
Combustion, or burning, is a high-temperature exothermic redox chemical reaction between a fuel and an oxidant, usually atmospheric oxygen, that produces oxidized, often gaseous products, in a mixture termed as smoke. Combustion does not always result in fire, because a flame is only visible when substances undergoing combustion vaporize, but when it does, a flame is a characteristic indicator of the reaction. While activation energy must be supplied to initiate combustion, the heat from a flame may provide enough energy to make the reaction self-sustaining.

In physics, physical chemistry and engineering, fluid dynamics is a subdiscipline of fluid mechanics that describes the flow of fluids—liquids and gases. It has several subdisciplines, including aerodynamics and hydrodynamics. Fluid dynamics has a wide range of applications, including calculating forces and moments on aircraft, determining the mass flow rate of petroleum through pipelines, predicting weather patterns, understanding nebulae in interstellar space and modelling fission weapon detonation.
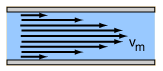
Laminar flow is the property of fluid particles in fluid dynamics to follow smooth paths in layers, with each layer moving smoothly past the adjacent layers with little or no mixing. At low velocities, the fluid tends to flow without lateral mixing, and adjacent layers slide past one another smoothly. There are no cross-currents perpendicular to the direction of flow, nor eddies or swirls of fluids. In laminar flow, the motion of the particles of the fluid is very orderly with particles close to a solid surface moving in straight lines parallel to that surface. Laminar flow is a flow regime characterized by high momentum diffusion and low momentum convection.
In fluid dynamics, turbulence or turbulent flow is fluid motion characterized by chaotic changes in pressure and flow velocity. It is in contrast to a laminar flow, which occurs when a fluid flows in parallel layers, with no disruption between those layers.

A flame is the visible, gaseous part of a fire. It is caused by a highly exothermic chemical reaction taking place in a thin zone. When flames are hot enough to have ionized gaseous components of sufficient density, they are then considered plasma.

In combustion, a diffusion flame is a flame in which the oxidizer and fuel are separated before burning. Contrary to its name, a diffusion flame involves both diffusion and convection processes. The name diffusion flame was first suggested by S.P. Burke and T.E.W. Schumann in 1928, to differentiate from premixed flame where fuel and oxidizer are premixed prior to burning. The diffusion flame is also referred to as nonpremixed flame. The burning rate is however still limited by the rate of diffusion. Diffusion flames tend to burn slower and to produce more soot than premixed flames because there may not be sufficient oxidizer for the reaction to go to completion, although there are some exceptions to the rule. The soot typically produced in a diffusion flame becomes incandescent from the heat of the flame and lends the flame its readily identifiable orange-yellow color. Diffusion flames tend to have a less-localized flame front than premixed flames.

A premixed flame is a flame formed under certain conditions during the combustion of a premixed charge of fuel and oxidiser. Since the fuel and oxidiser—the key chemical reactants of combustion—are available throughout a homogeneous stoichiometric premixed charge, the combustion process once initiated sustains itself by way of its own heat release. The majority of the chemical transformation in such a combustion process occurs primarily in a thin interfacial region which separates the unburned and the burned gases. The premixed flame interface propagates through the mixture until the entire charge is depleted. The propagation speed of a premixed flame is known as the flame speed which depends on the convection-diffusion-reaction balance within the flame, i.e. on its inner chemical structure. The premixed flame is characterised as laminar or turbulent depending on the velocity distribution in the unburned pre-mixture.

In fluid dynamics, an eddy is the swirling of a fluid and the reverse current created when the fluid is in a turbulent flow regime. The moving fluid creates a space devoid of downstream-flowing fluid on the downstream side of the object. Fluid behind the obstacle flows into the void creating a swirl of fluid on each edge of the obstacle, followed by a short reverse flow of fluid behind the obstacle flowing upstream, toward the back of the obstacle. This phenomenon is naturally observed behind large emergent rocks in swift-flowing rivers.
Turbulent diffusion is the transport of mass, heat, or momentum within a system due to random and chaotic time dependent motions. It occurs when turbulent fluid systems reach critical conditions in response to shear flow, which results from a combination of steep concentration gradients, density gradients, and high velocities. It occurs much more rapidly than molecular diffusion and is therefore extremely important for problems concerning mixing and transport in systems dealing with combustion, contaminants, dissolved oxygen, and solutions in industry. In these fields, turbulent diffusion acts as an excellent process for quickly reducing the concentrations of a species in a fluid or environment, in cases where this is needed for rapid mixing during processing, or rapid pollutant or contaminant reduction for safety.
In combustion engineering and explosion studies, the Markstein number characterizes the effect of local heat release of a propagating flame on variations in the surface topology along the flame and the associated local flame front curvature. The dimensionless Markstein number is defined as:
The Sugden Award is an annual award for contributions to combustion research. The prize is awarded by the British Section of The Combustion Institute for the published paper with at least one British Section member as author, which makes the most significant contribution to combustion research. The prize is named after Sir Morris Sugden.
Laminar flame speed is an intrinsic characteristic of premixed combustible mixtures. It is the speed at which an un-stretched laminar flame will propagate through a quiescent mixture of unburned reactants. Laminar flame speed is given the symbol sL. According to the thermal flame theory of Ernest-François Mallard and Le Chatelier, the un-stretched laminar flame speed is dependent on only three properties of a chemical mixture: the thermal diffusivity of the mixture, the reaction rate of the mixture and the temperature through the flame zone:
A laminar flow reactor (LFR) is a type of chemical reactor that uses laminar flow to control reaction rate, and/or reaction distribution. LFR is generally a long tube with constant diameter that is kept at constant temperature. Reactants are injected at one end and products are collected and monitored at the other. Laminar flow reactors are often used to study an isolated elementary reaction or multi-step reaction mechanism.
Combustion models for CFD refers to combustion models for computational fluid dynamics. Combustion is defined as a chemical reaction in which a hydrocarbon fuel reacts with an oxidant to form products, accompanied with the release of energy in the form of heat. Being the integral part of various engineering applications like: internal combustion engines, aircraft engines, rocket engines, furnaces, and power station combustors, combustion manifests itself as a wide domain during the design, analysis and performance characteristics stages of the above-mentioned applications. With the added complexity of chemical kinetics and achieving reacting flow mixture environment, proper modeling physics has to be incorporated during computational fluid dynamic (CFD) simulations of combustion. Hence the following discussion presents a general outline of the various adequate models incorporated with the Computational fluid dynamic code to model the process of combustion.
The simple chemical reacting system (SCRS) is one of the combustion models for computational fluid dynamics. This model helps us to determine the process of combustion which is a vital phenomenon used in many engineering applications like aircraft engines, internal combustion engines, rocket engines, industrial furnaces, and power station combustors. The simple chemical reacting system (SCRS) refers the global nature of the combustion process considering only the final species concentrations. The detailed kinetics of the process is generally neglected and it postulates that combustion does proceed via a global one-step without intermediates. Infinitely fast chemical reaction is assumed with oxidants reacting in stoichiometric proportions to form products. SCRS considers the reaction to be irreversible i.e. rate of reverse reaction is presumed to be very low.
Chemical reaction models transform physical knowledge into a mathematical formulation that can be utilized in computational simulation of practical problems in chemical engineering. Computer simulation provides the flexibility to study chemical processes under a wide range of conditions. Modeling of a chemical reaction involves solving conservation equations describing convection, diffusion, and reaction source for each component species.
The laminar flamelet model is a mathematical method for modelling turbulent combustion. The laminar flamelet model is formulated specifically as a model for non-premixed combustion
In combustion, a Burke–Schumann flame is a type of diffusion flame, established at the mouth of the two concentric ducts, by issuing fuel and oxidizer from the two region respectively. It is named after S.P. Burke and T.E.W. Schumann, who were able to predict the flame height and flame shape using their simple analysis of infinitely fast chemistry in 1928 at the First symposium on combustion.
Mixture fraction is a quantity used in combustion studies that measures the mass fraction of one stream of a mixture formed by two feed streams, one the fuel stream and the other the oxidizer stream. Both the feed streams are allowed to have inert gases. The mixture fraction definition is usually normalized such that it approaches unity in the fuel stream and zero in the oxidizer stream. The mixture-fraction variable is commonly used as a replacement for the physical coordinate normal to the flame surface, in nonpremixed combustion.

A high pressure jet is a stream of pressurized fluid that is released from an environment at a significantly higher pressure than ambient pressure from a nozzle or orifice, due to operational or accidental release. In the field of safety engineering, the release of toxic and flammable gases has been the subject of many R&D studies because of the major risk that they pose to the health and safety of workers, equipment and environment. Intentional or accidental release may occur in an industrial settings like natural gas processing plants, oil refineries and hydrogen storage facilities.
















