
A protein kinase is a kinase enzyme that modifies other proteins by chemically adding phosphate groups to them (phosphorylation). Phosphorylation usually results in a functional change of the target protein (substrate) by changing enzyme activity, cellular location, or association with other proteins. The human genome contains about 500 protein kinase genes and they constitute about 2% of all human genes. Protein kinases are also found in bacteria and plants. Up to 30% of all human proteins may be modified by kinase activity, and kinases are known to regulate the majority of cellular pathways, especially those involved in signal transduction.
The 70 kilodalton heat shock proteins are a family of conserved ubiquitously expressed heat shock proteins. Proteins with similar structure exist in virtually all living organisms. The Hsp70s are an important part of the cell's machinery for protein folding, and help to protect cells from stress.
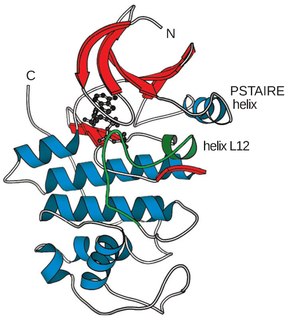
Cyclin-dependent kinases (CDKs) are the families of protein kinases first discovered for their role in regulating the cell cycle. They are also involved in regulating transcription, mRNA processing, and the differentiation of nerve cells. They are present in all known eukaryotes, and their regulatory function in the cell cycle has been evolutionarily conserved. In fact, yeast cells can proliferate normally when their CDK gene has been replaced with the homologous human gene. CDKs are relatively small proteins, with molecular weights ranging from 34 to 40 kDa, and contain little more than the kinase domain. By definition, a CDK binds a regulatory protein called a cyclin. Without cyclin, CDK has little kinase activity; only the cyclin-CDK complex is an active kinase. CDKs phosphorylate their substrates on serines and threonines, so they are serine-threonine kinases. The consensus sequence for the phosphorylation site in the amino acid sequence of a CDK substrate is [S/T*]PX[K/R], where S/T* is the phosphorylated serine or threonine, P is proline, X is any amino acid, K is lysine, and R is arginine.

Hsp90 is a chaperone protein that assists other proteins to fold properly, stabilizes proteins against heat stress, and aids in protein degradation. It also stabilizes a number of proteins required for tumor growth, which is why Hsp90 inhibitors are investigated as anti-cancer drugs.

14-3-3 proteins are a family of conserved regulatory molecules that are expressed in all eukaryotic cells. 14-3-3 proteins have the ability to bind a multitude of functionally diverse signaling proteins, including kinases, phosphatases, and transmembrane receptors. More than 200 signaling proteins have been reported as 14-3-3 ligands.

RAF proto-oncogene serine/threonine-protein kinase, also known as proto-oncogene c-RAF or simply c-Raf or even Raf-1, is an enzyme that in humans is encoded by the RAF1 gene. The c-Raf protein is part of the ERK1/2 pathway as a MAP kinase (MAP3K) that functions downstream of the Ras subfamily of membrane associated GTPases. C-Raf is a member of the Raf kinase family of serine/threonine-specific protein kinases, from the TKL (Tyrosine-kinase-like) group of kinases.

A serine/threonine protein kinase is a kinase enzyme that phosphorylates the OH group of serine or threonine. At least 125 of the 500+ human protein kinases are serine/threonine kinases (STK).
Cyclin G-associated kinase (GAK) is a serine/threonine kinase that in humans is encoded by the GAK gene.

BRAF is a human gene that encodes a protein called B-Raf. The gene is also referred to as proto-oncogene B-Raf and v-Raf murine sarcoma viral oncogene homolog B, while the protein is more formally known as serine/threonine-protein kinase B-Raf.

BAG family molecular chaperone regulator 1 is a protein that in humans is encoded by the BAG1 gene.

Serine/threonine-protein kinase A-Raf or simply A-Raf is an enzyme that in humans is encoded by the ARAF gene. A-Raf is a member of the Raf kinase family of serine/threonine-specific protein kinases.

BAG family molecular chaperone regulator 2 is a protein that in humans is encoded by the BAG2 gene.

BAG family molecular chaperone regulator 4 is a protein that in humans is encoded by the BAG4 gene.

BAG family molecular chaperone regulator 5 is a protein that in humans is encoded by the BAG5 gene.

Ankyrin repeat domain-containing protein 26 is a protein that in humans is encoded by the ANKRD26 gene. This protein has a function that is not currently understood.
The Walker A and Walker B motifs are protein sequence motifs, known to have highly conserved three-dimensional structures. These were first reported in ATP-binding proteins by Walker and co-workers in 1982.

Transmembrane protein 242 (TMEM242) is a protein that in humans is encoded by the TMEM242 gene. The tmem242 gene is located on chromosome 6, on the long arm, in band 2 section 5.3. This protein is also commonly called C6orf35, BM033, and UPF0463 Transmembrane Protein C6orf35. The tmem242 gene is 35,238 base pairs long, and the protein is 141 amino acids in length. The tmem242 gene contains 4 exons. The function of this protein is not well understood by the scientific community. This protein contains a DUF1358 domain.

EVI5L is a protein that in humans is encoded by the EVI5L gene. EVI5L is a member of the Ras superfamily of monomeric guanine nucleotide-binding (G) proteins, and functions as a GTPase-activating protein (GAP) with a broad specificity. Measurement of in vitro Rab-GAP activity has shown that EVI5L has significant Rab2A- and Rab10-GAP activity.

GrpE is a bacterial nucleotide exchange factor that is important for regulation of protein folding machinery, as well as the heat shock response. It is a heat-inducible protein and during stress it prevents unfolded proteins from accumulating in the cytoplasm. Accumulation of unfolded proteins in the cytoplasm can lead to cell death.

Serine/threonine-protein kinase RIO1 is an enzyme that in humans is encode by the RIOK1 gene.

















