
A dye is a colored substance that chemically bonds to the substrate to which it is being applied. This distinguishes dyes from pigments which do not chemically bind to the material they color. Dye is generally applied in an aqueous solution, and may require a mordant to improve the fastness of the dye on the fiber.

Histology, also known as microscopic anatomy or microanatomy, is the branch of biology which studies the microscopic anatomy of biological tissues. Histology is the microscopic counterpart to gross anatomy, which looks at larger structures visible without a microscope. Although one may divide microscopic anatomy into organology, the study of organs, histology, the study of tissues, and cytology, the study of cells, modern usage places all of these topics under the field of histology. In medicine, histopathology is the branch of histology that includes the microscopic identification and study of diseased tissue. In the field of paleontology, the term paleohistology refers to the histology of fossil organisms.

Haematoxylin or hematoxylin, also called natural black 1 or C.I. 75290, is a compound extracted from heartwood of the logwood tree with a chemical formula of C
16H
14O
6. This naturally derived dye has been used as a histologic stain, ink and as a dye in the textile and leather industry. As a dye, haematoxylin has been called Palo de Campeche, logwood extract, bluewood and blackwood. In histology, haematoxylin staining is commonly followed (counterstained), with eosin, when paired, this staining procedure is known as H&E staining, and is one of the most commonly used combinations in histology. In addition to its use in the H&E stain, haematoxylin is also a component of the Papanicolaou stain which is widely used in the study of cytology specimens.

Eosin is the name of several fluorescent acidic compounds which bind to and form salts with basic, or eosinophilic, compounds like proteins containing amino acid residues such as arginine and lysine, and stains them dark red or pink as a result of the actions of bromine on eosin. In addition to staining proteins in the cytoplasm, it can be used to stain collagen and muscle fibers for examination under the microscope. Structures, that stain readily with eosin, are termed eosinophilic. In the field of histology, Eosin Y is the form of eosin used most often as a histologic stain.

A mordant or dye fixative is a substance used to set dyes on fabrics by forming a coordination complex with the dye, which then attaches to the fabric. It may be used for dyeing fabrics or for intensifying stains in cell or tissue preparations. Although mordants are still used, especially by small batch dyers, it has been largely displaced in industry by directs.

Staining is a technique used to enhance contrast in samples, generally at the microscopic level. Stains and dyes are frequently used in histology, in cytology, and in the medical fields of histopathology, hematology, and cytopathology that focus on the study and diagnoses of diseases at the microscopic level. Stains may be used to define biological tissues, cell populations, or organelles within individual cells.

Histopathology refers to the microscopic examination of tissue in order to study the manifestations of disease. Specifically, in clinical medicine, histopathology refers to the examination of a biopsy or surgical specimen by a pathologist, after the specimen has been processed and histological sections have been placed onto glass slides. In contrast, cytopathology examines free cells or tissue micro-fragments.
Zenker's fixative is a rapid-acting fixative for animal tissues. It is employed to prepare specimens of animal or vegetable tissues for microscopic study. It provides excellent fixation of nuclear chromatin, connective tissue fibers and some cytoplasmic features but does not preserve delicate cytoplasmic organelles such as mitochondria. Helly's fixative is preferable for traditional dye staining of mitochondria.
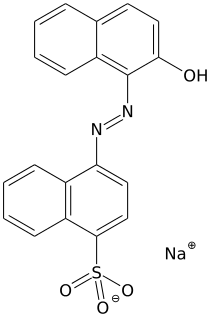
Acid dyes are anionic, soluble in water and are essentially applied from acidic bath. These dyes possess acidic groups, such as SO3H and COOH and are applied on wool, silk and nylon when ionic bond is established between protonated –NH2 group of fibre and acid group of dye. Overall wash fastness is poor although lightfastness is quite good. As dye and fibre contain opposite electrical nature, strike rate and uptake of acid dye on these fibres is faster; electrolyte at higher concentration is added to retard dye uptake and to form levelled shades. Acid generates cation on fibre and temperature helps to substitute negative part of acid with anionic dye molecules.
Trichrome staining is a histological staining method that uses two or more acid dyes in conjunction with a polyacid. Staining differentiates tissues by tinting them in contrasting colours. It increases the contrast of microscopic features in cells and tissues, which makes them easier to see when viewed through a microscope.

Reticular fibers, reticular fibres or reticulin is a type of fiber in connective tissue composed of type III collagen secreted by reticular cells. Reticular fibers crosslink to form a fine meshwork (reticulin). This network acts as a supporting mesh in soft tissues such as liver, bone marrow, and the tissues and organs of the lymphatic system.

Hematein or haematein is an oxidized derivative of haematoxylin, used in staining. Haematein should not be confused with haematin, which is a brown to black iron-containing pigment formed by decomposition of haemoglobin. In the Colour Index, haematein is called haematine.

Masson's trichrome is a three-colour staining procedure used in histology. The recipes evolved from Claude L. Pierre Masson's (1880–1959) original formulation have different specific applications, but all are suited for distinguishing cells from surrounding connective tissue.

Papanicolaou stain is a multichromatic (multicolored) cytological staining technique developed by George Papanicolaou in 1942. The Papanicolaou stain is one of the most widely used stains in cytology, where it is used to aid pathologists in making a diagnosis. Although most notable for its use in the detection of cervical cancer in the Pap test or Pap smear, it is also used to stain non-gynecological specimen preparations from a variety of bodily secretions and from small needle biopsies of organs and tissues. Papanicolaou published three formulations of this stain in 1942, 1954, and 1960.

Hematoxylin and eosin stain is one of the principal tissue stains used in histology. It is the most widely used stain in medical diagnosis and is often the gold standard. For example, when a pathologist looks at a biopsy of a suspected cancer, the histological section is likely to be stained with H&E.
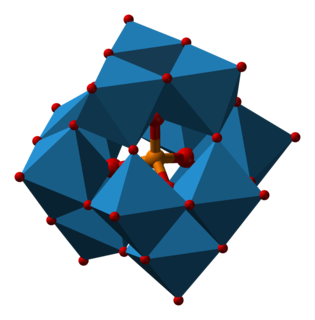
Phosphotungstic acid (PTA) or tungstophosphoric acid (TPA), is a heteropoly acid with the chemical formula H3PW12O40]. It forms hydrates H3[PW12O40]·nH2O. It is normally isolated as the n = 24 hydrate but can be desiccated to the hexahydrate. EPTA is the name of ethanolic phosphotungstic acid, its alcohol solution used in biology. It has the appearance of small, colorless-grayish or slightly yellow-green crystals, with melting point 89 °C. It is odorless and soluble in water. It is not especially toxic, but is a mild acidic irritant. The compound is known by a variety of names and acronyms.
In the fields of histology, pathology, and cell biology, fixation is the preservation of biological tissues from decay due to autolysis or putrefaction. It terminates any ongoing biochemical reactions and may also increase the treated tissues' mechanical strength or stability. Tissue fixation is a critical step in the preparation of histological sections, its broad objective being to preserve cells and tissue components and to do this in such a way as to allow for the preparation of thin, stained sections. This allows the investigation of the tissues' structure, which is determined by the shapes and sizes of such macromolecules as proteins and nucleic acids.

Brazilin is a naturally occurring red dye obtained from the wood of Paubrasilia echinata, Caesalpinia sappan, Caesalpinia violacea, and Haematoxylum brasiletto. Brazilin has been used since at least the Middle Ages to dye fabric, and has been used to make paints and inks as well. The specific color produced by the pigment depends on its manner of preparation: in an acidic solution brazilin will appear yellow, but in an alkaline preparation it will appear red. Brazilin is closely related to the blue-black dye precursor hematoxylin, having one fewer hydroxyl group. Brazilein, the active dye agent, is an oxidized form of brazilin.
Movat's stain is a pentachrome stain originally developed by Henry Zoltan Movat (1923–1995), a Hungarian-Canadian Pathologist in Toronto in 1955 to highlight the various constituents of connective tissue, especially cardiovascular tissue, by five colors in a single stained slide. In 1972, H. K. Russell, Jr. modified the technique so as to reduce the time for staining and to increase the consistency and reliability of the staining.
Bouin solution, or Bouin's solution, is a compound fixative used in histology. It was invented by French biologist Pol Bouin and is composed of picric acid, acetic acid and formaldehyde in an aqueous solution. Bouin's fluid is especially useful for fixation of gastrointestinal tract biopsies because this fixative allows crisper and better nuclear staining than 10% neutral-buffered formalin. It is not a good fixative when tissue ultrastructure must be preserved for electron microscopy. However, it is a good fixative when tissue structure with a soft and delicate texture must be preserved. The acetic acid in this fixative lyses red blood cells and dissolves small iron and calcium deposits in tissue. A variant in which the acetic acid is replaced with formic acid can be used for both fixation of tissue and decalcification. The effects of the three chemicals in Bouin solution balance each other. Formalin causes cytoplasm to become basophilic but this effect is balanced by the effect of the picric acid. This results in excellent nuclear and cytoplasmic H&E staining. The tissue hardening effect of formalin is balanced by the soft tissue fixation of picric and acetic acids. The tissue swelling effect of acetic acid is balanced by the tissue shrinking effect of picric acid.
















