
Amyloids are aggregates of proteins characterised by a fibrillar morphology of typically 7–13 nm in diameter, a β-sheet secondary structure and ability to be stained by particular dyes, such as Congo red. In the human body, amyloids have been linked to the development of various diseases. Pathogenic amyloids form when previously healthy proteins lose their normal structure and physiological functions (misfolding) and form fibrous deposits within and around cells. These protein misfolding and deposition processes disrupt the healthy function of tissues and organs.

Alpha-synuclein (aSyn) is a protein that, in humans, is encoded by the SNCA gene. Alpha-synuclein is a neuronal protein that regulates synaptic vesicle trafficking and subsequent neurotransmitter release.

Beta-secretase 2 is an enzyme that cleaves Glu-Val-Asn-Leu!Asp-Ala-Glu-Phe in the Swedish variant of Alzheimer's amyloid precursor protein. BACE2 is a close homolog of BACE1.
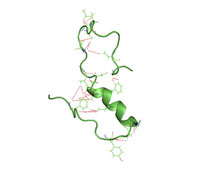
Amyloid beta denotes peptides of 36–43 amino acids that are the main component of the amyloid plaques found in the brains of people with Alzheimer's disease. The peptides derive from the amyloid-beta precursor protein (APP), which is cleaved by beta secretase and gamma secretase to yield Aβ in a cholesterol-dependent process and substrate presentation. Aβ molecules can aggregate to form flexible soluble oligomers which may exist in several forms. It is now believed that certain misfolded oligomers can induce other Aβ molecules to also take the misfolded oligomeric form, leading to a chain reaction akin to a prion infection. The oligomers are toxic to nerve cells. The other protein implicated in Alzheimer's disease, tau protein, also forms such prion-like misfolded oligomers, and there is some evidence that misfolded Aβ can induce tau to misfold.

Amyloid-beta precursor protein (APP) is an integral membrane protein expressed in many tissues and concentrated in the synapses of neurons. It functions as a cell surface receptor and has been implicated as a regulator of synapse formation, neural plasticity, antimicrobial activity, and iron export. It is coded for by the gene APP and regulated by substrate presentation. APP is best known as the precursor molecule whose proteolysis generates amyloid beta (Aβ), a polypeptide containing 37 to 49 amino acid residues, whose amyloid fibrillar form is the primary component of amyloid plaques found in the brains of Alzheimer's disease patients.

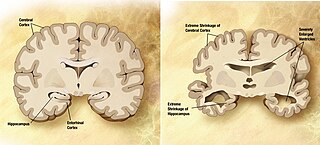
Amyloid plaques are extracellular deposits of the amyloid beta (Aβ) protein mainly in the grey matter of the brain. Degenerative neuronal elements and an abundance of microglia and astrocytes can be associated with amyloid plaques. Some plaques occur in the brain as a result of aging, but large numbers of plaques and neurofibrillary tangles are characteristic features of Alzheimer's disease. Abnormal neurites in amyloid plaques are tortuous, often swollen axons and dendrites. The neurites contain a variety of organelles and cellular debris, and many of them include characteristic paired helical filaments, the ultrastructural component of neurofibrillary tangles. The plaques are highly variable in shape and size; in tissue sections immunostained for Aβ, they comprise a log-normal size distribution curve with an average plaque area of 400-450 square micrometers (µm²). The smallest plaques, which often consist of diffuse deposits of Aβ, are particularly numerous. The apparent size of plaques is influenced by the type of stain used to detect them, and by the plane through which they are sectioned for analysis under the microscope. Plaques form when Aβ misfolds and aggregates into oligomers and longer polymers, the latter of which are characteristic of amyloid. Misfolded and aggregated Aβ is thought to be neurotoxic, especially in its oligomeric state.
A neurodegenerative disease is caused by the progressive loss of structure or function of neurons, in the process known as neurodegeneration. Such neuronal damage may ultimately involve cell death. Neurodegenerative diseases include amyotrophic lateral sclerosis, multiple sclerosis, Parkinson's disease, Alzheimer's disease, Huntington's disease, multiple system atrophy, and prion diseases. Neurodegeneration can be found in the brain at many different levels of neuronal circuitry, ranging from molecular to systemic. Because there is no known way to reverse the progressive degeneration of neurons, these diseases are considered to be incurable; however research has shown that the two major contributing factors to neurodegeneration are oxidative stress and inflammation. Biomedical research has revealed many similarities between these diseases at the subcellular level, including atypical protein assemblies and induced cell death. These similarities suggest that therapeutic advances against one neurodegenerative disease might ameliorate other diseases as well.
The biochemistry of Alzheimer's disease, the most common cause of dementia, is not yet very well understood. Alzheimer's disease (AD) has been identified as a proteopathy: a protein misfolding disease due to the accumulation of abnormally folded amyloid beta (Aβ) protein in the brain. Amyloid beta is a short peptide that is an abnormal proteolytic byproduct of the transmembrane protein amyloid-beta precursor protein (APP), whose function is unclear but thought to be involved in neuronal development. The presenilins are components of proteolytic complex involved in APP processing and degradation.

In medicine, proteinopathy, or proteopathy, protein conformational disorder, or protein misfolding disease, is a class of diseases in which certain proteins become structurally abnormal, and thereby disrupt the function of cells, tissues and organs of the body. Often the proteins fail to fold into their normal configuration; in this misfolded state, the proteins can become toxic in some way or they can lose their normal function. The proteinopathies include such diseases as Creutzfeldt–Jakob disease and other prion diseases, Alzheimer's disease, Parkinson's disease, amyloidosis, multiple system atrophy, and a wide range of other disorders. The term proteopathy was first proposed in 2000 by Lary Walker and Harry LeVine.

Insulin-degrading enzyme, also known as IDE, is an enzyme.

17-β-Hydroxysteroid dehydrogenase X (HSD10) also known as 3-hydroxyacyl-CoA dehydrogenase type-2 is a mitochondrial enzyme that in humans is encoded by the HSD17B10 gene. Several alternatively spliced transcript variants have been identified, but the full-length nature of only two transcript variants has been determined. Human HSD10 cDNA was cloned from the brain (NM_004493), and the resulting protein, a homotetramer, was first characterized as a short chain 3-hydroxyacyl-CoA dehydrogenase (SCHAD). Active sites of this enzyme can accommodate different substrates; 17β-HSD10 is involved in the oxidation of isoleucine, branched-chain fatty acids, and xenobiotics as well as the metabolism of sex hormones and neuroactive steroids.

DnaJ homolog subfamily B member 6 is a protein that in humans is encoded by the DNAJB6 gene.
Early-onset Alzheimer's disease (EOAD), also called Younger-onset Alzheimer's disease (YOAD), is Alzheimer's disease diagnosed before the age of 65. It is an uncommon form of Alzheimer's, accounting for only 5–10% of all Alzheimer's cases. About 60% have a positive family history of Alzheimer's and 13% of them are inherited in an autosomal dominant manner. Most cases of early-onset Alzheimer's share the same traits as the "late-onset" form and are not caused by known genetic mutations. Little is understood about how it starts.
Sheena Elizabeth Radford FRS FMedSci is a British biophysicist, and Astbury Professor of Biophysics in the Astbury Centre for Structural Molecular Biology, School of Molecular and Cellular Biology at the University of Leeds. Radford is the Associate Editor of the Journal of Molecular Biology.

G. Marius Clore MAE, FRSC, FRS is a British-born, Anglo-American molecular biophysicist and structural biologist. He was born in London, U.K. and is a dual U.S./U.K. Citizen. He is a Member of the National Academy of Sciences, a Fellow of the Royal Society, a NIH Distinguished Investigator, and the Chief of the Molecular and Structural Biophysics Section in the Laboratory of Chemical Physics of the National Institute of Diabetes and Digestive and Kidney Diseases at the U.S. National Institutes of Health. He is known for his foundational work in three-dimensional protein and nucleic acid structure determination by biomolecular NMR spectroscopy, for advancing experimental approaches to the study of large macromolecules and their complexes by NMR, and for developing NMR-based methods to study rare conformational states in protein-nucleic acid and protein-protein recognition. Clore's discovery of previously undetectable, functionally significant, rare transient states of macromolecules has yielded fundamental new insights into the mechanisms of important biological processes, and in particular the significance of weak interactions and the mechanisms whereby the opposing constraints of speed and specificity are optimized. Further, Clore's work opens up a new era of pharmacology and drug design as it is now possible to target structures and conformations that have been heretofore unseen.
Konrad Beyreuther is a German molecular biologist and chemist known for his work on neurodegenerative diseases.
Colin Louis MastersMD is an Australian neuropathologist who researches Alzheimer's disease and other neurodegenerative disorders. He is laureate professor of pathology at the University of Melbourne.
Ka Yee Christina Lee is Executive Vice President for Strategic Initiatives and the David Lee Shillinglaw Distinguished Service Professor in the Department of Chemistry University of Chicago. She works on membrane biophysics, including protein–lipid interactions, Alzheimer's disease and respiratory distress syndrome. She is a Fellow of the American Institute for Medical and Biological Engineering and American Physical Society.

Iseult Lynch is an Irish chemist and Professor of Enivornmental Nanoscience at the School of Geography, Earth and Environmental Sciences at the University of Birmingham. Her research focuses on the safety of nanoparticles in the environment and their interactions with biological entities.
Hilal Lashuel is an American-Yemeni neuroscientist and chemist, currently an associate professor at the EPFL. His research focuses on protein misfolding and aggregation in the pathogenesis of Alzheimer's and Parkinson's diseases.













