
Cyclins are proteins that control the progression of a cell through the cell cycle by activating cyclin-dependent kinases (CDK). [1]

Cyclins are proteins that control the progression of a cell through the cell cycle by activating cyclin-dependent kinases (CDK). [1]
Cyclins were originally discovered by R. Timothy Hunt in 1982 while studying the cell cycle of sea urchins. [2] [3]
In an interview for "The Life Scientific" (aired on 13/12/2011) hosted by Jim Al-Khalili, R. Timothy Hunt explained that the name "cyclin" was originally named after his hobby cycling. It was only after the naming did its importance in the cell cycle become apparent. As it was appropriate the name stuck. [4] R. Timothy Hunt: "By the way, the name cyclin, which I coined, was really a joke, it's because I liked cycling so much at the time, but they did come and go in the cell..." [4]

Cyclins were originally named because their concentration varies in a cyclical fashion during the cell cycle. (Note that the cyclins are now classified according to their conserved cyclin box structure, and not all these cyclins alter in level through the cell cycle. [5] ) The oscillations of the cyclins, namely fluctuations in cyclin gene expression and destruction by the ubiquitin mediated proteasome pathway, induce oscillations in Cdk activity to drive the cell cycle. A cyclin forms a complex with Cdk, which begins to activate but the complete activation requires phosphorylation, as well. Complex formation results in activation of the Cdk active site. Cyclins themselves have no enzymatic activity but have binding sites for some substrates and target the Cdks to specific subcellular locations. [5]
Cyclins, when bound with the dependent kinases, such as the p34/cdc2/cdk1 protein, form the maturation-promoting factor. MPFs activate other proteins through phosphorylation. These phosphorylated proteins, in turn, are responsible for specific events during cell division such as microtubule formation and chromatin remodeling. Cyclins can be divided into four classes based on their behaviour in the cell cycle of vertebrate somatic cells and yeast cells: G1 cyclins, G1/S cyclins, S cyclins, and M cyclins. This division is useful when talking about most cell cycles, but it is not universal as some cyclins have different functions or timing in different cell types.
G1/S Cyclins rise in late G1 and fall in early S phase. The Cdk- G1/S cyclin complex begins to induce the initial processes of DNA replication, primarily by arresting systems that prevent S phase Cdk activity in G1. The cyclins also promote other activities to progress the cell cycle, such as centrosome duplication in vertebrates or spindle pole body in yeast. The rise in presence of G1/S cyclins is paralleled by a rise in S cyclins.
G1 cyclins do not behave like the other cyclins, in that the concentrations increase gradually (with no oscillation), throughout the cell cycle based on cell growth and the external growth-regulatory signals. The presence of G cyclins coordinate cell growth with the entry to a new cell cycle.
S cyclins bind to Cdk and the complex directly induces DNA replication. The levels of S cyclins remain high, not only throughout S phase, but through G2 and early mitosis as well to promote early events in mitosis.
M cyclin concentrations rise as the cell begins to enter mitosis and the concentrations peak at metaphase. Cell changes in the cell cycle like the assembly of mitotic spindles and alignment of sister-chromatids along the spindles are induced by M cyclin- Cdk complexes. The destruction of M cyclins during metaphase and anaphase, after the Spindle Assembly Checkpoint is satisfied, causes the exit of mitosis and cytokinesis. [6] Expression of cyclins detected immunocytochemically in individual cells in relation to cellular DNA content (cell cycle phase), [7] or in relation to initiation and termination of DNA replication during S-phase, can be measured by flow cytometry. [8]
Kaposi sarcoma herpesvirus (KSHV) encodes a D-type cyclin (ORF72) that binds CDK6 and is likely to contribute to KSHV-related cancers. [9]
Cyclins are generally very different from each other in primary structure, or amino acid sequence. However, all members of the cyclin family are similar in 100 amino acids that make up the cyclin box. Cyclins contain two domains of a similar all-α fold, the first located at the N-terminus and the second at the C-terminus. All cyclins are believed to contain a similar tertiary structure of two compact domains of 5 α helices. The first of which is the conserved cyclin box, outside of which cyclins are divergent. For example, the amino-terminal regions of S and M cyclins contain short destruction-box motifs that target these proteins for proteolysis in mitosis.
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
There are several different cyclins that are active in different parts of the cell cycle and that cause the Cdk to phosphorylate different substrates. There are also several "orphan" cyclins for which no Cdk partner has been identified. For example, cyclin F is an orphan cyclin that is essential for G2/M transition. [12] [13] A study in C. elegans revealed the specific roles of mitotic cyclins. [14] [15] Notably, recent studies have shown that cyclin A creates a cellular environment that promotes microtubule detachment from kinetochores in prometaphase to ensure efficient error correction and faithful chromosome segregation. Cells must separate their chromosomes precisely, an event that relies on the bi-oriented attachment of chromosomes to spindle microtubules through specialized structures called kinetochores. In the early phases of division, there are numerous errors in how kinetochores bind to spindle microtubules. The unstable attachments promote the correction of errors by causing a constant detachment, realignment and reattachment of microtubules from kinetochores in the cells as they try to find the correct attachment. Protein cyclin A governs this process by keeping the process going until the errors are eliminated. In normal cells, persistent cyclin A expression prevents the stabilization of microtubules bound to kinetochores even in cells with aligned chromosomes. As levels of cyclin A decline, microtubule attachments become stable, allowing the chromosomes to be divided correctly as cell division proceeds. In contrast, in cyclin A-deficient cells, microtubule attachments are prematurely stabilized. Consequently, these cells may fail to correct errors, leading to higher rates of chromosome mis-segregation. [16]
There are two main groups of cyclins:
The specific cyclin subtypes along with their corresponding CDK (in brackets) are:
| Species | G1 | G1/S | S | M |
|---|---|---|---|---|
| S. cerevisiae | Cln3 (Cdk1) | Cln 1,2 (Cdk1) | Clb 5,6 (Cdk1) | Clb 1,2,3,4 (Cdk 1) |
| S. pombe | Puc1? (Cdc2) | Puc1, Cig1? (Cdc2) | Cig2, Cig1? (Cdc2) | Cdc13 (Cdc2) |
| D. melanogaster | cyclin D (Cdk4) | cyclin E (Cdk2) | cyclin E, A (Cdk2,1) | cyclin A, B, B3 (Cdk1) |
| X. laevis | either not known or not present | cyclin E (Cdk2) | cyclin E, A (Cdk2,1) | cyclin A, B, B3 (Cdk1) |
| H. sapiens | cyclin D 1,2,3 (Cdk4, Cdk6) | cyclin E (Cdk2) | cyclin A (Cdk2, Cdk1) | cyclin B (Cdk1) |
| family | members |
|---|---|
| A | CCNA1, CCNA2 |
| B | CCNB1, CCNB2, CCNB3 |
| C | CCNC |
| D | CCND1, CCND2, CCND3 |
| E | CCNE1, CCNE2 |
| F | CCNF |
| G | CCNG1, CCNG2 |
| H | CCNH |
| I | CCNI, CCNI2 |
| J | CCNJ, CCNJL |
| K | CCNK |
| L | CCNL1, CCNL2 |
| O | CCNO |
| P | CCNP |
| T | CCNT1, CCNT2 |
| Y | CCNY, CCNYL1, CCNYL2, CCNYL3 |
In addition, the following human protein contains a cyclin domain:
Leland H. Hartwell, R. Timothy Hunt, and Paul M. Nurse won the 2001 Nobel Prize in Physiology or Medicine for their discovery of cyclin and cyclin-dependent kinase. [17]

The cell cycle, or cell-division cycle, is the series of events that take place in a cell that causes it to divide into two daughter cells. These events include the duplication of its DNA and some of its organelles, and subsequently the partitioning of its cytoplasm, chromosomes and other components into two daughter cells in a process called cell division.
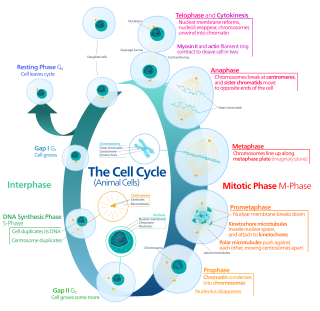
Mitosis is a part of the cell cycle in which replicated chromosomes are separated into two new nuclei. Cell division by mitosis is an equational division which gives rise to genetically identical cells in which the total number of chromosomes is maintained. Mitosis is preceded by the S phase of interphase and is followed by telophase and cytokinesis, which divide the cytoplasm, organelles, and cell membrane of one cell into two new cells containing roughly equal shares of these cellular components. The different stages of mitosis altogether define the mitotic phase of a cell cycle—the division of the mother cell into two daughter cells genetically identical to each other.

Cell division is the process by which a parent cell divides into two daughter cells. Cell division usually occurs as part of a larger cell cycle in which the cell grows and replicates its chromosome(s) before dividing. In eukaryotes, there are two distinct types of cell division: a vegetative division (mitosis), producing daughter cells genetically identical to the parent cell, and a cell division that produces haploid gametes for sexual reproduction (meiosis), reducing the number of chromosomes from two of each type in the diploid parent cell to one of each type in the daughter cells. Mitosis is a part of the cell cycle, in which, replicated chromosomes are separated into two new nuclei. Cell division gives rise to genetically identical cells in which the total number of chromosomes is maintained. In general, mitosis is preceded by the S stage of interphase and is followed by telophase and cytokinesis; which divides the cytoplasm, organelles, and cell membrane of one cell into two new cells containing roughly equal shares of these cellular components. The different stages of mitosis all together define the M phase of an animal cell cycle—the division of the mother cell into two genetically identical daughter cells. To ensure proper progression through the cell cycle, DNA damage is detected and repaired at various checkpoints throughout the cycle. These checkpoints can halt progression through the cell cycle by inhibiting certain cyclin-CDK complexes. Meiosis undergoes two divisions resulting in four haploid daughter cells. Homologous chromosomes are separated in the first division of meiosis, such that each daughter cell has one copy of each chromosome. These chromosomes have already been replicated and have two sister chromatids which are then separated during the second division of meiosis. Both of these cell division cycles are used in the process of sexual reproduction at some point in their life cycle. Both are believed to be present in the last eukaryotic common ancestor.
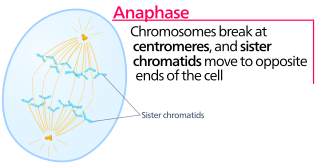
Anaphase is the stage of mitosis after the process of metaphase, when replicated chromosomes are split and the newly-copied chromosomes are moved to opposite poles of the cell. Chromosomes also reach their overall maximum condensation in late anaphase, to help chromosome segregation and the re-formation of the nucleus.

In cell biology, the spindle apparatus is the cytoskeletal structure of eukaryotic cells that forms during cell division to separate sister chromatids between daughter cells. It is referred to as the mitotic spindle during mitosis, a process that produces genetically identical daughter cells, or the meiotic spindle during meiosis, a process that produces gametes with half the number of chromosomes of the parent cell.

Telophase is the final stage in both meiosis and mitosis in a eukaryotic cell. During telophase, the effects of prophase and prometaphase are reversed. As chromosomes reach the cell poles, a nuclear envelope is re-assembled around each set of chromatids, the nucleoli reappear, and chromosomes begin to decondense back into the expanded chromatin that is present during interphase. The mitotic spindle is disassembled and remaining spindle microtubules are depolymerized. Telophase accounts for approximately 2% of the cell cycle's duration.
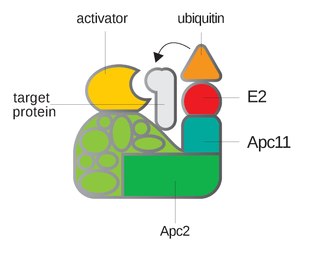
Anaphase-promoting complex is an E3 ubiquitin ligase that marks target cell cycle proteins for degradation by the 26S proteasome. The APC/C is a large complex of 11–13 subunit proteins, including a cullin (Apc2) and RING (Apc11) subunit much like SCF. Other parts of the APC/C have unknown functions but are highly conserved.

The spindle checkpoint, also known as the metaphase-to-anaphase transition, the spindle assembly checkpoint (SAC), the metaphase checkpoint, or the mitotic checkpoint, is a cell cycle checkpoint during metaphase of mitosis or meiosis that prevents the separation of the duplicated chromosomes (anaphase) until each chromosome is properly attached to the spindle. To achieve proper segregation, the two kinetochores on the sister chromatids must be attached to opposite spindle poles. Only this pattern of attachment will ensure that each daughter cell receives one copy of the chromosome. The defining biochemical feature of this checkpoint is the stimulation of the anaphase-promoting complex by M-phase cyclin-CDK complexes, which in turn causes the proteolytic destruction of cyclins and proteins that hold the sister chromatids together.

A kinetochore is a disc-shaped protein structure associated with duplicated chromatids in eukaryotic cells where the spindle fibers attach during cell division to pull sister chromatids apart. The kinetochore assembles on the centromere and links the chromosome to microtubule polymers from the mitotic spindle during mitosis and meiosis. The term kinetochore was first used in a footnote in a 1934 Cytology book by Lester W. Sharp and commonly accepted in 1936. Sharp's footnote reads: "The convenient term kinetochore has been suggested to the author by J. A. Moore", likely referring to John Alexander Moore who had joined Columbia University as a freshman in 1932.

G2 phase, Gap 2 phase, or Growth 2 phase, is the third subphase of interphase in the cell cycle directly preceding mitosis. It follows the successful completion of S phase, during which the cell’s DNA is replicated. G2 phase ends with the onset of prophase, the first phase of mitosis in which the cell’s chromatin condenses into chromosomes.
A spindle poison, also known as a spindle toxin, is a poison that disrupts cell division by affecting the protein threads that connect the centromere regions of chromosomes, known as spindles. Spindle poisons effectively cease the production of new cells by interrupting the mitosis phase of cell division at the spindle assembly checkpoint (SAC). However, as numerous and varied as they are, spindle poisons are not yet 100% effective at ending the formation of tumors (neoplasms). Although not 100% effective, substantive therapeutic efficacy has been found in these types of chemotherapeutic treatments. The mitotic spindle is composed of microtubules that aid, along with regulatory proteins, each other in the activity of appropriately segregating replicated chromosomes. Certain compounds affecting the mitotic spindle have proven highly effective against solid tumors and hematological malignancies.

Cell cycle checkpoints are control mechanisms in the eukaryotic cell cycle which ensure its proper progression. Each checkpoint serves as a potential termination point along the cell cycle, during which the conditions of the cell are assessed, with progression through the various phases of the cell cycle occurring only when favorable conditions are met. There are many checkpoints in the cell cycle, but the three major ones are: the G1 checkpoint, also known as the Start or restriction checkpoint or Major Checkpoint; the G2/M checkpoint; and the metaphase-to-anaphase transition, also known as the spindle checkpoint. Progression through these checkpoints is largely determined by the activation of cyclin-dependent kinases by regulatory protein subunits called cyclins, different forms of which are produced at each stage of the cell cycle to control the specific events that occur therein.
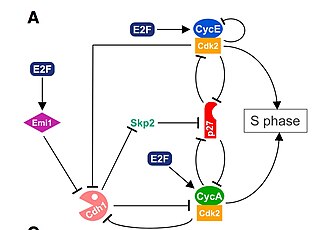
The G1/S transition is a stage in the cell cycle at the boundary between the G1 phase, in which the cell grows, and the S phase, during which DNA is replicated. It is governed by cell cycle checkpoints to ensure cell cycle integrity and the subsequent S phase can pause in response to improperly or partially replicated DNA. During this transition the cell makes decisions to become quiescent, differentiate, make DNA repairs, or proliferate based on environmental cues and molecular signaling inputs. The G1/S transition occurs late in G1 and the absence or improper application of this highly regulated checkpoint can lead to cellular transformation and disease states such as cancer.
Cyclin A is a member of the cyclin family, a group of proteins that function in regulating progression through the cell cycle. The stages that a cell passes through that culminate in its division and replication are collectively known as the cell cycle Since the successful division and replication of a cell is essential for its survival, the cell cycle is tightly regulated by several components to ensure the efficient and error-free progression through the cell cycle. One such regulatory component is cyclin A which plays a role in the regulation of two different cell cycle stages.
Cell synchronization is a process by which cells in a culture at different stages of the cell cycle are brought to the same phase. Cell synchrony is a vital process in the study of cells progressing through the cell cycle as it allows population-wide data to be collected rather than relying solely on single-cell experiments. The types of synchronization are broadly categorized into two groups; physical fractionization and chemical blockade.

The cell division cycle protein 20 homolog is an essential regulator of cell division that is encoded by the CDC20 gene in humans. To the best of current knowledge its most important function is to activate the anaphase promoting complex (APC/C), a large 11-13 subunit complex that initiates chromatid separation and entrance into anaphase. The APC/CCdc20 protein complex has two main downstream targets. Firstly, it targets securin for destruction, enabling the eventual destruction of cohesin and thus sister chromatid separation. It also targets S and M-phase (S/M) cyclins for destruction, which inactivates S/M cyclin-dependent kinases (Cdks) and allows the cell to exit from mitosis. A closely related protein, Cdc20homologue-1 (Cdh1) plays a complementary role in the cell cycle.
A series of biochemical switches control transitions between and within the various phases of the cell cycle. The cell cycle is a series of complex, ordered, sequential events that control how a single cell divides into two cells, and involves several different phases. The phases include the G1 and G2 phases, DNA replication or S phase, and the actual process of cell division, mitosis or M phase. During the M phase, the chromosomes separate and cytokinesis occurs.

Centrosomes are the major microtubule organizing centers (MTOC) in mammalian cells. Failure of centrosome regulation can cause mistakes in chromosome segregation and is associated with aneuploidy. A centrosome is composed of two orthogonal cylindrical protein assemblies, called centrioles, which are surrounded by a protein dense amorphous cloud of pericentriolar material (PCM). The PCM is essential for nucleation and organization of microtubules. The centrosome cycle is important to ensure that daughter cells receive a centrosome after cell division. As the cell cycle progresses, the centrosome undergoes a series of morphological and functional changes. Initiation of the centrosome cycle occurs early in the cell cycle in order to have two centrosomes by the time mitosis occurs.
Mitotic exit is an important transition point that signifies the end of mitosis and the onset of new G1 phase for a cell, and the cell needs to rely on specific control mechanisms to ensure that once it exits mitosis, it never returns to mitosis until it has gone through G1, S, and G2 phases and passed all the necessary checkpoints. Many factors including cyclins, cyclin-dependent kinases (CDKs), ubiquitin ligases, inhibitors of cyclin-dependent kinases, and reversible phosphorylations regulate mitotic exit to ensure that cell cycle events occur in correct order with fewest errors. The end of mitosis is characterized by spindle breakdown, shortened kinetochore microtubules, and pronounced outgrowth of astral (non-kinetochore) microtubules. For a normal eukaryotic cell, mitotic exit is irreversible.
Induced cell cycle arrest is the use of a chemical or genetic manipulation to artificially halt progression through the cell cycle. Cellular processes like genome duplication and cell division stop. It can be temporary or permanent. It is an artificial activation of naturally occurring cell cycle checkpoints, induced by exogenous stimuli controlled by an experimenter.