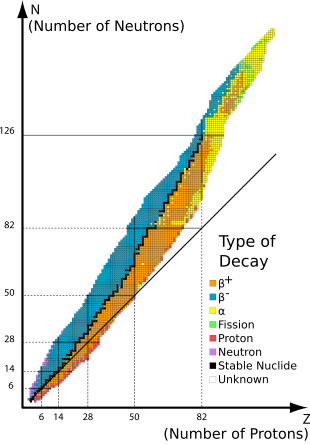A radionuclide (radioactive nuclide, radioisotope or radioactive isotope) is a nuclide that has excess numbers of either neutrons or protons, giving it excess nuclear energy, and making it unstable. This excess energy can be used in one of three ways: emitted from the nucleus as gamma radiation; transferred to one of its electrons to release it as a conversion electron; or used to create and emit a new particle (alpha particle or beta particle) from the nucleus. During those processes, the radionuclide is said to undergo radioactive decay. These emissions are considered ionizing radiation because they are energetic enough to liberate an electron from another atom. The radioactive decay can produce a stable nuclide or will sometimes produce a new unstable radionuclide which may undergo further decay. Radioactive decay is a random process at the level of single atoms: it is impossible to predict when one particular atom will decay. However, for a collection of atoms of a single nuclide the decay rate, and thus the half-life (t1/2) for that collection, can be calculated from their measured decay constants. The range of the half-lives of radioactive atoms has no known limits and spans a time range of over 55 orders of magnitude.

A nuclide is a class of atoms characterized by their number of protons, Z, their number of neutrons, N, and their nuclear energy state.

Neutron capture is a nuclear reaction in which an atomic nucleus and one or more neutrons collide and merge to form a heavier nucleus. Since neutrons have no electric charge, they can enter a nucleus more easily than positively charged protons, which are repelled electrostatically.
Natural tantalum (73Ta) consists of two stable isotopes: 181Ta (99.988%) and 180m
Ta
(0.012%).
Naturally occurring samarium (62Sm) is composed of five stable isotopes, 144Sm, 149Sm, 150Sm, 152Sm and 154Sm, and two extremely long-lived radioisotopes, 147Sm (half life: 1.06×1011 y) and 148Sm (6.3×1015 y), with 152Sm being the most abundant (26.75% natural abundance). 146Sm is also fairly long-lived, but is not long-lived enough to have survived in significant quantities from the formation of the Solar System on Earth, although it remains useful in radiometric dating in the Solar System as an extinct radionuclide. A 2012 paper revising the estimated half-life of 146Sm from 10.3(5)×107 y to 6.8(7)×107 y was retracted in 2023. It is the longest-lived nuclide that has not yet been confirmed to be primordial.
Naturally occurring xenon (54Xe) consists of seven stable isotopes and two very long-lived isotopes. Double electron capture has been observed in 124Xe and double beta decay in 136Xe, which are among the longest measured half-lives of all nuclides. The isotopes 126Xe and 134Xe are also predicted to undergo double beta decay, but this has never been observed in these isotopes, so they are considered to be stable. Beyond these stable forms, 32 artificial unstable isotopes and various isomers have been studied, the longest-lived of which is 127Xe with a half-life of 36.345 days. All other isotopes have half-lives less than 12 days, most less than 20 hours. The shortest-lived isotope, 108Xe, has a half-life of 58 μs, and is the heaviest known nuclide with equal numbers of protons and neutrons. Of known isomers, the longest-lived is 131mXe with a half-life of 11.934 days. 129Xe is produced by beta decay of 129I ; 131mXe, 133Xe, 133mXe, and 135Xe are some of the fission products of both 235U and 239Pu, so are used as indicators of nuclear explosions.

There are 37 known isotopes of iodine (53I) from 108I to 144I; all undergo radioactive decay except 127I, which is stable. Iodine is thus a monoisotopic element.

This page discusses each of the main elements in the mixture of fission products produced by nuclear fission of the common nuclear fuels uranium and plutonium. The isotopes are listed by element, in order by atomic number.
A table or chart of nuclides is a two-dimensional graph of isotopes of the elements, in which one axis represents the number of neutrons and the other represents the number of protons in the atomic nucleus. Each point plotted on the graph thus represents a nuclide of a known or hypothetical chemical element. This system of ordering nuclides can offer a greater insight into the characteristics of isotopes than the better-known periodic table, which shows only elements and not their isotopes. The chart of the nuclides is also known as the Segrè chart, after the Italian physicist Emilio Segrè.
An extinct radionuclide is a radionuclide that was formed by nucleosynthesis before the formation of the Solar System, about 4.6 billion years ago, but has since decayed to virtually zero abundance and is no longer detectable as a primordial nuclide. Extinct radionuclides were generated by various processes in the early Solar system, and became part of the composition of meteorites and protoplanets. All widely documented extinct radionuclides have half-lives shorter than 100 million years.

The neutron number is the number of neutrons in a nuclide.

Isotopes are distinct nuclear species of the same chemical element. They have the same atomic number and position in the periodic table, but differ in nucleon numbers due to different numbers of neutrons in their nuclei. While all isotopes of a given element have almost the same chemical properties, they have different atomic masses and physical properties.

In geochemistry, geophysics and nuclear physics, primordial nuclides, also known as primordial isotopes, are nuclides found on Earth that have existed in their current form since before Earth was formed. Primordial nuclides were present in the interstellar medium from which the solar system was formed, and were formed in, or after, the Big Bang, by nucleosynthesis in stars and supernovae followed by mass ejection, by cosmic ray spallation, and potentially from other processes. They are the stable nuclides plus the long-lived fraction of radionuclides surviving in the primordial solar nebula through planet accretion until the present; 286 such nuclides are known.

Isobars are atoms (nuclides) of different chemical elements that have the same number of nucleons. Correspondingly, isobars differ in atomic number but have the same mass number. An example of a series of isobars is 40S, 40Cl, 40Ar, 40K, and 40Ca. While the nuclei of these nuclides all contain 40 nucleons, they contain varying numbers of protons and neutrons.

A monoisotopic element is an element which has only a single stable isotope (nuclide). There are 26 such elements, as listed.

A radiogenic nuclide is a nuclide that is produced by a process of radioactive decay. It may itself be radioactive or stable.
The Mattauch isobar rule, formulated by Josef Mattauch in 1934, states that if two adjacent elements on the periodic table have isotopes of the same mass number, one of these isotopes must be radioactive. Two nuclides that have the same mass number (isobars) can both be stable only if their atomic numbers differ by more than one. In fact, for currently observationally stable nuclides, the difference can only be 2 or 4, and in theory, two nuclides that have the same mass number cannot be both stable, but many such nuclides which are theoretically unstable to double beta decay have not been observed to decay, e.g. 134Xe. However, this rule cannot make predictions on the half-lives of these radioisotopes.

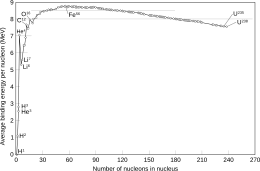
In nuclear physics, properties of a nucleus depend on evenness or oddness of its atomic number Z, neutron number N and, consequently, of their sum, the mass number A. Most importantly, oddness of both Z and N tends to lower the nuclear binding energy, making odd nuclei generally less stable. This effect is not only experimentally observed, but is included in the semi-empirical mass formula and explained by some other nuclear models, such as the nuclear shell model. This difference of nuclear binding energy between neighbouring nuclei, especially of odd-A isobars, has important consequences for beta decay.