Reactions
hisB-N
- D-erythro-1-(imidazol-4-yl)glycerol 3-phosphate 3-(imidazol-4-yl)-2-oxopropyl phosphate + H2O
hisB-C
- L-histidinol phosphate + H2O L-histidinol + phosphate
The hisB gene, found in the enterobacteria (such as E. coli ), in Campylobacter jejuni and in Xylella / Xanthomonas encodes a protein involved in catalysis of two step in histidine biosynthesis (the sixth and eight step), namely the bifunctional Imidazoleglycerol-phosphate dehydratase/histidinol-phosphatase. [1]
The former function (EC 4.2.1.19), found at the N-terminal, dehydrated d-erythroimidazoleglycerolphosphate to imidazoleacetolphosphate, the latter function (EC 3.1.3.15), found at the C-terminal, dephosphorylates l-histidinolphosphate producing histidinol. [2] [3] [4]
The firth step is catalysed instead by histadinolphosphate aminotransferase (encoded by hisC) [5]
The peptide is 40.5kDa and associates to form a hexamer (unless truncated) [6]
In E. coli hisB is found on the hisGDCBHAFI operon [7]
The phosphatase activity possess a substrate ambiguity and overexpression of hisB can rescue phosphoserine phosphatase (serB) knockouts. [8]
hisB-N
hisB-C
HIS3 from Saccharomyces cerevisiae is not a fused IGP dehydratase and hisidinol phosphatase, but an IGPD only (homologous to hisB-N). Whereas HIS2 is the HP (analogous to hisB-C, called hisJ in some prokaryotes).

Histidine (symbol His or H) is an α-amino acid that is used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated –NH3+ form under biological conditions), a carboxylic acid group (which is in the deprotonated –COO− form under biological conditions), and an imidazole side chain (which is partially protonated), classifying it as a positively charged amino acid at physiological pH. Initially thought essential only for infants, it has now been shown in longer-term studies to be essential for adults also. It is encoded by the codons CAU and CAC.

Pyridoxal phosphate (PLP, pyridoxal 5'-phosphate, P5P), the active form of vitamin B6, is a coenzyme in a variety of enzymatic reactions. The Enzyme commission has catalogued more than 140 PLP-dependent activities, corresponding to ~4% of all classified activities. The versatility of PLP arises from its ability to covalently bind the substrate, and then to act as an electrophilic catalyst, thereby stabilizing different types of carbanionic reaction intermediates.
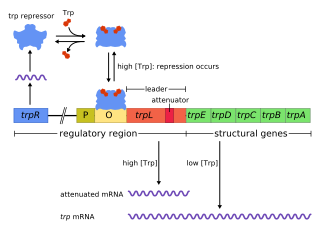
The trp operon is an operon—a group of genes that is used, or transcribed, together—that codes for the components for production of tryptophan. The trp operon is present in many bacteria, but was first characterized in Escherichia coli. The operon is regulated so that when tryptophan is present in the environment, the genes for tryptophan synthesis are not expressed. It was an important experimental system for learning about gene regulation, and is commonly used to teach gene regulation.
The gene rpoS encodes the sigma factor sigma-38, a 37.8 kD protein in Escherichia coli. Sigma factors are proteins that regulate transcription in bacteria. Sigma factors can be activated in response to different environmental conditions. rpoS is transcribed in late exponential phase, and RpoS is the primary regulator of stationary phase genes. RpoS is a central regulator of the general stress response and operates in both a retroactive and a proactive manner: it not only allows the cell to survive environmental challenges, but it also prepares the cell for subsequent stresses (cross-protection). The transcriptional regulator CsgD is central to biofilm formation, controlling the expression of the curli structural and export proteins, and the diguanylate cyclase, adrA, which indirectly activates cellulose production. The rpoS gene most likely originated in the gammaproteobacteria.

Amino acid synthesis is the set of biochemical processes by which the amino acids are produced. The substrates for these processes are various compounds in the organism's diet or growth media. Not all organisms are able to synthesize all amino acids. For example, humans can only synthesize 11 of the 20 standard amino acids, and in time of accelerated growth, histidine, can be considered an essential amino acid.
The L-arabinose operon, also called the ara or araBAD operon, is an operon required for the breakdown of the five-carbon sugar L-arabinose in Escherichia coli. The L-arabinose operon contains three structural genes: araB, araA, araD, which encode for three metabolic enzymes that are required for the metabolism of L-arabinose. AraB (ribulokinase), AraA, AraD produced by these genes catalyse conversion of L-arabinose to an intermediate of the pentose phosphate pathway, D-xylulose-5-phosphate.
In molecular genetics, a regulon is a group of genes that are regulated as a unit, generally controlled by the same regulatory gene that expresses a protein acting as a repressor or activator. This terminology is generally, although not exclusively, used in reference to prokaryotes, whose genomes are often organized into operons; the genes contained within a regulon are usually organized into more than one operon at disparate locations on the chromosome. Applied to eukaryotes, the term refers to any group of non-contiguous genes controlled by the same regulatory gene.
In enzymology, a 3-(imidazol-5-yl)lactate dehydrogenase (EC 1.1.1.111) is an enzyme that catalyzes the chemical reaction

In enzymology, a L-ribulose-5-phosphate 4-epimerase is an enzyme that catalyzes the interconversion of ribulose 5-phosphate and xylulose 5-phosphate in the oxidative phase of the Pentose phosphate pathway.

In enzymology, an imidazoleglycerol-phosphate dehydratase (EC 4.2.1.19) is an enzyme that catalyzes the chemical reaction

In enzymology, a histidinol-phosphatase (EC 3.1.3.15) is an enzyme that catalyzes the chemical reaction
In enzymology, a histidine transaminase is an enzyme that catalyzes the chemical reaction
In enzymology, a histidinol-phosphate transaminase is an enzyme that catalyzes the chemical reaction

In the field of molecular biology, a two-component regulatory system serves as a basic stimulus-response coupling mechanism to allow organisms to sense and respond to changes in many different environmental conditions. Two-component systems typically consist of a membrane-bound histidine kinase that senses a specific environmental stimulus and a corresponding response regulator that mediates the cellular response, mostly through differential expression of target genes. Although two-component signaling systems are found in all domains of life, they are most common by far in bacteria, particularly in Gram-negative and cyanobacteria; both histidine kinases and response regulators are among the largest gene families in bacteria. They are much less common in archaea and eukaryotes; although they do appear in yeasts, filamentous fungi, and slime molds, and are common in plants, two-component systems have been described as "conspicuously absent" from animals.
The gal operon is a prokaryotic operon, which encodes enzymes necessary for galactose metabolism. Repression of gene expression for this operon works via binding of repressor molecules to two operators. These repressors dimerize, creating a loop in the DNA. The loop as well as hindrance from the external operator prevent RNA polymerase from binding to the promoter, and thus prevent transcription. Additionally, since the metabolism of galactose in the cell is involved in both anabolic and catabolic pathways, a novel regulatory system using two promoters for differential repression has been identified and characterized within the context of the gal operon.
The Uptake of Hexose Phosphates (Uhp) is a protein system found in bacteria. It is a type of two-component sensory transduction pathway which helps bacteria react to their environment.

A response regulator is a protein that mediates a cell's response to changes in its environment as part of a two-component regulatory system. Response regulators are coupled to specific histidine kinases which serve as sensors of environmental changes. Response regulators and histidine kinases are two of the most common gene families in bacteria, where two-component signaling systems are very common; they also appear much more rarely in the genomes of some archaea, yeasts, filamentous fungi, and plants. Two-component systems are not found in metazoans.
Propionate kinase is an enzyme with systematic name ATP:propanoate phosphotransferase. This enzyme catalyses the following chemical reaction
D-glycero-beta-D-manno-heptose 1-phosphate adenylyltransferase is an enzyme with systematic name ATP:D-glycero-beta-D-manno-heptose 1-phosphate adenylyltransferase. This enzyme catalyses the following chemical reaction

The Phosphate (Pho) regulon is a bacterial regulatory mechanism used for the conservation and management of inorganic phosphate within the cell. It was first discovered in Escherichia coli as an operating system for the bacterial strain, and was later identified in other species. The Pho system is composed of various components including extracellular enzymes and transporters that are capable of phosphate assimilation in addition to extracting inorganic phosphate from organic sources. This is an essential process since phosphate plays an important role in cellular membranes, genetic expression, and metabolism within the cell. Under low nutrient availability, the Pho regulon helps the cell survive and thrive despite a depletion of phosphate within the environment. When this occurs, phosphate starvation-inducible (psi) genes activate other proteins that aid in the transport of inorganic phosphate.
| This gene article is a stub. You can help Wikipedia by expanding it. |