Immunodeficiency, also known as immunocompromisation, is a state in which the immune system's ability to fight infectious diseases and cancer is compromised or entirely absent. Most cases are acquired ("secondary") due to extrinsic factors that affect the patient's immune system. Examples of these extrinsic factors include HIV infection and environmental factors, such as nutrition. Immunocompromisation may also be due to genetic diseases/flaws such as SCID.

ICF syndrome is a very rare autosomal recessive immune disorder.
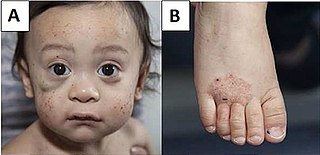
Wiskott–Aldrich syndrome (WAS) is a rare X-linked recessive disease characterized by eczema, thrombocytopenia, immune deficiency, and bloody diarrhea. It is also sometimes called the eczema-thrombocytopenia-immunodeficiency syndrome in keeping with Aldrich's original description in 1954. The WAS-related disorders of X-linked thrombocytopenia (XLT) and X-linked congenital neutropenia (XLN) may present with similar but less severe symptoms and are caused by mutations of the same gene.

X-linked agammaglobulinemia (XLA) is a rare genetic disorder discovered in 1952 that affects the body's ability to fight infection. As the form of agammaglobulinemia that is X-linked, it is much more common in males. In people with XLA, the white blood cell formation process does not generate mature B cells, which manifests as a complete or near-complete lack of proteins called gamma globulins, including antibodies, in their bloodstream. B cells are part of the immune system and normally manufacture antibodies, which defend the body from infections by sustaining a humoral immunity response. Patients with untreated XLA are prone to develop serious and even fatal infections. A mutation occurs at the Bruton's tyrosine kinase (Btk) gene that leads to a severe block in B cell development and a reduced immunoglobulin production in the serum. Btk is particularly responsible for mediating B cell development and maturation through a signaling effect on the B cell receptor BCR. Patients typically present in early childhood with recurrent infections, in particular with extracellular, encapsulated bacteria. XLA is deemed to have a relatively low incidence of disease, with an occurrence rate of approximately 1 in 200,000 live births and a frequency of about 1 in 100,000 male newborns. It has no ethnic predisposition. XLA is treated by infusion of human antibody. Treatment with pooled gamma globulin cannot restore a functional population of B cells, but it is sufficient to reduce the severity and number of infections due to the passive immunity granted by the exogenous antibodies.
Hypogammaglobulinemia is an immune system disorder in which not enough gamma globulins are produced in the blood. This results in a lower antibody count, which impairs the immune system, increasing risk of infection. Hypogammaglobulinemia may result from a variety of primary genetic immune system defects, such as common variable immunodeficiency, or it may be caused by secondary effects such as medication, blood cancer, or poor nutrition, or loss of gamma globulins in urine, as in nonselective glomerular proteinuria. Patients with hypogammaglobulinemia have reduced immune function; important considerations include avoiding use of live vaccines, and take precautionary measures when traveling to regions with endemic disease or poor sanitation such as receiving immunizations, taking antibiotics abroad, drinking only safe or boiled water, arranging appropriate medical cover in advance of travel, and ensuring continuation of any immunoglobulin infusions needed.

Immunodysregulation polyendocrinopathy enteropathy X-linked syndrome is a rare autoimmune disease. It is one of the autoimmune polyendocrine syndromes. Most often, IPEX presents with autoimmune enteropathy, dermatitis (eczema), and autoimmune endocrinopathy, but other presentations exist.
Hyperimmunoglobulinemia E syndrome (HIES), of which the autosomal dominant form is called Job's syndrome or Buckley syndrome, is a heterogeneous group of immune disorders. Job's is also very rare at about 300 cases currently in the literature.

X-linked severe combined immunodeficiency (X-SCID) is an immunodeficiency disorder in which the body produces very few T cells and NK cells.
Immune dysregulation is any proposed or confirmed breakdown or maladaptive change in molecular control of immune system processes. For example, dysregulation is a component in the pathogenesis of autoimmune diseases and some cancers. Immune system dysfunction, as seen in IPEX syndrome leads to immune dysfunction, polyendocrinopathy, enteropathy, X-linked (IPEX). IPEX typically presents during the first few months of life with diabetes mellitus, intractable diarrhea, failure to thrive, eczema, and hemolytic anemia. unrestrained or unregulated immune response.
Hyper IgM syndrome is a rare primary immune deficiency disorders characterized by low or absent levels of serum IgG, IgA, IgE and normal or increased levels of serum IgM.

Hyper IgM Syndrome Type 2 is a rare disease. Unlike other hyper-IgM syndromes, Type 2 patients identified thus far did not present with a history of opportunistic infections. One would expect opportunistic infections in any immunodeficiency syndrome. The responsible genetic lesion is in the AICDA gene found at 12p13.

The fifth type of hyper-IgM syndrome has been characterized in three patients from France and Japan. The symptoms are similar to hyper IgM syndrome type 2, but the AICDA gene is intact.
Primary immunodeficiencies are disorders in which part of the body's immune system is missing or does not function normally. To be considered a primary immunodeficiency (PID), the immune deficiency must be inborn, not caused by secondary factors such as other disease, drug treatment, or environmental exposure to toxins. Most primary immunodeficiencies are genetic disorders; the majority are diagnosed in children under the age of one, although milder forms may not be recognized until adulthood. While there are over 430 recognized inborn errors of immunity (IEIs) as of 2019, the vast majority of which are PIDs, most are very rare. About 1 in 500 people in the United States are born with a primary immunodeficiency. Immune deficiencies can result in persistent or recurring infections, auto-inflammatory disorders, tumors, and disorders of various organs. There are currently limited treatments available for these conditions; most are specific to a particular type of PID. Research is currently evaluating the use of stem cell transplants (HSCT) and experimental gene therapies as avenues for treatment in limited subsets of PIDs.

Netherton syndrome is a severe, autosomal recessive form of ichthyosis associated with mutations in the SPINK5 gene. It is named after Earl W. Netherton (1910–1985), an American dermatologist who discovered it in 1958.

IgG deficiency is a form of dysgammaglobulinemia where the proportional levels of the IgG isotype are reduced relative to other immunoglobulin isotypes.

Hyper-IgM syndrome type 3 is a form of hyper IgM syndrome characterized by mutations of the CD40 gene. In this type, Immature B cells cannot receive signal 2 from helper T cells which is necessary to mature into mature B cells.

Hyper-IgM syndrome type 4 is a form of Hyper IgM syndrome which is a defect in class switch recombination downstream of the AICDA gene that does not impair somatic hypermutation.

DOCK8 deficiency, also called DOCK8 immunodeficiency syndrome, is the autosomal recessive form of hyperimmunoglobulin E syndrome, a genetic disorder characterized by elevated immunoglobulin E levels, eosinophilia, and recurrent infections with staphylococcus and viruses. It is caused by a mutation in the DOCK8 gene.
Immunoglobulin therapy is the use of a mixture of antibodies to treat several health conditions. These conditions include primary immunodeficiency, immune thrombocytopenic purpura, chronic inflammatory demyelinating polyneuropathy, Kawasaki disease, certain cases of HIV/AIDS and measles, Guillain-Barré syndrome, and certain other infections when a more specific immunoglobulin is not available. Depending on the formulation it can be given by injection into muscle, a vein, or under the skin. The effects last a few weeks.





