Related Research Articles

Aspirin, also known as acetylsalicylic acid (ASA), is a nonsteroidal anti-inflammatory drug (NSAID) used to reduce pain, fever, and/or inflammation, and as an antithrombotic. Specific inflammatory conditions that aspirin is used to treat include Kawasaki disease, pericarditis, and rheumatic fever.

An antacid is a substance which neutralizes stomach acidity and is used to relieve heartburn, indigestion, or an upset stomach. Some antacids have been used in the treatment of constipation and diarrhea. Marketed antacids contain salts of aluminum, calcium, magnesium, or sodium. Some preparations contain a combination of two salts, such as magnesium carbonate and aluminum hydroxide.

A tablet is a pharmaceutical oral dosage form or solid unit dosage form. Tablets may be defined as the solid unit dosage form of medication with suitable excipients. It comprises a mixture of active substances and excipients, usually in powder form, that are pressed or compacted into a solid dose. The main advantages of tablets are that they ensure a consistent dose of medicine that is easy to consume.

In biochemistry, cellulose acetate refers to any acetate ester of cellulose, usually cellulose diacetate. It was first prepared in 1865. A bioplastic, cellulose acetate is used as a film base in photography, as a component in some coatings, and as a frame material for eyeglasses; it is also used as a synthetic fiber in the manufacture of cigarette filters and playing cards. In photographic film, cellulose acetate film replaced nitrate film in the 1950s, being far less flammable and cheaper to produce.
An excipient is a substance formulated alongside the active ingredient of a medication. They may be used to enhance the active ingredient’s therapeutic properties; to facilitate drug absorption; to reduce viscosity; to enhance solubility; to improve long-term stabilization ; or to add bulk to solid formulations that have small amounts of potent active ingredients. During the manufacturing process, excipients can improve the handling of active substances and facilitate powder flow. The choice of excipients depends on factors such as the intended route of administration, the dosage form, and compatibility with the active ingredient.
Microencapsulation is a process in which tiny particles or droplets are surrounded by a coating to give small capsules, with useful properties. In general, it is used to incorporate food ingredients, enzymes, cells or other materials on a micro metric scale. Microencapsulation can also be used to enclose solids, liquids, or gases inside a micrometric wall made of hard or soft soluble film, in order to reduce dosing frequency and prevent the degradation of pharmaceuticals.
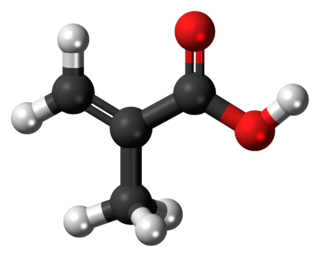
Methacrylic acid, abbreviated MAA, is an organic compound with the formula CH2=C(CH3)CO2H. This colorless, viscous liquid is a carboxylic acid with an acrid unpleasant odor. It is soluble in warm water and miscible with most organic solvents. Methacrylic acid is produced industrially on a large scale as a precursor to its esters, especially methyl methacrylate (MMA), and to poly(methyl methacrylate) (PMMA).

Dimethyl phthalate (DMP) is an organic compound and phthalate ester. it is a colourless and oily liquid that is soluble in organic solvents, but which is only poorly soluble in water.

Hypromellose (INN), short for hydroxypropyl methylcellulose (HPMC), is a semisynthetic, inert, viscoelastic polymer used in eye drops, as well as an excipient and controlled-delivery component in oral medicaments, found in a variety of commercial products.
Absorption is the journey of a drug travelling from the site of administration to the site of action.
Pharmaceutical formulation, in pharmaceutics, is the process in which different chemical substances, including the active drug, are combined to produce a final medicinal product. The word formulation is often used in a way that includes dosage form.

Cellulose acetate phthalate (CAP), also known as cellacefate (INN) and cellulosi acetas phthalas, is a commonly used polymer phthalate in the formulation of pharmaceuticals, such as the enteric coating of tablets or capsules and for controlled release formulations. It is a cellulose polymer where about half of the hydroxyls are esterified with acetyls, a quarter are esterified with one or two carboxyls of a phthalic acid, and the remainder are unchanged. It is a hygroscopic white to off-white free-flowing powder, granules, or flakes. It is tasteless and odorless, though may have a weak odor of acetic acid. Its main use in pharmaceutics is with enteric formulations. It can be used together with other coating agents, e.g. ethyl cellulose. Cellulose acetate phthalate is commonly plasticized with diethyl phthalate, a hydrophobic compound, or triethyl citrate, a hydrophilic compound; other compatible plasticizers are various phthalates, triacetin, dibutyl tartrate, glycerol, propylene glycol, tripropionin, triacetin citrate, acetylated monoglycerides, etc.
Modified-release dosage is a mechanism that delivers a drug with a delay after its administration or for a prolonged period of time or to a specific target in the body.

The gastric mucosal barrier is the property of the stomach that allows it to safely contain the gastric acid required for digestion.
Polyvinyl acetate phthalate (PVAP) is a commonly used polymer phthalate in the formulation of pharmaceuticals, such as the enteric coating of tablets or capsules. It is a vinyl acetate polymer that is partially hydrolyzed and then esterified with phthalic acid. Its main use in pharmaceutics is with enteric formulations and controlled release formulations.
Dissolving pulp, also called dissolving cellulose, is bleached wood pulp or cotton linters that has a high cellulose content. It has special properties including a high level of brightness and uniform molecular-weight distribution. This pulp is manufactured for uses that require a high chemical purity, and particularly low hemicellulose content, since the chemically similar hemicellulose can interfere with subsequent processes. Dissolving pulp is so named because it is not made into paper, but dissolved either in a solvent or by derivatization into a homogeneous solution, which makes it completely chemically accessible and removes any remaining fibrous structure. Once dissolved, it can be spun into textile fibers, or chemically reacted to produce derivatized celluloses, such cellulose triacetate, a plastic-like material formed into fibers or films, or cellulose ethers such as methyl cellulose, used as a thickener.

Effervescent or carbon tablets are tablets which are designed to dissolve in water and release carbon dioxide. The carbon dioxide is generated by a reaction of a compound containing bicarbonate, such as sodium bicarbonate or magnesium bicarbonate, with an acid such as citric acid or tartaric acid. Both compounds are present in the tablet in powder form and start reacting as soon as they dissolve in water.

The Poison Prevention Packaging Act of 1970 (PPPA); was signed into law by U.S. President Richard Nixon on December 30, 1970. It was enacted by the 91st United States Congress. This law required the use of child-resistant packaging for prescription drugs, over-the-counter (OTC) drugs, household chemicals, and other hazardous materials that could be considered dangerous for children.
A film coating is a thin polymer-based coat that is typically sprayed onto solid pharmaceutical dosage forms, such as tablets, capsules, pellets or granules. Film coating can impact both its appearance and its pharmacokinetics making it an essential process in making the final drug product.
References
- 1 2 Tarcha, Peter J. (1990). Polymers for Controlled Drug Delivery. CRC Press. ISBN 9780849356520.
- ↑ Bundgaard, Hans; Hansen, Anne Bagger; Kofod, Helmer (1982). Optimization of drug delivery: proceedings of the Alfred Benzon Symposium 17 held at the premises of the Royal Danish Academy of Sciences and Letters, Copenhagen 31 May-4 June 1981. Munksgaard. ISBN 9788716089793.
- 1 2 3 4 Wen, Hong; Park, Kinam (2011). Oral Controlled Release Formulation Design and Drug Delivery: Theory to Practice. John Wiley & Sons. ISBN 9781118060322.
- ↑ Juliano, R. L. (1980). Drug delivery systems: characteristics and biomedical applications. Oxford University Press. ISBN 9780195027006.
- ↑ Aulton, Michael; Cole, Graham; Hogan, John (1995). Pharmaceutical Coating Technology. Taylor & Francis. ISBN 9780136628910.
- ↑ Bukey, F. S.; Brew, Marjorie (1934). "A study of the emptying time of the stomach with reference to pills and tablets". Journal of the American Pharmaceutical Association. 23 (12): 1217–1219. doi:10.1002/jps.3080231216.
- ↑ Alpsten, M.; Bogentoft, C.; Ekenved, G.; Sölvell, L. (1982). "Gastric emptying and absorption of acetylsalicylic acid administered as enteric-coated micro-granules". European Journal of Clinical Pharmacology. 22 (1): 57–61. doi:10.1007/BF00606426. PMID 7094976. S2CID 5875048.
- 1 2 Kaniwa, N.; Aoyagi, N.; Ogata, H.; Ejima, A.; Motoyama, H.; Yasumi, H. (1988). "Gastric emptying rates of drug preparations. II. Effects of size and density of enteric-coated drug preparations and food on gastric emptying rates in humans". Journal of Pharmacobio-Dynamics. 11 (8): 571–575. doi: 10.1248/bpb1978.11.571 . PMID 3236215.
- ↑ Park, H. M.; Chernish, S. M.; Rosenek, B. D.; Brunelle, R. L.; Hargrove, B.; Wellman, H. N. (1984). "Gastric emptying of enteric-coated tablets". Digestive Diseases and Sciences. 29 (3): 207–212. doi:10.1007/bf01296253. PMID 6321117. S2CID 8366662.
- ↑ Maree, Andrew O.; Curtin, Ronan J.; Dooley, Michelle; Conroy, Ronan M.; Crean, Peter; Cox, Dermot; Fitzgerald, Desmond J. (2005-10-04). "Platelet response to low-dose enteric-coated aspirin in patients with stable cardiovascular disease". Journal of the American College of Cardiology. 46 (7): 1258–1263. doi:10.1016/j.jacc.2005.06.058. PMID 16198840.
- ↑ Patell, Mahesh K. (Oct 4, 1988), Enteric coated tablet and process for making , retrieved 2016-02-18
- ↑ Unna, Keratinirte Pillen, Pharm. Zentrahlle, 25, 577, 1884.
- ↑ Tbompsont, Herman O.; Lee, C.O. (1945). "History, literature, and theory of enteric coatings". Journal of the American Pharmaceutical Association (Scientific Ed.). 34 (5): 135–138. doi:10.1002/jps.3030340503.