
Immunosuppressive drugs, also known as immunosuppressive agents, immunosuppressants and antirejection medications, are drugs that inhibit or prevent activity of the immune system.

Rituximab, sold under the brand name Rituxan among others, is a monoclonal antibody medication used to treat certain autoimmune diseases and types of cancer. It is used for non-Hodgkin lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, idiopathic thrombocytopenic purpura, pemphigus vulgaris, myasthenia gravis and Epstein–Barr virus-positive mucocutaneous ulcers. It is given by slow injection into a vein. Biosimilars of Rituxan include Blitzima, Riabni, Ritemvia, Rituenza, Rixathon, Ruxience, and Truxima.

B-lymphocyte antigen CD20 or CD20 is expressed on the surface of all B-cells beginning at the pro-B phase and progressively increasing in concentration until maturity.
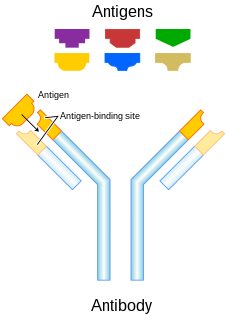
Monoclonal antibody therapy is a form of immunotherapy that uses monoclonal antibodies (mAbs) to bind monospecifically to certain cells or proteins. The objective is that this treatment will stimulate the patient's immune system to attack those cells. Alternatively, in radioimmunotherapy a radioactive dose localizes a target cell line, delivering lethal chemical doses. Antibodies have been used to bind to molecules involved in T-cell regulation to remove inhibitory pathways that block T-cell responses. This is known as immune checkpoint therapy.
Biological response modifiers (BRMs) are substances that modify immune responses. They can be both endogenous and exogenous, and they can either enhance an immune response or suppress it. Some of these substances arouse the body's response to an infection, and others can keep the response from becoming excessive. Thus they serve as immunomodulators in immunotherapy, which can be helpful in treating cancer and in treating autoimmune diseases, such as some kinds of arthritis and dermatitis. Most BRMs are biopharmaceuticals (biologics), including monoclonal antibodies, interleukin 2, interferons, and various types of colony-stimulating factors. "Immunotherapy makes use of BRMs to enhance the activity of the immune system to increase the body's natural defense mechanisms against cancer", whereas BRMs for rheumatoid arthritis aim to reduce inflammation.

Belimumab, sold under the brand name Benlysta, is a human monoclonal antibody that inhibits B-cell activating factor (BAFF), also known as B-lymphocyte stimulator (BLyS). It is approved in the United States, Canada, and the European Union to treat systemic lupus erythematosus (SLE).
Ocrelizumab, sold under the brand name Ocrevus, is a pharmaceutical drug for the treatment of multiple sclerosis (MS). It is a humanized anti-CD20 monoclonal antibody. It targets CD20 marker on B lymphocytes and hence is an immunosuppressive drug. Ocrelizumab binds to an epitope that overlaps with the epitope to which rituximab binds.
Epratuzumab is a humanized monoclonal antibody. Potential uses may be found in oncology and in treatment of inflammatory autoimmune disorders, such as systemic lupus erythematosus (SLE).
Teplizumab is a humanized anti-CD3 monoclonal antibody that is being evaluated for treatment and prevention of type 1 diabetes mellitus (T1DM) by the biopharmaceutical company Provention Bio. Teplizumab has also been evaluated for treatment of renal allograft rejection, for induction therapy in islet transplant recipients, and for psoriatic arthritis.
Otelixizumab, also known as TRX4, is a monoclonal antibody, which is being developed for the treatment of type 1 diabetes and other autoimmune diseases. The antibody is being developed by Tolerx, Inc. in collaboration with GlaxoSmithKline and is being manufactured by Abbott Laboratories.

Interferon alpha-1 is a protein that in humans is encoded by the IFNA1 gene.
Atacicept is a recombinant fusion protein designed to inhibit B cells, thereby suppressing autoimmune disease. The designer protein combines the binding site for two cytokines that regulate maturation, function, and survival of B cells - B-lymphocyte stimulator (BLyS) and A proliferation-inducing ligand (APRIL), with the constant region of immunoglobin. Atacicept blocks activation of B cells by the tumor necrosis factor receptor superfamily member 13B, a transmembrane receptor protein found predominantly on the surface of B cells. Like the monoclonal antibody belimumab, atacicept blocks the binding of BLyS, but it also blocks APRIL. Binding of these TACI ligands induces proliferation, activation, and longevity of B cells and thus their production of autoantibodies. Atacicept is thought to selectively impair mature B cells and plasma cells with less impact on progenitor cells and memory B cells.
Short Course Immune Induction Therapy or SCIIT, is a therapeutic strategy employing rapid, specific, short term-modulation of the immune system using a therapeutic agent to induce T-cell non-responsiveness, also known as operational tolerance. As an alternative strategy to immunosuppression and antigen-specific tolerance inducing therapies, the primary goal of SCIIT is to re-establish or induce peripheral immune tolerance in the context of autoimmune disease and transplant rejection through the use of biological agents. In recent years, SCIIT has received increasing attention in clinical and research settings as an alternative to immunosuppressive drugs currently used in the clinic, drugs which put the patients at risk of developing infection, cancer, and cardiovascular disease.
TOL101, is a murine-monoclonal antibody specific for the human αβ T cell receptor. In 2010 it was an Investigational New Drug under development by Tolera Therapeutics, Inc.
Blisibimod is a selective antagonist of B-cell activating factor, being developed by Anthera Pharmaceuticals as a treatment for systemic lupus erythematosus. It is currently under active investigation in clinical trials.
Tabalumab is an anti-B-cell activating factor (BAFF) human monoclonal antibody designed for the treatment of autoimmune diseases and B cell malignancies. Tabalumab was developed by Eli Lilly and Company.
Rinat Neuroscience Corporation was a privately held biotechnology company that discovered and developed antibody-based drugs including:
Tolerogenic therapy aims to induce immune tolerance where there is pathological or undesirable activation of the normal immune response. This can occur, for example, when an allogeneic transplantation patient develops an immune reaction to donor antigens, or when the body responds inappropriately to self antigens implicated in autoimmune diseases. It must provide absence of specific antibodies for exactly that antigenes.
Emactuzumab (RG-7155) is a humanized monoclonal antibody directed against CSF-1R expressed on macrophages and has demonstrated a profound antitumor effect through interference with the CSF-1/CSF-1R axis, along with a manageable safety profile in patients with diffuse-type tenosynovial giant cell tumors (d-TGCT)
Otilimab is a fully human antibody which has been developed by the biotechnology company MorphoSys. It can also be referred to as HuCAL antibody, HuCAL standing for Human Combinatorial Antibody Library and being a technology used to generate monoclonal antibodies. Otilimab is directed against the granulocyte-macrophage colony stimulating factor (GM-CSF), a monomeric glycoprotein functioning as a cytokine promoting both proliferation and activation of macrophages and neutrophils.







