Heat shock proteins (HSP) are a family of proteins produced by cells in response to exposure to stressful conditions. They were first described in relation to heat shock, but are now known to also be expressed during other stresses including exposure to cold, UV light and during wound healing or tissue remodeling. Many members of this group perform chaperone functions by stabilizing new proteins to ensure correct folding or by helping to refold proteins that were damaged by the cell stress. This increase in expression is transcriptionally regulated. The dramatic upregulation of the heat shock proteins is a key part of the heat shock response and is induced primarily by heat shock factor (HSF). HSPs are found in virtually all living organisms, from bacteria to humans.
In anatomy, a crystallin is a water-soluble structural protein found in the lens and the cornea of the eye accounting for the transparency of the structure. It has also been identified in other places such as the heart, and in aggressive breast cancer tumors. Since it has been shown that lens injury may promote nerve regeneration, crystallin has been an area of neural research. So far, it has been demonstrated that crystallin β b2 (crybb2) may be a neurite-promoting factor.

Desmin is a protein that in humans is encoded by the DES gene. Desmin is a muscle-specific, type III intermediate filament that integrates the sarcolemma, Z disk, and nuclear membrane in sarcomeres and regulates sarcomere architecture.
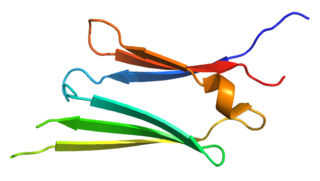
Heat shock protein 27 (Hsp27) also known as heat shock protein beta-1 (HSPB1) is a protein that in humans is encoded by the HSPB1 gene.

Heat shock protein 90kDa beta member 1 (HSP90B1), known also as endoplasmin, gp96, grp94, or ERp99, is a chaperone protein that in humans is encoded by the HSP90B1 gene.

Heat shock protein HSP 90-beta also called HSP90beta is a protein that in humans is encoded by the HSP90AB1 gene.

Heat shock protein beta-8 is a protein that in humans is encoded by the HSPB8 gene.

Heat shock 70 kDa protein 4 is a protein that in humans is encoded by the HSPA4 gene.

DnaJ homolog subfamily B member 1 is a protein that in humans is encoded by the DNAJB1 gene.

Hsc70-interacting protein also known as suppression of tumorigenicity 13 (ST13) is a protein that in humans is encoded by the ST13 gene.

Heat shock protein beta-2 is a protein that in humans is encoded by the HSPB2 gene.

Heat shock 70 kDa protein 1L is a protein that in humans is encoded by the HSPA1L gene on chromosome 6. As a member of the heat shock protein 70 (Hsp70) family and a chaperone protein, it facilitates the proper folding of newly translated and misfolded proteins, as well as stabilize or degrade mutant proteins. Its functions contribute to biological processes including signal transduction, apoptosis, protein homeostasis, and cell growth and differentiation. It has been associated with an extensive number of cancers, neurodegenerative diseases, cell senescence and aging, and Graft-versus-host disease.

LIM domain binding 3 (LDB3), also known as Z-band alternatively spliced PDZ-motif (ZASP), is a protein which in humans is encoded by the LDB3 gene. ZASP belongs to the Enigma subfamily of proteins and stabilizes the sarcomere during contraction, through interactions with actin in cardiac and skeletal muscles. Mutations in the ZASP gene has been associated with several muscular diseases.

Heat shock protein beta-6 (HSPB6) is a protein that in humans is encoded by the HSPB6 gene.

Alpha-crystallin A chain is a protein that in humans is encoded by the CRYAA gene.
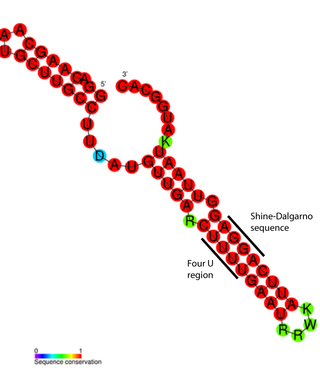
FourU thermometers are a class of non-coding RNA thermometers found in Salmonella. They are named 'FourU' due to the four highly conserved uridine nucleotides found directly opposite the Shine-Dalgarno sequence on hairpin II (pictured). RNA thermometers such as FourU control regulation of temperature via heat shock proteins in many prokaryotes. FourU thermometers are relatively small RNA molecules, only 57 nucleotides in length, and have a simple two-hairpin structure.
The heat shock protein Hsp20 family, also known as small heat shock proteins (sHSPs), is a family of heat shock proteins.

Protein moonlighting is a phenomenon by which a protein can perform more than one function. Ancestral moonlighting proteins originally possessed a single function but through evolution, acquired additional functions. Many proteins that moonlight are enzymes; others are receptors, ion channels or chaperones. The most common primary function of moonlighting proteins is enzymatic catalysis, but these enzymes have acquired secondary non-enzymatic roles. Some examples of functions of moonlighting proteins secondary to catalysis include signal transduction, transcriptional regulation, apoptosis, motility, and structural.

Heat shock protein beta-3 (HspB3) also known as heat shock 27kDa protein 3 is a protein that in humans is encoded by the HSPB3 gene.

Heat Shock Protein Family B (small) member 7 (HSPB7) in humans is a protein encoded by a gene of the same name with four exons that is located on chromosome 1p36.13.,. HSPB7 contains 170 amino acids and has a molecular weight of 18,611Da. HSPB7 is a member of human small heat shock protein (HSPB) family, which contains eleven family members of chaperone proteins. HSPB7 and its gene pair SRARP are located 5 kb apart on the opposite strands of chromosome 1p36.13.





















