.
Κ-casein, or kappa casein, is a mammalian milk protein involved in a number of important physiological processes. In the gut, the ingested protein is split into an insoluble peptide and a soluble hydrophilic glycopeptide (caseinomacropeptide). Caseinomacropeptide is responsible for increased efficiency of digestion, prevention of neonate hypersensitivity to ingested proteins, and inhibition of gastric pathogens.

A lipase is any enzyme that catalyzes the hydrolysis of fats (lipids). Lipases are a subclass of the esterases.
Keratinases are proteolytic enzymes that digest keratin.
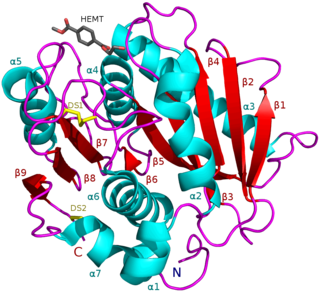
PETases are an esterase class of enzymes that catalyze the hydrolysis of polyethylene terephthalate (PET) plastic to monomeric mono-2-hydroxyethyl terephthalate (MHET). The idealized chemical reaction is (where n is the number of monomers in the polymer chain):
References
- ↑ Svendsen A (2000). "Lipase protein engineering". Biochim Biophys Acta. 1543 (2): 223–228. doi:10.1016/S0167-4838(00)00239-9. PMID 11150608.
- ↑ Jermyn, M. A. (1957). "Mechanism of Carbohydrase Action". Science. 125 (3236): 12–15. doi:10.1126/science.125.3236.12. PMID 13390960.
- ↑ Barrett A.J., Rawlings ND, Woessner JF. The Handbook of Proteolytic Enzymes, 2nd ed. Academic Press, 2003. ISBN 0-12-079610-4.
- ↑ Hedstrom L. Serine Protease Mechanism and Specificity. Chem Rev 2002;102:4501-4523.