 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
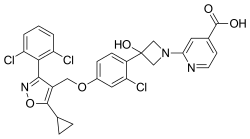
| Formula | C28H22Cl3N3O5 |
| Molar mass | 586.85 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Cilofexor (also known as GS-9674) is a nonsteroidal farnesoid X receptor (FXR) agonist in clinical trials for the treatment of non-alcoholic fatty liver disease (NAFLD), non-alcoholic steatohepatitis (NASH), [1] [2] [3] and primary sclerosing cholangitis (PSC). [4] [5] It is being investigated for use alone or in combination with firsocostat, selonsertib, [1] or semaglutide. [2] [6] In rat models [3] and human clinical trials [7] of NASH it has been shown to reduce fibrosis and steatosis, and in human clinical trials of PSC it improved cholestasis and reduced markers of liver injury. [4]
It is being developed by the pharmaceutical company Gilead Sciences. [8] [6]