Janus kinase (JAK) is a family of intracellular, non-receptor tyrosine kinases that transduce cytokine-mediated signals via the JAK-STAT pathway. They were initially named "just another kinase" 1 and 2, but were ultimately published as "Janus kinase". The name is taken from the two-faced Roman god of beginnings, endings and duality, Janus, because the JAKs possess two near-identical phosphate-transferring domains. One domain exhibits the kinase activity, while the other negatively regulates the kinase activity of the first.
Primary myelofibrosis (PMF) is a rare bone marrow blood cancer. It is classified by the World Health Organization (WHO) as a type of myeloproliferative neoplasm, a group of cancers in which there is activation and growth of mutated cells in the bone marrow. This is most often associated with a somatic mutation in the JAK2, CALR, or MPL genes. In PMF, the bony aspects of bone marrow are remodeled in a process called osteosclerosis; in addition, fibroblast secrete collagen and reticulin proteins that are collectively referred to as (fibrosis). These two pathological processes compromise the normal function of bone marrow resulting in decreased production of blood cells such as erythrocytes, granulocytes and megakaryocytes, the latter cells responsible for the production of platelets.

Lestaurtinib is a tyrosine kinase inhibitor structurally related to staurosporine. This semisynthetic derivative of the indolocarbazole K252a was investigated by Cephalon as a treatment for various types of cancer. It is an inhibitor of the kinases fms-like tyrosine kinase 3 (FLT3), Janus kinase 2 (JAK2), tropomyosin receptor kinase (trk) A (TrkA), TrkB and TrkC.
A Janus kinase inhibitor, also known as JAK inhibitor or jakinib, is a type of immune modulating medication, which inhibits the activity of one or more of the Janus kinase family of enzymes, thereby interfering with the JAK-STAT signaling pathway in lymphocytes.
Givinostat, sold under the brand name Duvyzat is a medication used for the treatment of Duchenne muscular dystrophy. It is a histone deacetylase inhibitor with potential anti-inflammatory, anti-angiogenic, and antineoplastic activities. It is a histone deacetylase (HDAC) inhibitor that works by targeting pathogenic processes to reduce inflammation and loss of muscle.

Ruxolitinib, sold under the brand name Jakafi among others, is a medication used for the treatment of intermediate or high-risk myelofibrosis, a type of myeloproliferative neoplasm that affects the bone marrow; polycythemia vera, when there has been an inadequate response to or intolerance of hydroxyurea; and steroid-refractory acute graft-versus-host disease. Ruxolitinib is a Janus kinase inhibitor. It was developed and marketed by Incyte Corp in the US under the brand name Jakafi, and by Novartis elsewhere in the world, under the brand name Jakavi.
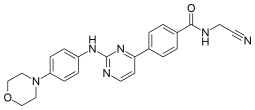
Fedratinib, sold under the brand name Inrebic, is an anti-cancer medication used to treat myeloproliferative diseases including myelofibrosis. It is used in the form of fedratinib hydrochloride capsules that are taken by mouth. It is a semi-selective inhibitor of Janus kinase 2 (JAK-2). It was approved by the FDA on 16 August 2019.

Baricitinib, sold under the brand name Olumiant among others, is an immunomodulatory medication used for the treatment of rheumatoid arthritis, alopecia areata, and COVID-19. It acts as an inhibitor of janus kinase (JAK), blocking the subtypes JAK1 and JAK2.

Trametinib, sold under the brand name Mekinist among others, is an anticancer medication used for the treatment of melanoma and glioma. It is a MEK inhibitor drug with anti-cancer activity. It inhibits MEK1 and MEK2. It is taken by mouth.

Pacritinib, sold under the brand name Vonjo, is an anti-cancer medication used to treat myelofibrosis. It is a macrocyclic protein kinase inhibitor. It mainly inhibits Janus kinase 2 (JAK2) and Fms-like tyrosine kinase 3\CD135 (FLT3).

Filgotinib, sold under the brand name Jyseleca, is a medication used for the treatment of rheumatoid arthritis (RA). It was developed by the Belgian-Dutch biotech company Galapagos NV.

Acalabrutinib, sold under the brand name Calquence, is a medication used to treat various types of non-Hodgkin lymphoma, including mantle cell lymphoma (MCL) and chronic lymphocytic leukemia/small lymphocytic Lymphoma (CLL/SLL). It may be used both in relapsed as well as in treatment-naive settings.

Oclacitinib, sold under the brand name Apoquel among others, is a veterinary medication used in the control of atopic dermatitis and pruritus from allergic dermatitis in dogs at least 12 months of age. Chemically, it is a synthetic cyclohexylamino pyrrolopyrimidine janus kinase inhibitor that is relatively selective for JAK1. It inhibits signal transduction when the JAK is activated and thus helps downregulate expression of inflammatory cytokines.

Upadacitinib, sold under the brand name Rinvoq, is a medication used for the treatment of rheumatoid arthritis, psoriatic arthritis, atopic dermatitis, ulcerative colitis, Crohn's disease, ankylosing spondylitis, and axial spondyloarthritis. Upadacitinib is a Janus kinase (JAK) inhibitor that works by blocking the action of enzymes called Janus kinases. These enzymes are involved in setting up processes that lead to inflammation, and blocking their effect brings inflammation in the joints under control.

Janus kinase 3 inhibitors, also called JAK3 inhibitors, are a new class of immunomodulatory agents that inhibit Janus kinase 3. They are used for the treatment of autoimmune diseases. The Janus kinases are a family of four nonreceptor tyrosine-protein kinases, JAK1, JAK2, JAK3, and TYK2. They signal via the JAK/STAT pathway, which is important in regulating the immune system. Expression of JAK3 is largely restricted to lymphocytes, while the others are ubiquitously expressed, so selective targeting of JAK3 over the other JAK isozymes is attractive as a possible treatment of autoimmune diseases.

Abrocitinib, sold under the brand name Cibinqo, is a medication used for the treatment of atopic dermatitis (eczema). It is a Janus kinase inhibitor and it was developed by Pfizer. It is taken by mouth.

Zotiraciclib (TG02) is a potent oral spectrum selective kinase inhibitor for the treatment of cancer. It was discovered in Singapore by S*BIO Pte Ltd and falls under the category of small molecule macrocycles. It crosses the blood brain barrier and acts by depleting Myc through the inhibition of cyclin-dependent kinase 9 (CDK9). It is one of a number of CDK inhibitors under investigation; others targeting CDK9 for the treatment of acute myeloid leukemia include alvocidib and atuveciclib. Myc overexpression is a known factor in many cancers, with 80 percent of glioblastomas characterized by this property. Zotiraciclib has been granted orphan drug designation by the US Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for the treatment of gliomas.

Pemigatinib, sold under the brand name Pemazyre, is an anti-cancer medication used for the treatment of bile duct cancer (cholangiocarcinoma). Pemigatinib works by blocking FGFR2 in tumor cells to prevent them from growing and spreading.

Mitapivat, sold under the brand name Pyrukynd, is a medication used to treat hemolytic anemia. It is taken as the sulfate hydrate salt by mouth. Mitapivat is a pyruvate kinase activator.

Deucravacitinib, sold under the brand name Sotyktu, is a medication used for the treatment of moderate-to-severe plaque psoriasis. It is a tyrosine kinase 2 (TYK2) inhibitor and it is taken by mouth. It was developed by Bristol Myers Squibb.