Ontogeny is the origination and development of an organism, usually from the time of fertilization of the egg to adult. The term can also be used to refer to the study of the entirety of an organism's lifespan.

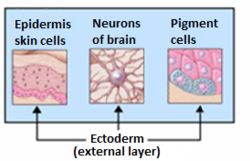
The mesoderm is the middle layer of the three germ layers that develops during gastrulation in the very early development of the embryo of most animals. The outer layer is the ectoderm, and the inner layer is the endoderm.
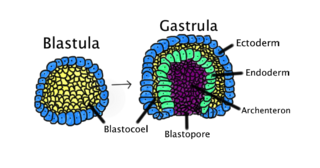
Gastrulation is the stage in the early embryonic development of most animals, during which the blastula, or in mammals the blastocyst, is reorganized into a two-layered or three-layered embryo known as the gastrula. Before gastrulation, the embryo is a continuous epithelial sheet of cells; by the end of gastrulation, the embryo has begun differentiation to establish distinct cell lineages, set up the basic axes of the body, and internalized one or more cell types including the prospective gut.
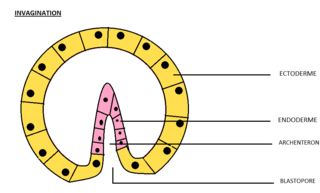
Invagination is the process of a surface folding in on itself to form a cavity, pouch or tube. In developmental biology, invagination is a mechanism that takes place during gastrulation. This mechanism or cell movement happens mostly in the vegetal pole. Invagination consists of the folding of an area of the exterior sheet of cells towards the inside of the blastula. In each organism, the complexity will be different depending on the number of cells. Invagination can be referenced as one of the steps of the establishment of the body plan. The term, originally used in embryology, has been adopted in other disciplines as well.

Neurulation refers to the folding process in vertebrate embryos, which includes the transformation of the neural plate into the neural tube. The embryo at this stage is termed the neurula.
A germ layer is a primary layer of cells that forms during embryonic development. The three germ layers in vertebrates are particularly pronounced; however, all eumetazoans produce two or three primary germ layers. Some animals, like cnidarians, produce two germ layers making them diploblastic. Other animals such as bilaterians produce a third layer between these two layers, making them triploblastic. Germ layers eventually give rise to all of an animal's tissues and organs through the process of organogenesis.
Organogenesis is the phase of embryonic development that starts at the end of gastrulation and continues until birth. During organogenesis, the three germ layers formed from gastrulation form the internal organs of the organism.

In developmental biology, animal embryonic development, also known as animal embryogenesis, is the developmental stage of an animal embryo. Embryonic development starts with the fertilization of an egg cell (ovum) by a sperm cell, (spermatozoon). Once fertilized, the ovum becomes a single diploid cell known as a zygote. The zygote undergoes mitotic divisions with no significant growth and cellular differentiation, leading to development of a multicellular embryo after passing through an organizational checkpoint during mid-embryogenesis. In mammals, the term refers chiefly to the early stages of prenatal development, whereas the terms fetus and fetal development describe later stages.

The neural plate is a key developmental structure that serves as the basis for the nervous system. Cranial to the primitive node of the embryonic primitive streak, ectodermal tissue thickens and flattens to become the neural plate. The region anterior to the primitive node can be generally referred to as the neural plate. Cells take on a columnar appearance in the process as they continue to lengthen and narrow. The ends of the neural plate, known as the neural folds, push the ends of the plate up and together, folding into the neural tube, a structure critical to brain and spinal cord development. This process as a whole is termed primary neurulation.
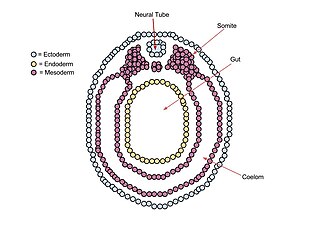
A neurula is a vertebrate embryo at the early stage of development in which neurulation occurs. The neurula stage is preceded by the gastrula stage; consequentially, neurulation is preceded by gastrulation. Neurulation marks the beginning of the process of organogenesis.
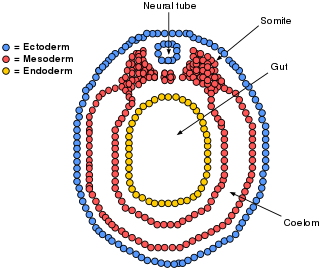
The lateral plate mesoderm is the mesoderm that is found at the periphery of the embryo. It is to the side of the paraxial mesoderm, and further to the axial mesoderm. The lateral plate mesoderm is separated from the paraxial mesoderm by a narrow region of intermediate mesoderm. The mesoderm is the middle layer of the three germ layers, between the outer ectoderm and inner endoderm.

The neural fold is a structure that arises during neurulation in the embryonic development of both birds and mammals among other organisms. This structure is associated with primary neurulation, meaning that it forms by the coming together of tissue layers, rather than a clustering, and subsequent hollowing out, of individual cells. In humans, the neural folds are responsible for the formation of the anterior end of the neural tube. The neural folds are derived from the neural plate, a preliminary structure consisting of elongated ectoderm cells. The folds give rise to neural crest cells, as well as bringing about the formation of the neural tube.

In amniote embryonic development, the epiblast is one of two distinct cell layers arising from the inner cell mass in the mammalian blastocyst, or from the blastula in reptiles and birds, the other layer is the hypoblast. It drives the embryo proper through its differentiation into the three primary germ layers, ectoderm, mesoderm and endoderm, during gastrulation. The amniotic ectoderm and extraembryonic mesoderm also originate from the epiblast.

Mesenchyme is a type of loosely organized animal embryonic connective tissue of undifferentiated cells that give rise to most tissues, such as skin, blood or bone. The interactions between mesenchyme and epithelium help to form nearly every organ in the developing embryo.

The bilaminar embryonic disc, bilaminar blastoderm or embryonic disc is the distinct two-layered structure of cells formed in an embryo. In the development of the human embryo this takes place by day eight. It is formed when the inner cell mass, also known as the embryoblast, forms a bilaminar disc of two layers, an upper layer called the epiblast and a lower layer called the hypoblast, which will eventually form into fetus. These two layers of cells are stretched between two fluid-filled cavities at either end: the primitive yolk sac and the amniotic sac.
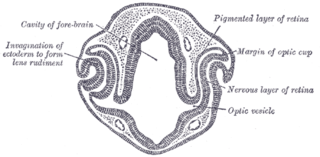
Eye formation in the human embryo begins at approximately three weeks into embryonic development and continues through the tenth week. Cells from both the mesodermal and the ectodermal tissues contribute to the formation of the eye. Specifically, the eye is derived from the neuroepithelium, surface ectoderm, and the extracellular mesenchyme which consists of both the neural crest and mesoderm.

The development of fishes is unique in some specific aspects compared to the development of other animals.

Human embryonic development or human embryogenesis is the development and formation of the human embryo. It is characterised by the processes of cell division and cellular differentiation of the embryo that occurs during the early stages of development. In biological terms, the development of the human body entails growth from a one-celled zygote to an adult human being. Fertilization occurs when the sperm cell successfully enters and fuses with an egg cell (ovum). The genetic material of the sperm and egg then combine to form the single cell zygote and the germinal stage of development commences. Embryonic development in the human, covers the first eight weeks of development; at the beginning of the ninth week the embryo is termed a fetus. The eight weeks have 23 stages.

The surface ectoderm, AKA external ectoderm, is one of the two early embryonic divisions of the ectoderm. The other early division of the ectoderm is the neuroectoderm.

In Xenopus laevis, the specification of the three germ layers occurs at the blastula stage. Great efforts have been made to determine the factors that specify the endoderm and mesoderm. On the other hand, only a few examples of genes that are required for ectoderm specification have been described in the last decade. The first molecule identified to be required for the specification of ectoderm was the ubiquitin ligase Ectodermin ; later, it was found that the deubiquitinating enzyme, FAM/USP9x, is able to overcome the effects of ubiquitination made by Ectodermin in Smad4. Two transcription factors have been proposed to control gene expression of ectodermal specific genes: POU91/Oct3/4 and FoxIe1/Xema. A new factor specific for the ectoderm, XFDL156, has shown to be essential for suppression of mesoderm differentiation from pluripotent cells.