
Dithiolene metal complexes are complexes containing 1,2-dithiolene ligands. 1,2-Dithiolene ligands, a particular case of 1,2-dichalcogenolene species along with 1,2-diselenolene derivatives, are unsaturated bidentate ligand wherein the two donor atoms are sulfur. 1,2-Dithiolene metal complexes are often referred to as "metal dithiolenes", "metallodithiolenes" or "dithiolene complexes". Most molybdenum- and tungsten-containing proteins have dithiolene-like moieties at their active sites, which feature the so-called molybdopterin cofactor bound to the Mo or W.

1,2-Bis(diphenylphosphino)ethane (dppe) is an organophosphorus compound with the formula (Ph2PCH2)2 (Ph = phenyl). It is a common symmetrical bidentate ligand in coordination chemistry. It is a white solid that is soluble in organic solvents.

In coordination chemistry, hapticity is the coordination of a ligand to a metal center via an uninterrupted and contiguous series of atoms. The hapticity of a ligand is described with the Greek letter η ('eta'). For example, η2 describes a ligand that coordinates through 2 contiguous atoms. In general the η-notation only applies when multiple atoms are coordinated. In addition, if the ligand coordinates through multiple atoms that are not contiguous then this is considered denticity, and the κ-notation is used once again. When naming complexes care should be taken not to confuse η with μ ('mu'), which relates to bridging ligands.

1,1′-Bis(diphenylphosphino)ferrocene, commonly abbreviated dppf, is an organophosphorus compound commonly used as a ligand in homogeneous catalysis. It contains a ferrocene moiety in its backbone, and is related to other bridged diphosphines such as 1,2-bis(diphenylphosphino)ethane (dppe).

Diphenylphosphine, also known as diphenylphosphane, is an organophosphorus compound with the formula (C6H5)2PH. This foul-smelling, colorless liquid is easily oxidized in air. It is a precursor to organophosphorus ligands for use as catalysts.

1,1-Bis(diphenylphosphino)methane (dppm), is an organophosphorus compound with the formula CH2(PPh2)2. Dppm, a white, crystalline powder, is used in inorganic and organometallic chemistry as a ligand. It is more specifically a chelating ligand because it is a ligand that can bond to metals with two phosphorus donor atoms. The natural bite angle is 73°.

1,2-Bis(diisopropylphosphino)ethane (dippe) is a commonly used bidentate ligand in coordination chemistry. This compound is similar to the ligand 1,2-bis(diphenylphosphino)ethane (dppe), with the substitution of isopropyl groups for phenyl groups.
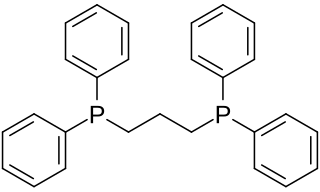
1,3-Bis(diphenylphosphino)propane (dppp) is an organophosphorus compound with the formula Ph2P(CH2)3PPh2. The compound is a white solid that is soluble in organic solvents. It is slightly air-sensitive, degrading in air to the phosphine oxide. It is classified as a diphosphine ligand in coordination chemistry and homogeneous catalysis.

1,2-Bis(dimethylphosphino)ethane (dmpe) is a diphosphine ligand in coordination chemistry. It is a colorless, air-sensitive liquid that is soluble in organic solvents. With the formula (CH2PMe2)2, dmpe is used as a compact strongly basic spectator ligand (Me = methyl), Representative complexes include V(dmpe)2(BH4)2, Mn(dmpe)2(AlH4)2, Tc(dmpe)2(CO)2Cl, and Ni(dmpe)Cl2.

A metal-phosphine complex is a coordination complex containing one or more phosphine ligands. Almost always, the phosphine is an organophosphine of the type R3P (R = alkyl, aryl). Metal phosphine complexes are useful in homogeneous catalysis. Prominent examples of metal phosphine complexes include Wilkinson's catalyst (Rh(PPh3)3Cl), Grubbs' catalyst, and tetrakis(triphenylphosphine)palladium(0).

trans-Bis(dinitrogen)bis[1,2-bis(diphenylphosphino)ethane]molybdenum(0) is a coordination complex with the formula Mo(N2)2(dppe)2. It is a relatively air stable yellow-orange solid. It is notable as being the first discovered dinitrogen containing complex of molybdenum.
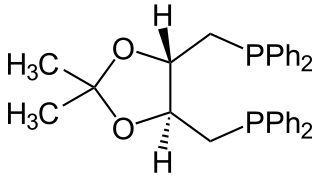
DIOP is an organophosphorus compound that is used as a chiral ligand in asymmetric catalysis. It is a white solid that is soluble in organic solvents.

Chlorobis(dppe)iron hydride is a coordination complex with the formula HFeCl(dppe)2, where dppe is the bidentate ligand 1,2-bis(diphenylphosphino)ethane. It is a red-violet solid. The compound has attracted much attention as a precursor to dihydrogen complexes.

In organic chemistry, peri-naphthalenes are particular derivatives of naphthalene with the formula C10H6-1,8-X2.
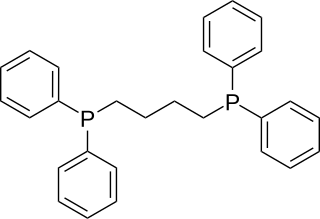
1,4-Bis(diphenylphosphino)butane (dppb) is an organophosphorus compound with the formula (Ph2PCH2CH2)2. It is less commonly used in coordination chemistry than other diphosphine ligands such as dppe. It is a white solid that is soluble in organic solvents.

Dichloro[1,2-bis(diphenylphosphino)ethane]nickel is a coordination complex with the formula NiCl2(dppe); where dppe is the diphosphine 1,2-bis(diphenylphosphino)ethane. It is used as a reagent and as a catalyst. The compound is a bright orange-red diamagnetic solid. The complex adopts a square planar geometry.

1,2-Bis(dichlorophosphino)benzene is an organophosphorus compound with the formula C6H4(PCl2)2. This viscous colorless liquid is a precursor to chelating diphosphines of the type C6H4(PR2)2. It is prepared from 1,2-dibromobenzene by sequential lithiation followed by treatment with (Et2N)2PCl (Et = ethyl), which affords C6H4[P(NEt2)2]2. This species is finally cleaved with hydrogen chloride:
1,2-Bis(dichlorophosphino)ethane is an organophosphorus compound with the formula (CH2PCl2)2. This colorless liquid is a precursor to chelating diphosphines.
cis-1,2-Bis(diphenylphosphino)ethylene (dppv) is an organophosphorus compound with the formula C2H2(PPh2)2 (Ph = C6H5). Both the cis and trans isomers are known, but the cis isomer is of primary interest. Classified as a diphosphine ligand, it is a bidentate ligand in coordination chemistry. For example it gives rise to the complex Ni(dppv)2 and the coordination polymer [Ni(dppv)]n. As a chelating ligand, dppv is very similar to 1,2-bis(diphenylphosphino)benzene.
Palladium forms a variety of ionic, coordination, and organopalladium compounds, typically with oxidation state Pd0 or Pd2+. Palladium(III) compounds have also been reported. Palladium compounds are frequently used as catalysts in cross-coupling reactions such as the Sonogashira coupling and Suzuki reaction.

















