Related Research Articles

Orexin, also known as hypocretin, is a neuropeptide that regulates arousal, wakefulness, and appetite. The most common form of narcolepsy, type 1, in which the individual experiences brief losses of muscle tone, is caused by a lack of orexin in the brain due to destruction of the cells that produce it. It exists in the forms of orexin-A and orexin-B.
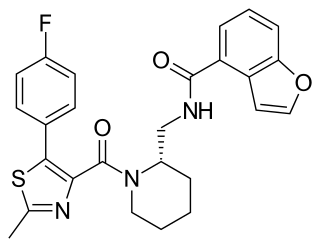
SB-649868 is a dual orexin receptor antagonist that was being developed by GlaxoSmithKline as a treatment for insomnia.

Orexin receptor type 1 (Ox1R or OX1), also known as hypocretin receptor type 1 (HcrtR1), is a protein that in humans is encoded by the HCRTR1 gene.

Orexin receptor type 2 (Ox2R or OX2), also known as hypocretin receptor type 2 (HcrtR2), is a protein that in humans is encoded by the HCRTR2 gene.

Almorexant, also known by its development code ACT-078573, is an orexin antagonist, acting as a competitive antagonist of the OX1 and OX2 orexin receptors, which was being developed by the pharmaceutical companies Actelion and GSK for the treatment of insomnia. Development of the drug was abandoned in January 2011 due to concerns over the hepatic safety of almorexant after transient increases in liver enzymes were observed in trials.
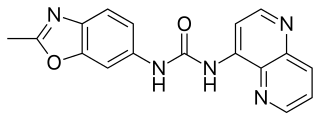
SB-334867 is an orexin antagonist. It was the first non-peptide antagonist developed that is selective for the orexin receptor subtype OX1, with around 50x selectivity for OX1 over OX2 receptors. It has been shown to produce sedative and anorectic effects in animals, and has been useful in characterising the orexinergic regulation of brain systems involved with appetite and sleep, as well as other physiological processes. The hydrochloride salt of SB-334867 has been demonstrated to be hydrolytically unstable, both in solution and as the solid. Orexin antagonists have multiple potential clinical applications including the treatment of drug addiction, insomnia, obesity and diabetes.
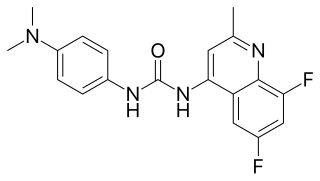
SB-408124 is a drug which is a non-peptide antagonist selective for the orexin receptor subtype OX1, with around 70x selectivity for OX1 over OX2 receptors, and improved oral bioavailability compared to the older OX1 antagonist SB-334867. It is used in scientific research into the function of orexinergic neurons in the body.

Suvorexant, sold under the brand name Belsomra, is an orexin antagonist medication which is used in the treatment of insomnia. It is indicated specifically for the treatment of insomnia characterized by difficulties with sleep onset and/or maintenance in adults. Suvorexant helps with falling asleep faster, sleeping longer, being awake less in the middle of the night, and having better quality of sleep. Its effectiveness is modest, and is similar to that of other orexin antagonists, but is lower than that of benzodiazepines and Z-drugs. Suvorexant is taken by mouth.
An orexin receptor antagonist, or orexin antagonist, is a drug that inhibits the effect of orexin by acting as a receptor antagonist of one (selective orexin receptor antagonist or SORA) or both (dual orexin receptor antagonis or DORA) of the orexin receptors, OX1 and OX2. Medical applications include treatment of sleep disorders such as insomnia.
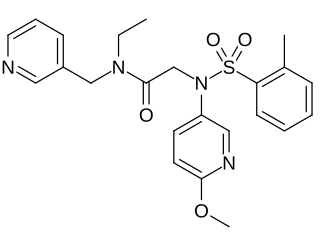
EMPA is a selective antagonist of the OX2 receptor, with 900-fold selectivity in binding for OX2 over OX1.

Filorexant (INNTooltip International Nonproprietary Name, USANTooltip United States Approved Name; developmental code name MK-6096) is an orexin antagonist which was under development by Merck for the treatment of insomnia, depression, diabetic neuropathy, and migraine. It is a dual antagonist of the orexin OX1 and OX2 receptors. It has a relatively short elimination half-life of 3 to 6 hours. However, it dissociates slowly from the orexin receptors and may thereby have a longer duration. Possibly in relation to this, filorexant shows next-day somnolence similarly to suvorexant. In phase 2 clinical trials, filorexant was found to be effective in the treatment of insomnia, but was not effective in the treatment of major depressive disorder, painful diabetic neuropathy, or migraine. As of May 2015, filorexant was no longer listed on Merck's online development pipeline and hence development of the drug appears to have been discontinued. Development of filorexant may have been discontinued due to lack of differentiation from suvorexant (which was also developed by Merck).

Seltorexant, also known by its developmental code names MIN-202 and JNJ-42847922, is an orexin antagonist medication which is under development for the treatment of depression and insomnia. It is a selective antagonist of the orexin OX2 receptor (2-SORA). The medication is taken by mouth. As of February 2022, seltorexant is in phase 3 clinical trials for treatment of major depressive disorder (MDD) and phase 2 trials for treatment of insomnia. It was also under investigation for the treatment of sleep apnea, but no recent development has been reported for this indication. Seltorexant is under development by Minerva Neurosciences and Johnson & Johnson's Janssen Pharmaceuticals.

Lemborexant, sold under the brand name Dayvigo, is an orexin antagonist medication which is used in the treatment of insomnia. It is indicated specifically for the treatment of insomnia characterized by difficulties with sleep onset and/or maintenance in adults. The medication is taken by mouth.

Daridorexant, sold under the brand name Quviviq, is an orexin antagonist medication which is used for the treatment of insomnia. Daridorexant is taken by mouth.

Vornorexant, also known by its developmental code names ORN-0829 and TS-142, is an orexin antagonist medication which is under development for the treatment of insomnia and sleep apnea. It is a dual orexin OX1 and OX2 receptor antagonist (DORA). The medication is taken by mouth. As of June 2021, vornorexant is in phase 2 clinical trials for insomnia and phase 1 trials for sleep apnea. It is under development by Taisho Pharmaceutical.

Firazorexton (INNTooltip International Nonproprietary Name; development code TAK-994) is an experimental orexin 2 (OX2) receptor agonist first described in a 2019 patent filed by Takeda Pharmaceutical Company.

ACT-462206 is a dual orexin receptor antagonist (IC50 for OX1 = 60nM, OX2 = 11nM) which has been investigated for the treatment of insomnia. In human trials, ACT-462206 produced dose-dependent sedative effects and was generally well tolerated, with residual sleepiness and headache being the most common adverse events.

JNJ-61393215 is an orexin antagonist medication which is under development for the treatment of depression and anxiety disorders. It is an orally active compound and acts as a selective antagonist of the orexin OX1 receptor (1-SORA). Preliminary clinical findings suggest that JNJ-61393215 may have anti-panic effects in humans. As of November 2021, JNJ-61393215 is in phase 2 clinical trials for the treatment of major depressive disorder and is in the preclinical stage of development for treatment of panic disorder, while no further development has been reported for treatment of other anxiety disorders. The drug was originated and developed by Janssen Pharmaceuticals.
References
- ↑ Spinazzi R, Andreis PG, Rossi GP, Nussdorfer GG (2006). "Orexins in the regulation of the hypothalamic-pituitary-adrenal axis". Pharmacol. Rev. 58 (1): 46–57. doi:10.1124/pr.58.1.4. PMID 16507882. S2CID 17941978.
- 1 2 3 Smart D, Jerman JC, Brough SJ, Rushton SL, Murdock PR, Jewitt F, Elshourbagy NA, Ellis CE, Middlemiss DN, Brown F (September 1999). "Characterization of recombinant human orexin receptor pharmacology in a Chinese hamster ovary cell-line using FLIPR". Br. J. Pharmacol. 128 (1): 1–3. doi:10.1038/sj.bjp.0702780. PMC 1571615 . PMID 10498827.
- 1 2 3 Langmead CJ, Jerman JC, Brough SJ, Scott C, Porter RA, Herdon HJ (January 2004). "Characterisation of the binding of [3H]-SB-674042, a novel nonpeptide antagonist, to the human orexin-1 receptor". British Journal of Pharmacology. 141 (2): 340–6. doi:10.1038/sj.bjp.0705610. PMC 1574197 . PMID 14691055.
- ↑ Yin J, Mobarec JC, Kolb P, Rosenbaum DM (December 2014). "Crystal Structure of the Human Ox2 Orexin Receptor Bound to the Insomnia Drug Suvorexant". Nature. 519 (7542): 247–250. doi:10.1038/nature14035. PMID 25533960. S2CID 4405254.
- ↑ "Merck's Insomnia Medicine Belsomra C-IV Now Available in US". Sleep Review. 3 February 2015. Retrieved 2019-12-06.
- ↑ "New Data Presented at World Sleep Congress Demonstrate Early Signs of Efficacy for TAK-925, a Selective Orexin Type-2 Receptor (OX2R) Agonist, in Patients with Narcolepsy Type 1". www.takeda.com. Retrieved 2019-12-06.
- ↑ Heifetz A, Morris GB, Biggin PC, Barker O, Fryatt T, Bentley J, Hallett D, Manikowski DP, Pal S, Reifegerste R, Slack M, Law R (2012). "Study of Human Orexin-1 and -2 G-Protein-Coupled Receptors with Novel and Published Antagonists by Modeling, Molecular Dynamics Simulations, and Site-Directed Mutagenesis". Biochemistry. 51 (15): 3178–3197. doi:10.1021/bi300136h. PMID 22448975. S2CID 42765328.
- ↑ Chow M, Cao M (2016). "The hypocretin/orexin system in sleep disorders: preclinical insights and clinical progress". Nat Sci Sleep. 8: 81–6. doi: 10.2147/NSS.S76711 . PMC 4803263 . PMID 27051324.
- ↑ Baxter CA, Cleator ED, Karel MJ, Edwards JS, Reamer RA, Sheen FJ, Stewart GW, Strotman NA, Wallace DJ (2011). "The First Large-Scale Synthesis of MK-4305: A Dual Orexin Receptor Antagonist for the Treatment of Sleep Disorder". Organic Process Research & Development. 15 (2): 367–375. doi:10.1021/op1002853.
- ↑ Hoch M, van Gorsel H, van Gerven J, Dingemanse J (Sep 2014). "Entry-into-humans study with ACT-462206, a novel dual orexin receptor antagonist, comparing its pharmacodynamics with almorexant". J Clin Pharmacol. 54 (9): 979–86. doi:10.1002/jcph.297. PMID 24691844. S2CID 40714628.
- 1 2 "WHO Drug Information, Vol. 34, No. 2, 2020 Proposed INN: List 123 : International Nonproprietary Names for Pharmaceutical Substances (INN)" (PDF). Who.int. Retrieved 1 December 2021.
- 1 2 WOapplication 2019027058,Kajita, Yuichi; Mikami, Satoshi& Miyanohana, Yuheiet al.,"Heterocyclic compound and use therof",published 2019-02-07, assigned to Takeda Pharmaceutical Company
- ↑ "Wave 1 Pipeline Market Opportunity Conference Call" (PDF). Takeda Pharmaceutical Company Limited. 8 December 2020. Archived from the original (PDF) on 2021-10-20.
TAK-861, a second oral OX2R agonist will begin clinical testing in 2H FY20