Related Research Articles
Matrix metalloproteinases (MMPs), also known as matrix metallopeptidases or matrixins, are metalloproteinases that are calcium-dependent zinc-containing endopeptidases; other family members are adamalysins, serralysins, and astacins. The MMPs belong to a larger family of proteases known as the metzincin superfamily.

Neural cell adhesion molecule (NCAM), also called CD56, is a homophilic binding glycoprotein expressed on the surface of neurons, glia and skeletal muscle. Although CD56 is often considered a marker of neural lineage commitment due to its discovery site, CD56 expression is also found in, among others, the hematopoietic system. Here, the expression of CD56 is mostly associated with, but not limited to, natural killer cells. CD56 has been detected on other lymphoid cells, including gamma delta (γδ) Τ cells and activated CD8+ T cells, as well as on dendritic cells. NCAM has been implicated as having a role in cell–cell adhesion, neurite outgrowth, synaptic plasticity, and learning and memory.

Interferon regulatory factor 6 also known as IRF6 is a protein that in humans is encoded by the IRF6 gene.

Synemin, also known as desmuslin, is a protein that in humans is encoded by the SYNM gene. Synemin is an intermediate filament (IF) family member. IF proteins are cytoskeletal proteins that confer resistance to mechanical stress and are encoded by a dispersed multigene family. This protein has been found to form a linkage between desmin, which is a subunit of the IF network, and the extracellular matrix, and provides an important structural support in muscle.

72 kDa type IV collagenase also known as matrix metalloproteinase-2 (MMP-2) and gelatinase A is an enzyme that in humans is encoded by the MMP2 gene. The MMP2 gene is located on chromosome 16 at position 12.2.
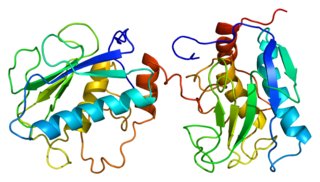
Stromelysin-1 also known as matrix metalloproteinase-3 (MMP-3) is an enzyme that in humans is encoded by the MMP3 gene. The MMP3 gene is part of a cluster of MMP genes which localize to chromosome 11q22.3. MMP-3 has an estimated molecular weight of 54 kDa.
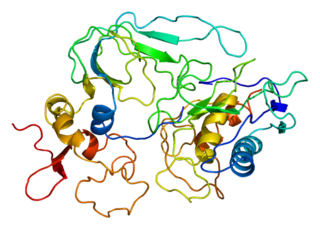
Tissue inhibitor of metalloproteinases 2 (TIMP2) is a gene and a corresponding protein. The gene is a member of the TIMP gene family. The protein is thought to be a metastasis suppressor.

Matrilysin also known as matrix metalloproteinase-7 (MMP-7), pump-1 protease (PUMP-1), or uterine metalloproteinase is an enzyme in humans that is encoded by the MMP7 gene. The enzyme has also been known as matrin, putative metalloproteinase-1, matrix metalloproteinase pump 1, PUMP-1 proteinase, PUMP, metalloproteinase pump-1, putative metalloproteinase, MMP). Human MMP-7 has a molecular weight around 30 kDa.
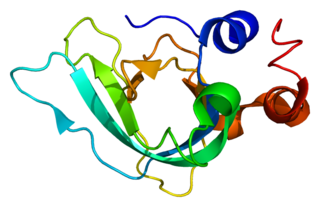
TIMP metallopeptidase inhibitor 1, also known as TIMP1, a tissue inhibitor of metalloproteinases, is a glycoprotein with a molecular weight of 28 kDa. TIMP1 is expressed from several tissues of organisms.

Metalloproteinase inhibitor 3 is a protein that in humans is encoded by the TIMP3 gene.

Matrix metalloproteinase-26 also known as matrilysin-2 and endometase is an enzyme that in humans is encoded by the MMP26 gene.

Metalloproteinase inhibitor 4 is an enzyme that in humans is encoded by the TIMP4 gene.

Interferon-induced protein with tetratricopeptide repeats 2 or IFIT2 is a protein found in humans which is encoded by the IFIT2 gene.

Dimethylaniline monooxygenase [N-oxide-forming] 4 is an enzyme that in humans is encoded by the FMO4 gene.

Olfactomedin 1, also known as noelin 1 or pancortin, is a protein that in humans is encoded by the OLFM1 gene. The name noelin stands for "neuronal olfactomedin-related endoplasmic reticulum-localized 1".
Metalloprotease inhibitors are cellular inhibitors of the Matrix metalloproteinases (MMPs). MMPs belong to a family of zinc-dependent neutral endopeptidases. These enzymes have the ability to break down connective tissue. The expression of MMPs is increased in various pathological conditions like inflammatory conditions, metabolic bone disease, to cancer invasion, metastasis and angiogenesis. Examples of diseases are periodontitis, hepatitis, glomerulonephritis, atherosclerosis, emphysema, asthma, autoimmune disorders of skin and dermal photoaging, rheumatoid arthritis, osteoarthritis, multiple sclerosis, Alzheimer's disease, chronic ulcerations, uterine involution, corneal epithelial defects, bone resorption and tumor progression and metastasis. Due to the role of MMPs in pathological conditions, inhibitors of MMPs may have therapeutic potential. Several other proteins have similar inhibitory effects, however none as effective. They might have other biological activities which have yet been fully characterised.

Aldehyde dehydrogenase 1 family, member A3, also known as ALDH1A3 or retinaldehyde dehydrogenase 3 (RALDH3), is an enzyme that in humans is encoded by the ALDH1A3 gene,
Angiogenesis is the process of forming new blood vessels from existing blood vessels, formed in vasculogenesis. It is a highly complex process involving extensive interplay between cells, soluble factors, and the extracellular matrix (ECM). Angiogenesis is critical during normal physiological development, but it also occurs in adults during inflammation, wound healing, ischemia, and in pathological conditions such as rheumatoid arthritis, hemangioma, and tumor growth. Proteolysis has been indicated as one of the first and most sustained activities involved in the formation of new blood vessels. Numerous proteases including matrix metalloproteinases (MMPs), a disintegrin and metalloproteinase domain (ADAM), a disintegrin and metalloproteinase domain with throbospondin motifs (ADAMTS), and cysteine and serine proteases are involved in angiogenesis. This article focuses on the important and diverse roles that these proteases play in the regulation of angiogenesis.

Iroquois-class homeodomain protein IRX-2, also known as Iroquois homeobox protein 2, is a protein that in humans is encoded by the IRX2 gene.

Putative sodium-coupled neutral amino acid transporter 10, also known as solute carrier family 38 member 10, is a protein that in humans is encoded by the SLC38A10 gene.
References
- ↑ Brew K, Dinakarpandian D, Nagase H (2000). "Tissue inhibitors of metalloproteinases: evolution, structure and function". Biochim Biophys Acta. 1477 (1–2): 267–83. doi:10.1016/S0167-4838(99)00279-4. PMID 10708863.
- 1 2 Rotondo JC, Bosi S, Bassi C, Ferracin M, Lanza G, Gafà R, Magri E, Selvatici R, Torresani S, Marci R, Garutti P, Negrini M, Tognon M, Martini F (April 2015). "Gene expression changes in progression of cervical neoplasia revealed by microarray analysis of cervical neoplastic keratinocytes". J Cell Physiol. 230 (4): 802–812. doi:10.1002/jcp.24808. hdl: 11392/2066612 . PMID 25205602. S2CID 24986454.
- Tissue+Inhibitor+of+Metalloproteinases at the U.S. National Library of Medicine Medical Subject Headings (MeSH)