
In genetics, the phenotype is the set of observable characteristics or traits of an organism. The term covers the organism's morphology, its developmental processes, its biochemical and physiological properties, its behavior, and the products of behavior. An organism's phenotype results from two basic factors: the expression of an organism's genetic code and the influence of environmental factors. Both factors may interact, further affecting the phenotype. When two or more clearly different phenotypes exist in the same population of a species, the species is called polymorphic. A well-documented example of polymorphism is Labrador Retriever coloring; while the coat color depends on many genes, it is clearly seen in the environment as yellow, black, and brown. Richard Dawkins in 1978 and then again in his 1982 book The Extended Phenotype suggested that one can regard bird nests and other built structures such as caddisfly larva cases and beaver dams as "extended phenotypes".
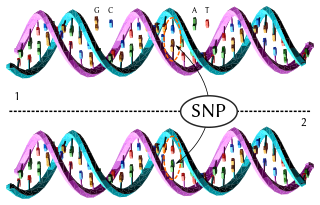
In genetics, a single-nucleotide polymorphism is a germline substitution of a single nucleotide at a specific position in the genome and is present in a sufficiently large fraction of the population. Single nucleotide substitutions with an allele frequency of less than 1% are called "single-nucleotide variants", not SNPs.

Pharmacogenomics is the study of the role of the genome in drug response. Its name reflects its combining of pharmacology and genomics. Pharmacogenomics analyzes how the genetic makeup of an individual affects their response to drugs. It deals with the influence of acquired and inherited genetic variation on drug response in patients by correlating DNA mutations with pharmacokinetic, pharmacodynamic, and/or immunogenic endpoints.
A phenome, similar to phenotype, is the set of all traits expressed by a cell, tissue, organ, organism, or species.
The candidate gene approach to conducting genetic association studies focuses on associations between genetic variation within pre-specified genes of interest, and phenotypes or disease states. This is in contrast to genome-wide association studies (GWAS), which is a hypothesis-free approach that scans the entire genome for associations between common genetic variants and traits of interest. Candidate genes are most often selected for study based on a priori knowledge of the gene's biological functional impact on the trait or disease in question. The rationale behind focusing on allelic variation in specific, biologically relevant regions of the genome is that certain alleles within a gene may directly impact the function of the gene in question and lead to variation in the phenotype or disease state being investigated. This approach often uses the case-control study design to try to answer the question, "Is one allele of a candidate gene more frequently seen in subjects with the disease than in subjects without the disease?" Candidate genes hypothesized to be associated with complex traits have generally not been replicated by subsequent GWASs or highly powered replication attempts. The failure of candidate gene studies to shed light on the specific genes underlying such traits has been ascribed to insufficient statistical power, low prior probability that scientists can correctly guess a specific allele within a specific gene that is related to a trait, poor methodological practices, and data dredging.

Gene–environment interaction is when two different genotypes respond to environmental variation in different ways. A norm of reaction is a graph that shows the relationship between genes and environmental factors when phenotypic differences are continuous. They can help illustrate GxE interactions. When the norm of reaction is not parallel, as shown in the figure below, there is a gene by environment interaction. This indicates that each genotype responds to environmental variation in a different way. Environmental variation can be physical, chemical, biological, behavior patterns or life events.
Genetic association is when one or more genotypes within a population co-occur with a phenotypic trait more often than would be expected by chance occurrence.
A tag SNP is a representative single nucleotide polymorphism (SNP) in a region of the genome with high linkage disequilibrium that represents a group of SNPs called a haplotype. It is possible to identify genetic variation and association to phenotypes without genotyping every SNP in a chromosomal region. This reduces the expense and time of mapping genome areas associated with disease, since it eliminates the need to study every individual SNP. Tag SNPs are useful in whole-genome SNP association studies in which hundreds of thousands of SNPs across the entire genome are genotyped.

In genomics, a genome-wide association study, also known as whole genome association study, is an observational study of a genome-wide set of genetic variants in different individuals to see if any variant is associated with a trait. GWA studies typically focus on associations between single-nucleotide polymorphisms (SNPs) and traits like major human diseases, but can equally be applied to any other genetic variants and any other organisms.

The Single Nucleotide Polymorphism Database (dbSNP) is a free public archive for genetic variation within and across different species developed and hosted by the National Center for Biotechnology Information (NCBI) in collaboration with the National Human Genome Research Institute (NHGRI). Although the name of the database implies a collection of one class of polymorphisms only, it in fact contains a range of molecular variation: (1) SNPs, (2) short deletion and insertion polymorphisms (indels/DIPs), (3) microsatellite markers or short tandem repeats (STRs), (4) multinucleotide polymorphisms (MNPs), (5) heterozygous sequences, and (6) named variants. The dbSNP accepts apparently neutral polymorphisms, polymorphisms corresponding to known phenotypes, and regions of no variation. It was created in September 1998 to supplement GenBank, NCBI’s collection of publicly available nucleic acid and protein sequences.
In genetics, association mapping, also known as "linkage disequilibrium mapping", is a method of mapping quantitative trait loci (QTLs) that takes advantage of historic linkage disequilibrium to link phenotypes to genotypes, uncovering genetic associations.

Exome sequencing, also known as whole exome sequencing (WES), is a genomic technique for sequencing all of the protein-coding regions of genes in a genome. It consists of two steps: the first step is to select only the subset of DNA that encodes proteins. These regions are known as exons—humans have about 180,000 exons, constituting about 1% of the human genome, or approximately 30 million base pairs. The second step is to sequence the exonic DNA using any high-throughput DNA sequencing technology.

CDKN2B-AS, also known as ANRIL is a long non-coding RNA consisting of 19 exons, spanning 126.3kb in the genome, and its spliced product is a 3834bp RNA. It is located within the p15/CDKN2B-p16/CDKN2A-p14/ARF gene cluster, in the antisense direction. Single nucleotide polymorphisms (SNPs) which alter the expression of CDKN2B-AS are associated with human healthy life expectancy, as well as with multiple diseases, including coronary artery disease, diabetes and many cancers. It binds to chromobox 7 (CBX7) within the polycomb repressive complex 1 and to SUZ12, a component of polycomb repression complex 2 and through these interactions is involved in transcriptional repression.

Interferon lambda 3 encodes the IFNL3 protein. IFNL3 was formerly named IL28B, but the Human Genome Organization Gene Nomenclature Committee renamed this gene in 2013 while assigning a name to the then newly discovered IFNL4 gene. Together with IFNL1 and IFNL2, these genes lie in a cluster on chromosomal region 19q13. IFNL3 shares ~96% amino-acid identity with IFNL2, ~80% identity with IFNL1 and ~30% identity with IFNL4.
The Pharmacogenomics Knowledgebase (PharmGKB) is a publicly available, online knowledge base responsible for the aggregation, curation, integration and dissemination of knowledge regarding the impact of human genetic variation on drug response. It is funded by the National Institutes of Health (NIH) National Institute of General Medical Sciences (NIGMS), and is a partner of the NIH Pharmacogenomics Research Network (PGRN). It has been managed at Stanford University since its inception in 2000.

The genotype-first approach is a type of strategy used in genetic epidemiological studies to associate specific genotypes to apparent clinical phenotypes of a complex disease or trait. As opposed to “phenotype-first”, the traditional strategy that has been guiding genome-wide association studies (GWAS) so far, this approach characterizes individuals first by a statistically common genotype based on molecular tests prior to clinical phenotypic classification. This method of grouping leads to patient evaluations based on a shared genetic etiology for the observed phenotypes, regardless of their suspected diagnosis. Thus, this approach can prevent initial phenotypic bias and allow for identification of genes that pose a significant contribution to the disease etiology.
A human disease modifier gene is a modifier gene that alters expression of a human gene at another locus that in turn causes a genetic disease. Whereas medical genetics has tended to distinguish between monogenic traits, governed by simple, Mendelian inheritance, and quantitative traits, with cumulative, multifactorial causes, increasing evidence suggests that human diseases exist on a continuous spectrum between the two.

In genetics, a polygenic score (PGS), also called a polygenic risk score (PRS), polygenic index (PGI), genetic risk score, or genome-wide score, is a number that summarizes the estimated effect of many genetic variants on an individual's phenotype, typically calculated as a weighted sum of trait-associated alleles. It reflects an individual's estimated genetic predisposition for a given trait and can be used as a predictor for that trait. In other words, it gives an estimate of how likely an individual is to have a given trait only based on genetics, without taking environmental factors into account. Polygenic scores are widely used in animal breeding and plant breeding due to their efficacy in improving livestock breeding and crops. In humans, polygenic scores are typically generated from genome-wide association study (GWAS) data.

Interferon lambda 4 is one of the most recently discovered human genes and the newest addition to the interferon lambda protein family. This gene encodes the IFNL4 protein, which is involved in immune response to viral infection.

Cancer pharmacogenomics is the study of how variances in the genome influences an individual’s response to different cancer drug treatments. It is a subset of the broader field of pharmacogenomics, which is the area of study aimed at understanding how genetic variants influence drug efficacy and toxicity.











