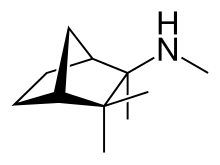
Quetiapine, sold under the brand name Seroquel among others, is an atypical antipsychotic medication used for the treatment of schizophrenia, bipolar disorder, and major depressive disorder. Despite being widely used as a sleep aid due to its sedating effect, the benefits of such use do not appear to generally outweigh the side effects. It is taken orally.

Bupropion, sold under the brand name Wellbutrin among others, is an atypical antidepressant primarily used to treat major depressive disorder and to support smoking cessation. It is also popular as an add-on medication in the cases of "incomplete response" to the first-line selective serotonin reuptake inhibitor (SSRI) antidepressant. Bupropion has several features that distinguish it from other antidepressants: it does not usually cause sexual dysfunction; it is not associated with weight gain and sleepiness, and it is more effective than SSRIs at improving symptoms of hypersomnia and fatigue. Bupropion, particularly the immediate release formulation, carries a higher risk of seizure than many other antidepressants, hence caution is recommended in patients with a history of seizure disorder.

Doxepin is a medication belonging to the tricyclic antidepressant (TCA) class of drugs used to treat major depressive disorder, anxiety disorders, chronic hives, and insomnia. For hives it is a less preferred alternative to antihistamines. It has a mild to moderate benefit for sleeping problems. It is used as a cream for itchiness due to atopic dermatitis or lichen simplex chronicus.

Viloxazine, sold under the brand name Qelbree and formerly as Vivalan among others, is a selective norepinephrine reuptake inhibitor medication which is used in the treatment of attention deficit hyperactivity disorder (ADHD) in children and adults. It was marketed for almost 30 years as an antidepressant for the treatment of depression before being discontinued and subsequently repurposed as a treatment for ADHD. Viloxazine is taken by mouth. It was used as an antidepressant in an immediate-release form and is used in ADHD in an extended-release form.

Amisulpride is an antiemetic and antipsychotic medication used at lower doses intravenously to prevent and treat postoperative nausea and vomiting; and at higher doses by mouth to treat schizophrenia and acute psychotic episodes. It is sold under the brand names Barhemsys and Solian, Socian, Deniban and others. At very low doses it is also used to treat dysthymia.

Vandetanib, sold under the brand name Caprelsa, is an anti-cancer medication that is used for the treatment of certain tumours of the thyroid gland. It acts as a kinase inhibitor of a number of cell receptors, mainly the vascular endothelial growth factor receptor (VEGFR), the epidermal growth factor receptor (EGFR), and the RET-tyrosine kinase. The drug was developed by AstraZeneca who later sold the rights to Sanofi in 2015.
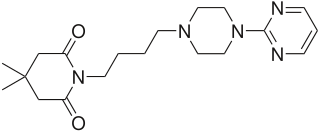
Gepirone, sold under the brand name Exxua, is a medication used for the treatment of major depressive disorder. It is taken orally.

Esketamine, also known as (S)-ketamine or S(+)-ketamine, is the S(+) enantiomer of ketamine. It is a dissociative hallucinogen drug used as a general anesthetic and as an antidepressant for treatment of depression. It is sold under the brand names Spravato, Ketanest, among others. Esketamine is the active enantiomer of ketamine in terms of NMDA receptor antagonism and is more potent than racemic ketamine.
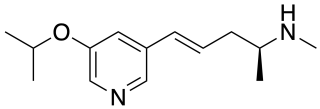
Ispronicline is an experimental drug which acts as a partial agonist at neural nicotinic acetylcholine receptors. It progressed to phase II clinical trials for the treatment of dementia and Alzheimer's disease, but is no longer under development.

Remacemide is a drug which acts as a low-affinity NMDA antagonist with sodium channel blocking properties. It has been studied for the treatment of acute ischemic stroke, epilepsy, Huntington's disease, and Parkinson's disease.

Bradanicline is a drug which was being developed by Targacept that acts as a partial agonist at the α7 subtype of the neural nicotinic acetylcholine receptors. It showed cognitive enhancing effects in animal studies, and was being developed through a collaboration between Targacept and AstraZeneca as a potential treatment for schizophrenia and attention deficit disorder. Phase I clinical trials were completed successfully, and it was in phase II trials.

TC-2216 is a drug developed by Targacept which acts as a partial agonist at neural nicotinic acetylcholine receptors and was researched for the treatment of anxiety and depression. It was unsuccessful as a therapeutic but is still used in pharmacological research as an alpha4beta2-selective antagonist.

Vortioxetine, sold under the brand names Trintellix and Brintellix among others, is a medication used to treat major depressive disorder. Its effectiveness is viewed as similar to that of other antidepressants. It is taken by mouth.
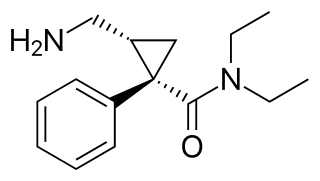
Levomilnacipran is an antidepressant which was approved in the United States in 2013 for the treatment of major depressive disorder (MDD) in adults. It is the levorotatory enantiomer of milnacipran, and has similar effects and pharmacology, acting as a serotonin–norepinephrine reuptake inhibitor (SNRI).

Zibotentan is an experimental anti-cancer drug candidate in development by AstraZeneca. It is an endothelin receptor antagonist.
Brodalumab, sold under the brand name Siliq in the US and Kyntheum in the EU, is a human monoclonal antibody designed for the treatment of inflammatory diseases.

Buprenorphine/samidorphan is a combination formulation of buprenorphine and samidorphan which is under development as an add on to antidepressants in treatment-resistant depression (TRD).

Brilaroxazine, also known as oxaripiprazole, is an investigational atypical antipsychotic which is under development by Reviva Pharmaceuticals for the treatment of neuropsychiatric and inflammatory disorders. It has currently completed the first of two phase III clinical trials for schizophrenia. Reviva Pharmaceuticals also intends to investigate brilaroxazine for the treatment of bipolar disorder, major depressive disorder, attention deficit hyperactivity disorder (ADD/ADHD), psychosis/agitation associated with Alzheimer's disease, Parkinson's disease psychosis, as well as the inflammatory disorders pulmonary arterial hypertension (PAH), idiopathic pulmonary fibrosis (IPF), and psoriasis. The FDA granted brilaroxazine orphan drug designation for the treatment of PAH and IPF.

Tezepelumab, sold under the brand name Tezspire, is a human monoclonal antibody used for the treatment of asthma. Tezepelumab blocks thymic stromal lymphopoietin (TSLP), an epithelial cytokine that has been suggested to be critical in the initiation and persistence of airway inflammation.

Dextromethorphan/bupropion (DXM/BUP), sold under the brand name Auvelity, is a combination medication for the treatment of major depressive disorder (MDD). Its active components are dextromethorphan (DXM) and bupropion. Patients who stayed on the medication had an average of 11% greater reduction in depressive symptoms than placebo in an FDA approval trial. It is taken as a tablet by mouth.