In chemistry, a chemical formula is a way of presenting information about the chemical proportions of atoms that constitute a particular chemical compound or molecule, using chemical element symbols, numbers, and sometimes also other symbols, such as parentheses, dashes, brackets, commas and plus (+) and minus (−) signs. These are limited to a single typographic line of symbols, which may include subscripts and superscripts. A chemical formula is not a chemical name, and it contains no words. Although a chemical formula may imply certain simple chemical structures, it is not the same as a full chemical structural formula. Chemical formulae can fully specify the structure of only the simplest of molecules and chemical substances, and are generally more limited in power than chemical names and structural formulae.
In organic chemistry, a hydrocarbon is an organic compound consisting entirely of hydrogen and carbon. Hydrocarbons are examples of group 14 hydrides. Hydrocarbons are generally colourless and hydrophobic; their odor is usually faint, and may be similar to that of gasoline or lighter fluid. They occur in a diverse range of molecular structures and phases: they can be gases, liquids, low melting solids or polymers.
In chemistry, a structural isomer of a compound is another compound whose molecule has the same number of atoms of each element, but with logically distinct bonds between them. The term metamer was formerly used for the same concept.

Chlorofluorocarbons (CFCs) and hydrochlorofluorocarbons (HCFCs) are fully or partly halogenated hydrocarbons that contain carbon (C), hydrogen (H), chlorine (Cl), and fluorine (F), produced as volatile derivatives of methane, ethane, and propane.
Chloroform, or trichloromethane (often abbreviated as TCM), is an organic compound with the formula CHCl3 and a common organic solvent. It is a very volatile, colorless, strong-smelling, dense liquid produced on a large scale as a precursor to PTFE and refrigerants and is a trihalomethane that serves as a powerful anesthetic, euphoriant, anxiolytic, and sedative when inhaled or ingested. Chloroform was frequently used as an anaesthetic between 1847 and the first half of the 20th century. It is also part of a wider class of substances known as volatile organic compounds. Chloroform is miscible with many solvents but it is only very slightly soluble in water (only 8 g/L at 20 °C).
The compound hydrogen chloride has the chemical formula HCl and as such is a hydrogen halide. At room temperature, it is a colourless gas, which forms white fumes of hydrochloric acid upon contact with atmospheric water vapor. Hydrogen chloride gas and hydrochloric acid are important in technology and industry. Hydrochloric acid, the aqueous solution of hydrogen chloride, is also commonly given the formula HCl.

The organic compound 1,1,1-trichloroethane, also known as methyl chloroform, is a chloroalkane with the chemical formula CH3CCl3. It is an isomer of 1,1,2-trichloroethane. This colorless, sweet-smelling liquid was once produced industrially in large quantities for use as a solvent. It is regulated by the Montreal Protocol as an ozone-depleting substance and its use is being rapidly phased out.
Halomethane compounds are derivatives of methane with one or more of the hydrogen atoms replaced with halogen atoms. Halomethanes are both naturally occurring, especially in marine environments, and human-made, most notably as refrigerants, solvents, propellants, and fumigants. Many, including the chlorofluorocarbons, have attracted wide attention because they become active when exposed to ultraviolet light found at high altitudes and destroy the Earth's protective ozone layer.

Trichloroacetic acid is an analogue of acetic acid in which the three hydrogen atoms of the methyl group have all been replaced by chlorine atoms. Salts and esters of trichloroacetic acid are called trichloroacetates.
In organic chemistry, free-radical halogenation is a type of halogenation. This chemical reaction is typical of alkanes and alkyl-substituted aromatics under application of UV light. The reaction is used for the industrial synthesis of chloroform (CHCl3), dichloromethane (CH2Cl2), and hexachlorobutadiene. It proceeds by a free-radical chain mechanism.

Phosphorus pentachloride is the chemical compound with the formula PCl5. It is one of the most important phosphorus chlorides, others being PCl3 and POCl3. PCl5 finds use as a chlorinating reagent. It is a colourless, water-sensitive solid, although commercial samples can be yellowish and contaminated with hydrogen chloride.
In chemistry, the inductive effect in a molecule is a local change in the electron density due to electron-withdrawing or electron-donating groups elsewhere in the molecule, resulting in a permanent dipole in a bond. It is present in a σ (sigma) bond, unlike the electromeric effect which is present in a π (pi) bond.
The Reed reaction is a chemical reaction that utilizes light to oxidize hydrocarbons to alkylsulfonyl chlorides. This reaction is employed in modifying polyethylene to give chlorosulfonated polyethylene (CSPE), which noted for its toughness.
In chemistry, a reaction intermediate or an intermediate is a molecular entity that is formed from the reactants but is consumed in further reactions in stepwise chemical reactions that contain multiple elementary steps. Intermediates are the reaction product of one elementary step, but do not appear in the chemical equation for an overall chemical equation.
In group theory, geometry, representation theory and molecular geometry, a symmetry operation is a geometric transformation of an object that leaves the object looking the same after it has been carried out. For example, as transformations of an object in space, rotations, reflections and inversions are all symmetry operations. Such symmetry operations are performed with respect to symmetry elements. In the context of molecular symmetry, a symmetry operation is a permutation of atoms such that the molecule or crystal is transformed into a state indistinguishable from the starting state. Two basic facts follow from this definition, which emphasizes its usefulness.
- Physical properties must be invariant with respect to symmetry operations.
- Symmetry operations can be collected together in groups which are isomorphic to permutation groups.

In chemistry, isomers are molecules or polyatomic ions with identical molecular formula – that is, same number of atoms of each element – but distinct arrangements of atoms in space. Isomerism is existence or possibility of isomers.

The trichloromethyl group is a functional group that has the formula –CCl3. The naming of is group is derived from the methyl group (which has the formula –CH3), by replacing each hydrogen atom by a chlorine atom. Compounds with this group are a subclass of the organochlorines. Some notable examples of compounds with this group are trichloromethane H–CCl
3, 1,1,1-trichloroethane H
3C–CCl
3, and chloral HOC–CCl
3.

1,1,2,2,3,3-Hexachloropropane is a compound of chlorine, hydrogen, and carbon, with structural formula Cl2HC−CCl2−CCl2H. Its molecule can be described as that of propane with chlorine atoms substituted for six hydrogen atoms, two on each carbon. It is a liquid at ambient temperature.

Octachloropropane or perchloropropane is the chemical compound with elemental formula C3Cl8 and structural formula Cl3C−CCl2−CCl3. Its molecule has a simple chain of three carbon atoms connected by single bonds, with chlorine atoms filling their remaining bonds. It is a chlorocarbon, specifically the third simplest perchloroalkane. It can be described as a derivative of propane C3H8, with all hydrogen atoms replaced by chlorine.
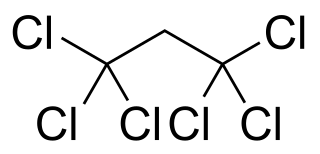
1,1,1,3,3,3-Hexachloropropane is a compound of chlorine, hydrogen, and carbon, with chemical formula C3Cl6H2, specifically Cl3C−CH2−CCl3. Its molecule can be described as that of propane with chlorine atoms substituted for the six hydrogen atoms on the extremal carbons.










