Mechanism
The hallmark feature of oxytosis/ferroptosis is the iron-dependent accumulation of oxidatively damaged phospholipids, i.e., lipid peroxides. The implication of Fenton chemistry via iron is crucial for the generation of reactive oxygen species and this feature can be exploited by sequestering iron in lysosomes. [32] Ferroptosis has been shown to involve distinct cellular organelles, [33] which includes peroxisomes, [34] mitochondria, [35] the endoplasmic reticulum (ER) [36] and lysosomes. [37] [38] [39] It has been a debate in the scientific community, where ferroptosis is initiated in the cell, and now research points to the lysosome, [39] where the chemical environment (iron, pH an hydrogen peroxide) are favorable. Oxidation of phospholipids can occur when free radicals abstract electrons from a lipid molecule (typically affecting polyunsaturated fatty acids), thereby promoting their oxidation.
The primary cellular mechanism of protection against oxytosis/ferroptosis is mediated by the selenoprotein GPX4, a glutathione-dependent hydroperoxidase that converts lipid peroxides into non-toxic lipid alcohols. [40] [41] Recently, a second parallel protective pathway was independently discovered by two labs that involves the oxidoreductase FSP1 (also known as AIFM2). [42] [43] FSP1 enzymatically reduces non-mitochondrial coenzyme Q10 (CoQ10), thereby generating a potent lipophilic antioxidant that suppresses the propagation of lipid peroxides. [42] [43] Vitamin K is also reduced by FSP1 to a hydroquinone species that also acts as a radical-trapping antoxidant and suppressor of ferroptosis. [44] A similar mechanism for a cofactor moonlighting as a diffusable antioxidant was discovered in the same year for tetrahydrobiopterin (BH4), a product of the rate-limiting enzyme GTP cyclohdrolase 1 (GCH1). [45] [46]
Replacing natural polyunsaturated fatty acids (PUFA) with deuterated PUFA (dPUFA), which have deuterium in place of the bis-allylic hydrogens, can prevent cell death induced by erastin or RSL3. [47] These deuterated PUFAs effectively inhibit ferroptosis and various chronic degenerative diseases associated with ferroptosis. [48]
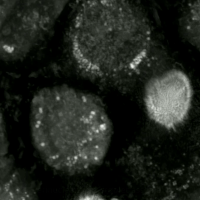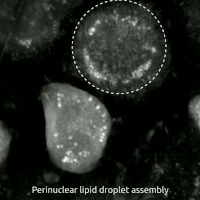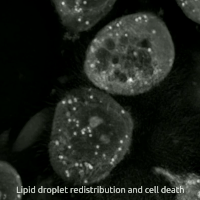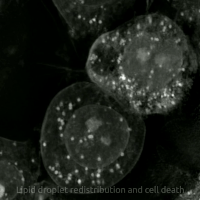
Live-cell imaging has been used to observe the morphological changes that cells undergo during oxytosis/ferroptosis. Initially the cell contracts and then begins to swell. Perinuclear lipid assembly is observed immediately before oxytosis/ferroptosis occurs. After the process is complete, lipid droplets are redistributed throughout the cell (see GIF on right side). [ citation needed ]





