
Monoamine oxidase inhibitors (MAOIs) are a class of drugs that inhibit the activity of one or both monoamine oxidase enzymes: monoamine oxidase A (MAO-A) and monoamine oxidase B (MAO-B). They are best known as effective antidepressants, especially for treatment-resistant depression and atypical depression. They are also used to treat panic disorder, social anxiety disorder, Parkinson's disease, and several other disorders.

Serotonin syndrome (SS) is a group of symptoms that may occur with the use of certain serotonergic medications or drugs. The symptoms can range from mild to severe, and are potentially fatal. Symptoms in mild cases include high blood pressure and a fast heart rate; usually without a fever. Symptoms in moderate cases include high body temperature, agitation, increased reflexes, tremor, sweating, dilated pupils, and diarrhea. In severe cases, body temperature can increase to greater than 41.1 °C (106.0 °F). Complications may include seizures and extensive muscle breakdown.

Psychopharmacology is the scientific study of the effects drugs have on mood, sensation, thinking, behavior, judgment and evaluation, and memory. It is distinguished from neuropsychopharmacology, which emphasizes the correlation between drug-induced changes in the functioning of cells in the nervous system and changes in consciousness and behavior.
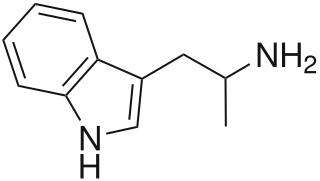
α-Methyltryptamine is a psychedelic, stimulant, and entactogen drug of the tryptamine class. It was originally developed as an antidepressant by chemists at Upjohn in the 1960s, and was used briefly as an antidepressant in Russia under the trade name Indopan before being discontinued.

Phenelzine, sold under the brand name Nardil, among others, is a non-selective and irreversible monoamine oxidase inhibitor (MAOI) of the hydrazine class which is primarily used as an antidepressant and anxiolytic. Along with tranylcypromine and isocarboxazid, phenelzine is one of the few non-selective and irreversible MAOIs still in widespread clinical use.

Tranylcypromine, sold under the brand name Parnate among others, is a monoamine oxidase inhibitor (MAOI). More specifically, tranylcypromine acts as nonselective and irreversible inhibitor of the enzyme monoamine oxidase (MAO). It is used as an antidepressant and anxiolytic agent in the clinical treatment of mood and anxiety disorders, respectively.

Tyramine, also known under several other names, is a naturally occurring trace amine derived from the amino acid tyrosine. Tyramine acts as a catecholamine releasing agent. Notably, it is unable to cross the blood-brain barrier, resulting in only non-psychoactive peripheral sympathomimetic effects following ingestion. A hypertensive crisis can result, however, from ingestion of tyramine-rich foods in conjunction with the use of monoamine oxidase inhibitors (MAOIs).
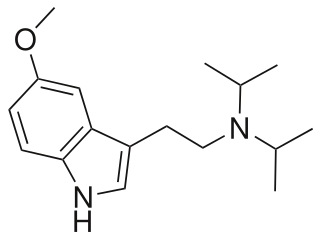
5-Methoxy-N,N-diisopropyltryptamine is a psychedelic tryptamine and the methoxy derivative of diisopropyltryptamine (DiPT).
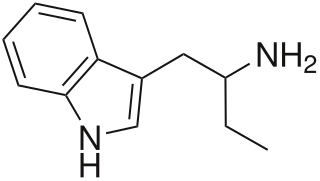
α-Ethyltryptamine, also known as etryptamine, is a psychedelic, stimulant, and entactogenic drug of the tryptamine class. It was originally developed and marketed as an antidepressant under the brand name Monase by Upjohn in the 1960s.

para-Methoxyamphetamine (PMA), also known as 4-methoxyamphetamine (4-MA), is a designer drug of the amphetamine class with serotonergic effects. Unlike other similar drugs of this family, PMA does not produce stimulant, euphoriant, or entactogen effects, and behaves more like an antidepressant in comparison, though it does have some psychedelic properties.

Harmala alkaloids are several alkaloids that increase effects of reward system neurotransmitter dopamine by acting as monoamine oxidase inhibitors (MAOIs). These alkaloids are found in the seeds of Peganum harmala, as well as leaves of tobacco and coffee beans. The alkaloids include harmine, harmaline, harmalol, and their derivatives, which have similar chemical structures, hence the name "harmala alkaloids". These alkaloids are of interest for their use in Amazonian shamanism, where they are derived from other plants. Harmine, once known as telepathine and banisterine, is a naturally occurring beta-carboline alkaloid that is structurally related to harmaline, and also found in the vine Banisteriopsis caapi. Tetrahydroharmine is also found in B. caapi and P. harmala. Dr. Alexander Shulgin has suggested that harmine may be a breakdown product of harmaline. Harmine and harmaline are reversible inhibitors of monoamine oxidase A (RIMAs). They can stimulate the central nervous system by inhibiting the metabolism of monoamine compounds such as serotonin and norepinephrine.

Selegiline, also known as L-deprenyl and sold under the brand names Eldepryl, Emsam, Selgin, among other names, is a medication which is used in the treatment of Parkinson's disease and major depressive disorder. It is provided in the form of a capsule or tablet taken by mouth or orally disintegrating tablets taken on the tongue for Parkinson's disease and as a patch applied to skin for depression.

Deliriants are a subclass of hallucinogen. The term was coined in the early 1980s to distinguish these drugs from psychedelics such as LSD and dissociatives such as ketamine, due to their primary effect of causing delirium, as opposed to the more lucid and less disturbed states produced by other types of hallucinogens. The term generally refers to anticholinergic drugs, which are substances that inhibit the function of the neurotransmitter acetylcholine. Common examples of deliriants include plants of the genera Datura and Brugmansia as well as higher than recommended dosages of diphenhydramine (Benadryl). A number of plant deliriants such as that of the Solanaceae family, particularly in the Americas have been used by some indigenous cultures to reach delirious and altered states for traditions or rituals, such as rites of passage, divination or communicating with the ancestors. Despite their long history of use, deliriants are the least-studied class of hallucinogens in terms of their behavioral and neurological effects.

Dopaminergic means "related to dopamine" (literally, "working on dopamine"), dopamine being a common neurotransmitter. Dopaminergic substances or actions increase dopamine-related activity in the brain. Dopaminergic brain pathways facilitate dopamine-related activity. For example, certain proteins such as the dopamine transporter (DAT), vesicular monoamine transporter 2 (VMAT2), and dopamine receptors can be classified as dopaminergic, and neurons that synthesize or contain dopamine and synapses with dopamine receptors in them may also be labeled as dopaminergic. Enzymes that regulate the biosynthesis or metabolism of dopamine such as aromatic L-amino acid decarboxylase or DOPA decarboxylase, monoamine oxidase (MAO), and catechol O-methyl transferase (COMT) may be referred to as dopaminergic as well. Also, any endogenous or exogenous chemical substance that acts to affect dopamine receptors or dopamine release through indirect actions (for example, on neurons that synapse onto neurons that release dopamine or express dopamine receptors) can also be said to have dopaminergic effects, two prominent examples being opioids, which enhance dopamine release indirectly in the reward pathways, and some substituted amphetamines, which enhance dopamine release directly by binding to and inhibiting VMAT2.

Moclobemide, sold under the brand names Amira, Aurorix, Clobemix, Depnil and Manerix among others, is a reversible inhibitor of monoamine oxidase A (RIMA) drug primarily used to treat depression and social anxiety. It is not approved for use in the United States, but is approved in other Western countries such as Canada, the UK and Australia. It is produced by affiliates of the Hoffmann–La Roche pharmaceutical company. Initially, Aurorix was also marketed by Roche in South Africa, but was withdrawn after its patent rights expired and Cipla Medpro's Depnil and Pharma Dynamic's Clorix became available at half the cost.

Iproniazid is a non-selective, irreversible monoamine oxidase inhibitor (MAOI) of the hydrazine class. It is a xenobiotic that was originally designed to treat tuberculosis, but was later most prominently used as an antidepressant drug. However, it was withdrawn from the market because of its hepatotoxicity. The medical use of iproniazid was discontinued in most of the world in the 1960s, but remained in use in France until its discontinuation in 2015.

para-Methoxy-N-methylamphetamine, chemically known as methyl-MA, 4-methoxy-N-methylamphetamine, and 4-MMA is a stimulant and psychedelic drug closely related to the amphetamine-class serotonergic drug para-methoxyamphetamine (PMA). PMMA is the 4-methoxy analog of methamphetamine. Little is known about the pharmacological properties, metabolism, and toxicity of PMMA; because of its structural similarity to PMA, which has known toxicity in humans, it is thought to have considerable potential to cause harmful side effects or death in overdose. In the early 2010s, a number of deaths in users of the drug MDMA were linked to misrepresented tablets and capsules of PMMA.
An adrenergic storm is a sudden and dramatic increase in serum levels of the catecholamines adrenaline and noradrenaline, with a less significant increase in dopamine transmission. It is a life-threatening condition because of extreme tachycardia and hypertension, and is especially dire for those with prior heart problems. If treatment is prompt, prognosis is good; typically large amounts of diazepam or other benzodiazepines are administered alongside beta blockers. Beta blockers are contraindicated in some patients, so other anti-hypertensive medication such as clonidine may be used. Antipsychotics are also used to treat the most severe psychiatric reactions such as psychosis, paranoia or terror, after their use was formerly discouraged because of their potential to prolong the QT interval; however, more recent research performed since 2019 has revealed that this and other severe side effects are rare and their occurrence does not warrant banning antipsychotics from the treatment of adrenergic crises for which they can be extremely useful.

Amiflamine (FLA-336) is a reversible inhibitor of monoamine oxidase A (MAO-A), thereby being a RIMA, and, to a lesser extent, semicarbazide-sensitive amine oxidase (SSAO), as well as a serotonin releasing agent (SRA). It is a derivative of the phenethylamine and amphetamine chemical classes. The (+)-enantiomer is the active stereoisomer.
Amphetamine type stimulants (ATS) are a group of synthetic drugs that are chemical derivatives of the parent compound alpha-methylphenethylamine, also known as amphetamine. Common ATS includes amphetamine, methamphetamine, ephedrine, pseudoephedrine, 3,4-methylenedioxymethamphetamine (MDMA), 3,4-methylenedioxyamphetamine (MDA) and 3,4-methylenedioxyethylamphetamine (MDEA). ATS when used illicitly has street names including ice, meth, crystal, crank, bennies, and speed. Within the group of amphetamine-type stimulants, there are also prescription drugs including mixed amphetamine salts, dextroamphetamine, and lisdexamfetamine.





















