
Meiosis is a special type of cell division of germ cells and apicomplexans in sexually-reproducing organisms that produces the gametes, such as sperm or egg cells. It involves two rounds of division that ultimately result in four cells with only one copy of each chromosome (haploid). Additionally, prior to the division, genetic material from the paternal and maternal copies of each chromosome is crossed over, creating new combinations of code on each chromosome. Later on, during fertilisation, the haploid cells produced by meiosis from a male and a female will fuse to create a cell with two copies of each chromosome again, the zygote.
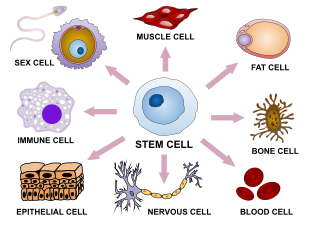
Cellular differentiation is the process in which a stem cell changes from one type to a differentiated one. Usually, the cell changes to a more specialized type. Differentiation happens multiple times during the development of a multicellular organism as it changes from a simple zygote to a complex system of tissues and cell types. Differentiation continues in adulthood as adult stem cells divide and create fully differentiated daughter cells during tissue repair and during normal cell turnover. Some differentiation occurs in response to antigen exposure. Differentiation dramatically changes a cell's size, shape, membrane potential, metabolic activity, and responsiveness to signals. These changes are largely due to highly controlled modifications in gene expression and are the study of epigenetics. With a few exceptions, cellular differentiation almost never involves a change in the DNA sequence itself. However, metabolic composition does get altered quite dramatically where stem cells are characterized by abundant metabolites with highly unsaturated structures whose levels decrease upon differentiation. Thus, different cells can have very different physical characteristics despite having the same genome.
Gametogenesis is a biological process by which diploid or haploid precursor cells undergo cell division and differentiation to form mature haploid gametes. Depending on the biological life cycle of the organism, gametogenesis occurs by meiotic division of diploid gametocytes into various gametes, or by mitosis. For example, plants produce gametes through mitosis in gametophytes. The gametophytes grow from haploid spores after sporic meiosis. The existence of a multicellular, haploid phase in the life cycle between meiosis and gametogenesis is also referred to as alternation of generations.

A germ cell is any cell that gives rise to the gametes of an organism that reproduces sexually. In many animals, the germ cells originate in the primitive streak and migrate via the gut of an embryo to the developing gonads. There, they undergo meiosis, followed by cellular differentiation into mature gametes, either eggs or sperm. Unlike animals, plants do not have germ cells designated in early development. Instead, germ cells can arise from somatic cells in the adult, such as the floral meristem of flowering plants.

Oogenesis, ovogenesis, or oögenesis is the differentiation of the ovum into a cell competent to further develop when fertilized. It is developed from the primary oocyte by maturation. Oogenesis is initiated in the embryonic stage.

An ovarian follicle is a roughly spheroid cellular aggregation set found in the ovaries. It secretes hormones that influence stages of the menstrual cycle. At the time of puberty, women have approximately 200,000 to 300,000 follicles, each with the potential to release an egg cell (ovum) at ovulation for fertilization. These eggs are developed once every menstrual cycle with around 450–500 being ovulated during a woman's reproductive lifetime.
A germ layer is a primary layer of cells that forms during embryonic development. The three germ layers in vertebrates are particularly pronounced; however, all eumetazoans produce two or three primary germ layers. Some animals, like cnidarians, produce two germ layers making them diploblastic. Other animals such as bilaterians produce a third layer between these two layers, making them triploblastic. Germ layers eventually give rise to all of an animal's tissues and organs through the process of organogenesis.

Spermatocytes are a type of male gametocyte in animals. They derive from immature germ cells called spermatogonia. They are found in the testis, in a structure known as the seminiferous tubules. There are two types of spermatocytes, primary and secondary spermatocytes. Primary and secondary spermatocytes are formed through the process of spermatocytogenesis.
An oogonium is a small diploid cell which, upon maturation, forms a primordial follicle in a female fetus or the female gametangium of certain thallophytes.
In biology, reprogramming refers to erasure and remodeling of epigenetic marks, such as DNA methylation, during mammalian development or in cell culture. Such control is also often associated with alternative covalent modifications of histones.

The inner cell mass (ICM) or embryoblast is a structure in the early development of an embryo. It is the mass of cells inside the blastocyst that will eventually give rise to the definitive structures of the fetus. The inner cell mass forms in the earliest stages of embryonic development, before implantation into the endometrium of the uterus. The ICM is entirely surrounded by the single layer of trophoblast cells of the trophectoderm.
In developmental biology, the cells that give rise to the gametes are often set aside during embryonic cleavage. During development, these cells will differentiate into primordial germ cells, migrate to the location of the gonad, and form the germline of the animal.
Gonocytes are the precursors of spermatogonia that differentiate in the testis from primordial germ cells around week 7 of embryonic development and exist up until the postnatal period, when they become spermatogonia. Despite some uses of the term to refer to the precursors of oogonia, it was generally restricted to male germ cells. Germ cells operate as vehicles of inheritance by transferring genetic and epigenetic information from one generation to the next. Male fertility is centered around continual spermatogonia which is dependent upon a high stem cell population. Thus, the function and quality of a differentiated sperm cell is dependent upon the capacity of its originating spermatogonial stem cell (SSC).

Rex1 (Zfp-42) is a known marker of pluripotency, and is usually found in undifferentiated embryonic stem cells. In addition to being a marker for pluripotency, its regulation is also critical in maintaining a pluripotent state. As the cells begin to differentiate, Rex1 is severely and abruptly downregulated.

Cell potency is a cell's ability to differentiate into other cell types. The more cell types a cell can differentiate into, the greater its potency. Potency is also described as the gene activation potential within a cell, which like a continuum, begins with totipotency to designate a cell with the most differentiation potential, pluripotency, multipotency, oligopotency, and finally unipotency.
After the blastocyst stage, once an embryo implanted in endometrium, the inner cell mass (ICM) of a fertilized embryo segregates into two layers: hypoblast and epiblast. The epiblast cells are the functional progenitors of soma and germ cells which later differentiate into three layers: definitive endoderm, mesoderm and ectoderm. Stem cells derived from epiblast are pluripotent. These cells are called epiblast-derived stem cells (EpiSCs) and have several different cellular and molecular characteristics with Embryonic Stem Cells (ESCs). Pluripotency in EpiSCs is essentially different from that of embryonic stem cells. The pluripotency of EpiSCs is primed pluripotency: primed to differentiate into specific cell lineages. Naïve pluripotent stem cells and primed pluripotent stem cells not only sustain the ability to self-renew but also maintain the capacity to differentiate. Since the cell status is primed to differentiate in EpiSCs, however, one copy of the X chromosome in XX cells in EpiSCs is silenced (XaXi). EpiSCs is unable to colonize and is not available to be used to produce chimeras. Conversely, XX cells in ESCs are both active and can produce chimera when inserted into a blastocyst. Both ESC and EpiSC induce teratoma when injected in the test animals which proves pluripotency. EpiSC display several distinctive characteristics distinct from ESCs. The cellular status of human ESCs (hESCs) is similar to primed state mouse stem cells rather than naïve state.

A spermatogonial stem cell (SSC), also known as a type A spermatogonium, is a spermatogonium that does not differentiate into a spermatocyte, a precursor of sperm cells. Instead, they continue dividing into other spermatogonia or remain dormant to maintain a reserve of spermatogonia. Type B spermatogonia, on the other hand, differentiate into spermatocytes, which in turn undergo meiosis to eventually form mature sperm cells.
The signaled by retinoic acid 8 (Stra8) gene is activated only upon stimulation by retinoic acid and expresses a cytoplasmic protein in the gonads of male and female vertebrates. This protein functions to initiate the transition between mitosis and meiosis, aiding in spermatogenesis and oogenesis. In females, its signaling begins 12.5 days after conception, is localized in the primordial germ cells of female ovaries, and ushers in the first stage of meiosis. Male expression begins postnatally and continues throughout life, matching the need of spermatogenesis compared to the limited window of oogenesis in females. Sperm of mice that had induced null mutations for Stra8 gene were able to undergo mitotic divisions, and while some sperm were able to transition into the early stages of meiosis I, but could not transition into further sub-stages of meiosis I. Errors in chromosome pairing and chromosome condensation were observed following these failures. In female mice, loss of Stra8 signaling shows failure to enter into meiosis. Both males and females are left infertile if Stra8 signaling is absent.

Primordial germ cell (PGC) migration is the process of distribution of primordial germ cells throughout the embryo during embryogenesis.
The germ cell nest forms in the ovaries during their development. The nest consists of multiple interconnected oogonia formed by incomplete cell division. The interconnected oogonia are surrounded by somatic cells called granulosa cells. Later on in development, the germ cell nests break down through invasion of granulosa cells. The result is individual oogonia surrounded by a single layer of granulosa cells. There is also a comparative germ cell nest structure in the developing spermatogonia, with interconnected intracellular cytoplasmic bridges.










