
Vulcanization is a range of processes for hardening rubbers. The term originally referred exclusively to the treatment of natural rubber with sulfur, which remains the most common practice. It has also grown to include the hardening of other (synthetic) rubbers via various means. Examples include silicone rubber via room temperature vulcanizing and chloroprene rubber (neoprene) using metal oxides.

N,N-Dimethylaniline (DMA) is an organic chemical compound, a substituted derivative of aniline. It is a tertiary amine, featuring a dimethylamino group attached to a phenyl group. This oily liquid is colourless when pure, but commercial samples are often yellow. It is an important precursor to dyes such as crystal violet.

p-Phenylenediamine (PPD) is an organic compound with the formula C6H4(NH2)2. This derivative of aniline is a white solid, but samples can darken due to air oxidation. It is mainly used as a component of engineering polymers and composites like kevlar. It is also an ingredient in hair dyes and is occasionally used as a substitute for henna.
Accelerants, or accelerators, are substances that increase the rate of a natural or artificial chemical process. They play a major role in chemistry, as most chemical reactions can be hastened with an accelerant. Understanding accelerants is crucial in forensic science, engineering, and other fields where controlled chemical reactions are essential. Accelerants function by either altering a chemical bond, speeding up a chemical process, or changing the reaction conditions. Unlike catalysts, accelerants may be consumed during the process.
In organic chemistry, ozonolysis is an organic reaction where the unsaturated bonds are cleaved with ozone. Multiple carbon–carbon bond are replaced by carbonyl groups, such as aldehydes, ketones, and carboxylic acids. The reaction is predominantly applied to alkenes, but alkynes and azo compounds are also susceptible to cleavage. The outcome of the reaction depends on the type of multiple bond being oxidized and the work-up conditions.

Triphenylphosphine (IUPAC name: triphenylphosphane) is a common organophosphorus compound with the formula P(C6H5)3 and often abbreviated to PPh3 or Ph3P. It is versatile compound that is widely used as a reagent in organic synthesis and as a ligand for transition metal complexes, including ones that serve as catalysts in organometallic chemistry. PPh3 exists as relatively air stable, colorless crystals at room temperature. It dissolves in non-polar organic solvents such as benzene and diethyl ether.

Benzothiazole, or more specifically 1,3-benzothiazole, is an aromatic heterocyclic compound with the chemical formula C
7H
5NS. It is colorless, slightly viscous liquid. Although the parent compound, benzothiazole is not widely used, many of its derivatives are found in commercial products or in nature. Firefly luciferin can be considered a derivative of benzothiazole.

Dimethylamine is an organic compound with the formula (CH3)2NH. This secondary amine is a colorless, flammable gas with an ammonia-like odor. Dimethylamine is commonly encountered commercially as a solution in water at concentrations up to around 40%. An estimated 270,000 tons were produced in 2005.

o-Phenylenediamine (OPD) is an organic compound with the formula C6H4(NH2)2. This aromatic diamine is an important precursor to many heterocyclic compounds. OPD is a white compound although samples appear darker owing to oxidation by air. It is isomeric with m-phenylenediamine and p-phenylenediamine.

Thiocarbanilide is an organic chemical compound with the formula (C6H5NH)2CS. This white solid is a derivative of thiourea. It is prepared by the reaction of aniline and carbon disulfide.
A diamine is an amine with exactly two amino groups. Diamines are used as monomers to prepare polyamides, polyimides, and polyureas. The term diamine refers mostly to primary diamines, as those are the most reactive.

In organic chemistry, a dithiocarbamate is a functional group with the general formula R2N−C(=S)−S−R and structure >N−C(=S)−S−. It is the analog of a carbamate in which both oxygen atoms are replaced by sulfur atoms.

tert-Butylamine (also erbumine and other names) is an organic chemical compound with the formula (CH3)3CNH2. It is a colorless liquid with a typical amine-like odor. tert-Butylamine is one of the four isomeric amines of butane, the others being n-butylamine, sec-butylamine and isobutylamine.
Azobenzene reductase also known as azoreductase (EC 1.7.1.6) is an enzyme that catalyzes the chemical reaction:

An antiozonant, also known as anti-ozonant, is an organic compound that prevents or retards damage caused by ozone. The most important antiozonants are those which prevent degradation of elastomers like rubber. A number of research projects study the application of another type of antiozonants to protect plants as well as salmonids that are affected by the chemicals.
George Oenslager was a Goodrich chemist who discovered that a derivative of aniline accelerated the vulcanization of rubber with sulfur. He first introduced carbon black as a rubber reinforcing agent in 1912.
David Spence was one of the pioneering rubber chemists. He helped the war effort during the Second World War by devising new ways of extracting natural rubbers from plants, and worked to improve the processing of the rubber. Over the course of his career, he worked to improve the dyeing processes for rubber products and the vulcanization of rubber, and in developing new accelerants for strengthening lower-quality natural rubber. In 1941, he became the first recipient of the Charles Goodyear Medal, awarded by the American Chemical Society.

2-Mercaptobenzothiazole is an organosulfur compound with the formula C6H4(NH)SC=S. A white solid, it is used in the sulfur vulcanization of rubber.

Sulfur vulcanization is a chemical process for converting natural rubber or related polymers into materials of varying hardness, elasticity, and mechanical durability by heating them with sulfur or sulfur-containing compounds. Sulfur forms cross-linking bridges between sections of polymer chains which affects the mechanical and electronic properties. Many products are made with vulcanized rubber, including tires, shoe soles, hoses, and conveyor belts. The term vulcanization is derived from Vulcan, the Roman god of fire.
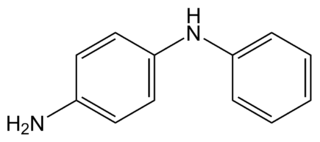
4-Aminodiphenylamine is a diphenylamine with an additional amine substituent. This dimer of aniline has various industrial uses, including as a hair dye ingredient, but also has raised concerns about toxicity by skin contact. It is also a starting material for the synthesis of 6PPD, an antiozonant for various rubber products. A colorimetric test for the quantitative analysis of nitrite, at levels below 100 nanograms per milliliter, is based on nitrite-catalyzed coupling of 4-aminodiphenylamine with N,N-dimethylaniline.















