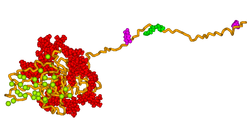
Osteopontin (OPN), also known as bone /sialoprotein I, early T-lymphocyte activation (ETA-1), secreted phosphoprotein 1 (SPP1), 2ar and Rickettsia resistance (Ric), is a protein that in humans is encoded by the SPP1 gene. The murine ortholog is Spp1. Osteopontin is a SIBLING (glycoprotein) that was first identified in 1986 in osteoblasts.

Pleiotrophin (PTN) also known as heparin-binding brain mitogen (HBBM) or heparin-binding growth factor 8 (HBGF-8) or neurite growth-promoting factor 1 (NEGF1) or heparin affinity regulatory peptide (HARP) or heparin binding growth associated molecule (HB-GAM) is a protein that in humans is encoded by the PTN gene. Pleiotrophin is an 18-kDa growth factor that has a high affinity for heparin. It is structurally related to midkine and retinoic acid induced heparin-binding protein.
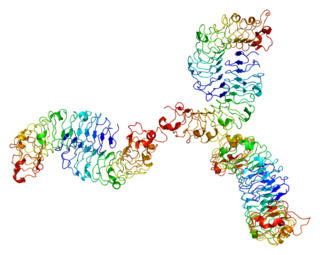
Biglycan is a small leucine-rich repeat proteoglycan (SLRP) which is found in a variety of extracellular matrix tissues, including bone, cartilage and tendon. In humans, biglycan is encoded by the BGN gene which is located on the X chromosome.

Osteonectin (ON) also known as secreted protein acidic and rich in cysteine (SPARC) or basement-membrane protein 40 (BM-40) is a protein that in humans is encoded by the SPARC gene.

Transforming growth factor-beta 2 (TGF-β2) is a secreted protein known as a cytokine that performs many cellular functions and has a vital role during embryonic development. It is an extracellular glycosylated protein. It is known to suppress the effects of interleukin dependent T-cell tumors. There are two named isoforms of this protein, created by alternative splicing of the same gene.
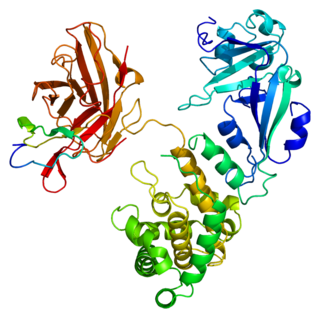
Heparin-binding EGF-like growth factor (HB-EGF) is a member of the EGF family of proteins that in humans is encoded by the HBEGF gene.

Y box binding protein 1 also known as Y-box transcription factor or nuclease-sensitive element-binding protein 1 is a protein that in humans is encoded by the YBX1 gene.

Glycylpeptide N-tetradecanoyltransferase 1 also known as myristoyl-CoA:protein N-myristoyltransferase 1 (NMT-1) is an enzyme that in humans is encoded by the NMT1 gene. It belongs to the protein N-terminal methyltransferase and glycylpeptide N-tetradecanoyltransferase family of enzymes.

Collagen alpha-2(IV) chain is a protein that in humans is encoded by the COL4A2 gene.

Laminin subunit alpha-1 is a protein that in humans is encoded by the LAMA1 gene.
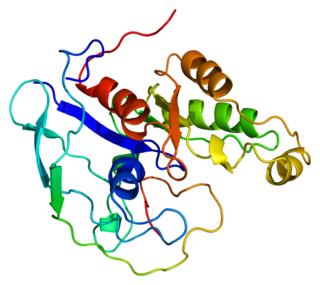
Activated CDC42 kinase 1, also known as ACK1, is an enzyme that in humans is encoded by the TNK2 gene. TNK2 gene encodes a non-receptor tyrosine kinase, ACK1, that binds to multiple receptor tyrosine kinases e.g. EGFR, MERTK, AXL, HER2 and insulin receptor (IR). ACK1 also interacts with Cdc42Hs in its GTP-bound form and inhibits both the intrinsic and GTPase-activating protein (GAP)-stimulated GTPase activity of Cdc42Hs. This binding is mediated by a unique sequence of 47 amino acids C-terminal to an SH3 domain. The protein may be involved in a regulatory mechanism that sustains the GTP-bound active form of Cdc42Hs and which is directly linked to a tyrosine phosphorylation signal transduction pathway. Several alternatively spliced transcript variants have been identified from this gene, but the full-length nature of only two transcript variants has been determined.

A disintegrin and metalloproteinase with thrombospondin motifs 1 is an enzyme that in humans is encoded by the ADAMTS1 gene.

Disintegrin and metalloproteinase domain-containing protein 9 is an enzyme that in humans is encoded by the ADAM9 gene.

Retinol binding protein 1, cellular, also known as RBP1, is a protein that in humans is encoded by the RBP1 gene.

Dentin matrix acidic phosphoprotein 1 is a protein that in humans is encoded by the DMP1 gene.

Sterile alpha motif and leucine zipper containing kinase AZK, also known as ZAK, is a human gene.

Hyaluronan synthase 1 is an enzyme that in humans is encoded by the HAS1 gene.

Hyaluronan synthase 3 is an enzyme that in humans is encoded by the HAS3 gene.

Neutrophil collagenase, also known as matrix metalloproteinase-8 (MMP-8) or PMNL collagenase (MNL-CL), is a collagen cleaving enzyme which is present in the connective tissue of most mammals. In humans, the MMP-8 protein is encoded by the MMP8 gene. The gene is part of a cluster of MMP genes which localize to chromosome 11q22.3. Most MMP's are secreted as inactive proproteins which are activated when cleaved by extracellular proteinases. However, the enzyme encoded by this gene is stored in secondary granules within neutrophils and is activated by autolytic cleavage.
Dentin sialophosphoprotein is a precursor protein for other proteins found in the teeth. It is produced by cells (odontoblasts) inside the teeth, and in smaller quantities by bone tissues. It is required for normal hardening (mineralisation) of teeth. During teeth development, it is broken down into three proteins such as dentin sialoprotein (DSP), dentin glycoprotein (DGP), and dentin phosphoprotein (DPP). These proteins become the major non-collagenous components of teeth. Their distribution in the collagen matrix of the forming dentin suggests these proteins play an important role in the regulation of mineral deposition. Additional evidence for this correlation is phenotypically manifested in patients with mutant forms of dentin sialophosphoprotein. Such patients suffer dental anomalies including type III dentinogenesis imperfecta.