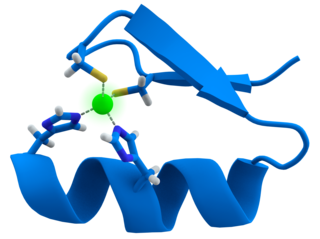
A zinc finger is a small protein structural motif that is characterized by the coordination of one or more zinc ions (Zn2+) in order to stabilize the fold. It was originally coined to describe the finger-like appearance of a hypothesized structure from the African clawed frog (Xenopus laevis) transcription factor IIIA. However, it has been found to encompass a wide variety of differing protein structures in eukaryotic cells. Xenopus laevis TFIIIA was originally demonstrated to contain zinc and require the metal for function in 1983, the first such reported zinc requirement for a gene regulatory protein followed soon thereafter by the Krüppel factor in Drosophila. It often appears as a metal-binding domain in multi-domain proteins.

DNA-binding proteins are proteins that have DNA-binding domains and thus have a specific or general affinity for single- or double-stranded DNA. Sequence-specific DNA-binding proteins generally interact with the major groove of B-DNA, because it exposes more functional groups that identify a base pair. However, there are some known minor groove DNA-binding ligands such as netropsin, distamycin, Hoechst 33258, pentamidine, DAPI and others.

Helix-turn-helix is a DNA-binding protein (DBP). The helix-turn-helix (HTH) is a major structural motif capable of binding DNA. Each monomer incorporates two α helices, joined by a short strand of amino acids, that bind to the major groove of DNA. The HTH motif occurs in many proteins that regulate gene expression. It should not be confused with the helix–loop–helix motif.

EGR-1 also known as ZNF268 or NGFI-A is a protein that in humans is encoded by the EGR1 gene.
A DNA-binding domain (DBD) is an independently folded protein domain that contains at least one structural motif that recognizes double- or single-stranded DNA. A DBD can recognize a specific DNA sequence or have a general affinity to DNA. Some DNA-binding domains may also include nucleic acids in their folded structure.

GATA-binding factor 1 or GATA-1 is the founding member of the GATA family of transcription factors. This protein is widely expressed throughout vertebrate species. In humans and mice, it is encoded by the GATA1 and Gata1 genes, respectively. These genes are located on the X chromosome in both species.
In molecular genetics, the Krüppel-like family of transcription factors (KLFs) are a set of eukaryotic C2H2 zinc finger DNA-binding proteins that regulate gene expression. This family has been expanded to also include the Sp transcription factor and related proteins, forming the Sp/KLF family.
Therapeutic gene modulation refers to the practice of altering the expression of a gene at one of various stages, with a view to alleviate some form of ailment. It differs from gene therapy in that gene modulation seeks to alter the expression of an endogenous gene whereas gene therapy concerns the introduction of a gene whose product aids the recipient directly.
In a zinc finger protein, certain sequences of amino acid residues are able to recognise and bind to an extended target-site of four or even five nucleotides When this occurs in a ZFP in which the three-nucleotide subsites are contiguous, one zinc finger interferes with the target-site of the zinc finger adjacent to it, a situation known as target-site overlap. For example, a zinc finger containing arginine at position -1 and aspartic acid at position 2 along its alpha-helix will recognise an extended sequence of four nucleotides of the sequence 5'-NNG(G/T)-3'. The hydrogen bond between Asp2 and the N4 of either a cytosine or adenine base paired to the guanine or thymine, respectively defines these two nucleotides at the 3' position, defining a sequence that overlaps into the subsite of any zinc finger that may be attached N-terminally.

Transcriptional repressor CTCF also known as 11-zinc finger protein or CCCTC-binding factor is a transcription factor that in humans is encoded by the CTCF gene. CTCF is involved in many cellular processes, including transcriptional regulation, insulator activity, V(D)J recombination and regulation of chromatin architecture.

T-cell acute lymphocytic leukemia protein 1 is a protein that in humans is encoded by the TAL1 gene.

GATA2 or GATA-binding factor 2 is a transcription factor, i.e. a nuclear protein which regulates the expression of genes. It regulates many genes that are critical for the embryonic development, self-renewal, maintenance, and functionality of blood-forming, lympathic system-forming, and other tissue-forming stem cells. GATA2 is encoded by the GATA2 gene, a gene which often suffers germline and somatic mutations which lead to a wide range of familial and sporadic diseases, respectively. The gene and its product are targets for the treatment of these diseases.

LIM domain only 2, also known as LMO2, RBTNL1, RBTN2, RHOM2, LIM Domain Only Protein 2, TTG2, and T-Cell Translocation Protein 2, is a protein which in humans is encoded by the LMO2 gene.

C-terminal-binding protein 1 also known as CtBP1 is a protein that in humans is encoded by the CTBP1 gene. CtBP1 is one of two CtBP proteins, the other protein being CtBP2.

Zinc finger protein 143 is a protein that in humans is encoded by the ZNF143 gene.

Zinc finger protein ZFPM1(FOG-1) also known as friend of GATA protein 1 is a protein that in humans is encoded by the ZFPM1 gene. It is a cofactor of the GATA1 transcription factor.

Zinc finger and BTB domain-containing protein 32 is a protein that in humans is encoded by the 1960 bp ZBTB32 gene. The 52 kDa protein is a transcriptional repressor and the gene is expressed in T and B cells upon activation, but also significantly in testis cells. It is a member of the Poxviruses and Zinc-finger (POZ) and Krüppel (POK) family of proteins, and was identified in multiple screens involving either immune cell tumorigenesis or immune cell development.

Zinc finger protein 804A is a protein that in humans is encoded by the ZNF804A gene. The human gene maps to chromosome 2 q32.1 and consists of 4 exons that code for a protein of 1210 amino acids.

Archaeal transcription factor B is a protein family of extrinsic transcription factors that guide the initiation of RNA transcription in organisms that fall under the domain of Archaea. It is homologous to eukaryotic TFIIB and, more distantly, to bacterial sigma factor. Like these proteins, it is involved in forming transcription preinitiation complexes. Its structure includes several conserved motifs which interact with DNA and other transcription factors, notably the single type of RNA polymerase that performs transcription in Archaea.

Zinc finger protein 226 is a protein that in humans is encoded by the ZNF226 gene.